| |||
| Names | |||
|---|---|---|---|
| Other names
Sodium cobalticarborane
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| EC Number |
| ||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H22B18CoNa | |||
| Molar mass | 346.72 g·mol−1 | ||
| Appearance | Red crystals or yellow powder | ||
| Melting point | >300°C | ||
| Hazards | |||
| GHS labelling: [1] | |||

| |||
| Warning | |||
| H315, H319, H335 | |||
| P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 | |||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
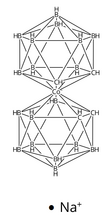
Dicarbollylcobaltate(III) anion is a dicarbollide cluster compound containing cobaltic cation (III) as a metal center. The dicarbollylcobaltate(III) anion can be abbreviated to [COSAN]− or [CoD]−. The center cobaltic cation is sandwiched by two dicarbollide clusters, [2] so that it can be regarded as the carboranyl version of Cp2Co+.
The countercation of dicarbollylcobaltate(III) could be Na+, [3][ failed verification] Cs+, [4] H+, [5] NH4+, [6] and other transition and main group metals. Among them, Na+ is the most commonly used cation.
Synthesis
Sodium dicarbollylcobaltate could be synthesized from o-carborane. Lewis bases like butylamine and sodium hydroxide could attack the boron that is connected by two carbon atoms and subsequently remove this boron atom from the carborane skeleton, affording sodium 7,8-dicarbaundecaborate(1-).

Then, two equivalents of sodium 7,8-dicarbaundecaborate could react with cobalt acetate tetrahydrate at high temperature, leading the formation of one equivlent of sodium acetate and one equivlent of sodium dicarbollylcobaltate.

The synthesis of sodium dicarbollylcobaltate could also be accomplished by ion exchange. First, the aqueous solution of the cesium salt could be acidified by sulfuric acid and be extracted in organic phase. Then using sodium carbonate as sodium source to react with acidified dicarbollylcobaltate to get the sodium dicarbaundecaborate. [2] [7] [8] [9][ failed verification]

Reactivity
The reactivity of the B-H bond in dicarbollylcobaltate is similar to that in o-carborane compounds. Until now, dicarbollylcobaltate anion has mostly been used as a spectator ion in the fields of catalytic chemistry, structural chemistry, and supramolecular chemistry. However, there is an example of the functionalization of the dicarbollylcobaltate anion. [10]
The preparation of iodonium zwitterions of dicarbollylcobaltate could be accomplished by the reaction of dicarbollylcobaltate with ArI(OAc)2 (Ar = Ph and 4-MeOC6H4) in 70% aqueous AcOH or CF3COOH.

The iodonium zwitterions could subsequently transform to other functionalized dicarbollylcobaltate by adding the appropriate nucleophiles. The reaction of the iodonium zwitterions with Me2NCHS and NEt4+CN− lead to corresponding closo-borate derivatives.

Applications
Dicarbollylcobaltate anion is stable enough in common organic solvent and water. Because of the uniqueness of its hydridic B-H vertices and charge delocalization, the [CoD]− anion exhibits surface activity and has an amphiphilic character. [11] [12] [CoD]- could be also used in radioactive ion extraction. [13] Due to the abundant boron atoms of [CoD]−, it also could be used in biomedical chemistry. [12] ( Boron neutron capture therapy is an important targeted radiation cancer therapy) Besides, because [CoD]− accumulates at interfaces and is soluble in both oil and water, [CoD]− could be also used in self-assembly in water. [14]
Structural analysis
The 1,2-B9C2H112- ion could be regarded as a ligand donating 6 electrons. The two carbon atoms and three boron atoms have approximately sp3 orbitals directed at the vacant 3-vertex. Therefore, with cobalt(III) as the metal center and a negative charge, the ion satisfies the 18-electron rule.



