| |
| Names | |
|---|---|
| Other names
Sodium trimethylsilanoate, Sodium trimethylsilanolate
| |
| Identifiers | |
| ECHA InfoCard | 100.038.112 |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| Properties | |
| C3H9NaOSi | |
| Molar mass | 112.179 g·mol−1 |
| Appearance | white solid |
| Density | 1.255 g/cm3 |
| Melting point | 147–150 °C (297–302 °F; 420–423 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
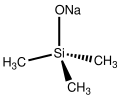
Sodium trimethylsiloxide is an organosilicon compound with the formula NaOSi(CH3)3. It is the sodium salt of the conjugate base derived from trimethylsilanol. [1] A white solid, its molecular structure consists of a cluster with Na-O-Na linkages on the basis of closely related compounds. [2]
The salt is used to prepare trimethylsiloxide complexes by salt metathesis. Trimethylsiloxide is a lipophilic pseudohalide. [3] [4]
It is a source of oxide dianion. [5] [6]
- Sodium silox, NaOSi(tBu)3 (tBu = C(CH3)3)
- Potassium trimethylsilanolate [7]
- ^ Sommer, L. H.; Pietrusza, E. W.; Whitmore, F. C. (1946). "Properties of the Silicon—Hydroxyl Bond in Trialkylsilanols1". Journal of the American Chemical Society. 68 (11): 2282–2284. doi: 10.1021/ja01215a047.
- ^ Mehring, Michael; Nolde, Christof; Schürmann, Markus (2004). "Crystallographic Report: A polymorph of Undecasodium Decatrimethylsilanolate Hydroxide:[Na11(OSiMe3)10(OH)]". Applied Organometallic Chemistry. 18 (9): 489–490. doi: 10.1002/aoc.684.
- ^ Krempner, Clemens (2011). "Role of Siloxides in Transition Metal Chemistry and Homogeneous Catalysis". European Journal of Inorganic Chemistry. 2011 (11): 1689–1698. doi: 10.1002/ejic.201100044.
- ^ Chisholm, Malcolm H.; Eilerts, Nancy W.; Huffman, John C.; Iyer, Suri S.; Pacold, Martha; Phomphrai, Khamphee (2000). "Polylactide Formation by Achiral and Chiral Magnesium and Zinc Alkoxides, (η3-L)MOR, Where L = Trispyrazolyl- and Trisindazolylborate Ligands". Journal of the American Chemical Society. 122 (48): 11845–11854. doi: 10.1021/ja002160g.
- ^ Do, Y.; Simhon, E. D.; Holm, R. H. (1983). "Improved Syntheses of Tetrachlorodi-μ-sulfidodiferrate Dianion ([Fe2S2Cl42-) and Hexachloro-μ-oxodiferrate2- ([Fe2OCl62-) and Oxo/Sulfido Ligand Substitution by Use of Silylsulfide Reagents". Inorg. Chem. 22: 3809-12. doi: 10.1021/ic00167a027.
- ^ Laganis, E. D.; Chenard, B. L. (1984). "Metal Silanolates: Organic Soluble Equivalents for O−2". Tetrahedron Letters. 25 (51): 5831–5834. doi: 10.1016/S0040-4039(01)81697-X.
- ^ Delaney, Connor P.; Heyboer, E. M.; Denmark, S. E. (2020). "Anhydrous, Homogeneous, Suzuki-Miyaura Cross-Coupling of Boronic Esters using Potassium Trimethylsilanolate". Organic Syntheses. 97: 245–261. doi: 10.15227/orgsyn.097.0245. PMC 7808858. PMID 33456091.

| |
| Names | |
|---|---|
| Other names
Sodium trimethylsilanoate, Sodium trimethylsilanolate
| |
| Identifiers | |
| ECHA InfoCard | 100.038.112 |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| Properties | |
| C3H9NaOSi | |
| Molar mass | 112.179 g·mol−1 |
| Appearance | white solid |
| Density | 1.255 g/cm3 |
| Melting point | 147–150 °C (297–302 °F; 420–423 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium trimethylsiloxide is an organosilicon compound with the formula NaOSi(CH3)3. It is the sodium salt of the conjugate base derived from trimethylsilanol. [1] A white solid, its molecular structure consists of a cluster with Na-O-Na linkages on the basis of closely related compounds. [2]
The salt is used to prepare trimethylsiloxide complexes by salt metathesis. Trimethylsiloxide is a lipophilic pseudohalide. [3] [4]
It is a source of oxide dianion. [5] [6]
- Sodium silox, NaOSi(tBu)3 (tBu = C(CH3)3)
- Potassium trimethylsilanolate [7]
- ^ Sommer, L. H.; Pietrusza, E. W.; Whitmore, F. C. (1946). "Properties of the Silicon—Hydroxyl Bond in Trialkylsilanols1". Journal of the American Chemical Society. 68 (11): 2282–2284. doi: 10.1021/ja01215a047.
- ^ Mehring, Michael; Nolde, Christof; Schürmann, Markus (2004). "Crystallographic Report: A polymorph of Undecasodium Decatrimethylsilanolate Hydroxide:[Na11(OSiMe3)10(OH)]". Applied Organometallic Chemistry. 18 (9): 489–490. doi: 10.1002/aoc.684.
- ^ Krempner, Clemens (2011). "Role of Siloxides in Transition Metal Chemistry and Homogeneous Catalysis". European Journal of Inorganic Chemistry. 2011 (11): 1689–1698. doi: 10.1002/ejic.201100044.
- ^ Chisholm, Malcolm H.; Eilerts, Nancy W.; Huffman, John C.; Iyer, Suri S.; Pacold, Martha; Phomphrai, Khamphee (2000). "Polylactide Formation by Achiral and Chiral Magnesium and Zinc Alkoxides, (η3-L)MOR, Where L = Trispyrazolyl- and Trisindazolylborate Ligands". Journal of the American Chemical Society. 122 (48): 11845–11854. doi: 10.1021/ja002160g.
- ^ Do, Y.; Simhon, E. D.; Holm, R. H. (1983). "Improved Syntheses of Tetrachlorodi-μ-sulfidodiferrate Dianion ([Fe2S2Cl42-) and Hexachloro-μ-oxodiferrate2- ([Fe2OCl62-) and Oxo/Sulfido Ligand Substitution by Use of Silylsulfide Reagents". Inorg. Chem. 22: 3809-12. doi: 10.1021/ic00167a027.
- ^ Laganis, E. D.; Chenard, B. L. (1984). "Metal Silanolates: Organic Soluble Equivalents for O−2". Tetrahedron Letters. 25 (51): 5831–5834. doi: 10.1016/S0040-4039(01)81697-X.
- ^ Delaney, Connor P.; Heyboer, E. M.; Denmark, S. E. (2020). "Anhydrous, Homogeneous, Suzuki-Miyaura Cross-Coupling of Boronic Esters using Potassium Trimethylsilanolate". Organic Syntheses. 97: 245–261. doi: 10.15227/orgsyn.097.0245. PMC 7808858. PMID 33456091.