References
- ^ "Sodium cobalticarborane". pubchem.ncbi.nlm.nih.gov.
- ^ a b c Hawthorne, M. Frederick; Young, Donald Clifford; Andrews, Timothy D.; Howe, David V.; Pilling, Richard L.; Pitts, A. Denise; Reintjes, Marten; Warren, Leslie F.; Wegner, Patrick A. (February 1968). ".pi.-Dicarbollyl derivatives of the transition metals. Metallocene analogs". Journal of the American Chemical Society. 90 (4): 879–896. doi: 10.1021/ja01006a008. ISSN 0002-7863.
- ^ Schweiger, Manuela; Seidel, S. Russell; Arif, Atta M.; Stang, Peter J. (2002-05-01). "Solution and Solid State Studies of a Triangle−Square Equilibrium: Anion-Induced Selective Crystallization in Supramolecular Self-Assembly". Inorganic Chemistry. 41 (9): 2556–2559. doi: 10.1021/ic0112692. ISSN 0020-1669. PMID 11978126.
- ^ González-Cardoso, Patricia; Stoica, Anca-Iulia; Farràs, Pau; Pepiol, Ariadna; Viñas, Clara; Teixidor, Francesc (2010-06-11). "Additive Tuning of Redox Potential in Metallacarboranes by Sequential Halogen Substitution". Chemistry - A European Journal. 16 (22): 6660–6665. doi: 10.1002/chem.200902558. hdl: 10379/5951. PMID 20411531.
- ^ Tarrés, Màrius; Arderiu, Víctor S.; Zaulet, Adnana; Viñas, Clara; Biani, Fabrizia Fabrizi de; Teixidor, Francesc (2015-06-23). "How to get the desired reduction voltage in a single framework! Metallacarborane as an optimal probe for sequential voltage tuning". Dalton Transactions. 44 (26): 11690–11695. doi: 10.1039/C5DT01464F. hdl: 11365/983458. ISSN 1477-9234. PMID 26054738.
- ^ Makrlík, E. (2002-08-01). "Solvent extraction of barium into nitrobenzene by using ammonium dicarbollylcobaltate in the presence of 15-crown-5". Journal of Radioanalytical and Nuclear Chemistry. 253 (2): 327–329. doi: 10.1023/A:1019678615764. ISSN 1588-2780. S2CID 93764779.
- ^ Rak, Jakub; Jakubek, Milan; Kaplánek, Robert; Král, Vladimír (2012-04-02). "Low-Melting Salts Based on a Glycolated Cobalt Bis(dicarbollide) Anion". Inorganic Chemistry. 51 (7): 4099–4107. doi: 10.1021/ic2023665. ISSN 0020-1669. PMID 22432551.
- ^ Guerrero, Isabel; Viñas, Clara; Romero, Isabel; Teixidor, Francesc (2021-12-13). "A stand-alone cobalt bis(dicarbollide) photoredox catalyst epoxidates alkenes in water at extremely low catalyst load". Green Chemistry. 23 (24): 10123–10131. doi: 10.1039/D1GC03119H. hdl: 10256/20224. ISSN 1463-9270. S2CID 244096222.
- ^ Vrbata, David; Ďorďovič, Vladimír; Seitsonen, Jani; Ruokolainen, Janne; Janoušková, Olga; Uchman, Mariusz; Matějíček, Pavel (2019-03-05). "Preparation of membrane-mimicking lamellar structures by molecular confinement of hybrid nanocomposites". Chemical Communications. 55 (20): 2900–2903. doi: 10.1039/C8CC09399G. ISSN 1364-548X. PMID 30698594. S2CID 206128749.
- ^ Kaszyński, Piotr; Ringstrand, Bryan (2015-05-26). "Functionalization of closo -Borates via Iodonium Zwitterions". Angewandte Chemie International Edition. 54 (22): 6576–6581. doi: 10.1002/anie.201411858. PMID 25877873.
- ^ Chevrot, G.; Schurhammer, R.; Wipff, G. (2006-04-22). "Surfactant Behavior of "Ellipsoidal" Dicarbollide Anions: A Molecular Dynamics Study". The Journal of Physical Chemistry B. 110 (19): 9488–9498. doi: 10.1021/jp060930q. ISSN 1520-6106. PMID 16686495.
- ^ a b Gassin, Pierre-Marie; Girard, Luc; Martin-Gassin, Gaelle; Brusselle, Damien; Jonchère, Alban; Diat, Olivier; Viñas, Clara; Teixidor, Francesc; Bauduin, Pierre (2015-02-16). "Surface Activity and Molecular Organization of Metallacarboranes at the Air–Water Interface Revealed by Nonlinear Optics". Langmuir. 31 (8): 2297–2303. doi: 10.1021/acs.langmuir.5b00125. ISSN 0743-7463. PMID 25644035.
- ^ Hosmane, Narayan S., ed. (2016-04-19). Boron Science. CRC Press. doi: 10.1201/b11199. ISBN 978-0-429-06273-5.
- ^ Uchman, Mariusz; Ďorďovič, Vladimír; Tošner, Zdeněk; Matějíček, Pavel (2015-11-16). "Classical Amphiphilic Behavior of Nonclassical Amphiphiles: A Comparison of Metallacarborane Self-Assembly with SDS Micellization". Angewandte Chemie International Edition. 54 (47): 14113–14117. doi: 10.1002/anie.201506545. PMID 26425966.
| |||
| Names | |||
|---|---|---|---|
| Other names
Sodium cobalticarborane
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| EC Number |
| ||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H22B18CoNa | |||
| Molar mass | 346.72 g·mol−1 | ||
| Appearance | Red crystals or yellow powder | ||
| Melting point | >300°C | ||
| Hazards | |||
| GHS labelling: [1] | |||

| |||
| Warning | |||
| H315, H319, H335 | |||
| P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 | |||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Dicarbollylcobaltate(III) anion is a dicarbollide cluster compound containing cobaltic cation (III) as a metal center. The dicarbollylcobaltate(III) anion can be abbreviated to [COSAN]− or [CoD]−. The center cobaltic cation is sandwiched by two dicarbollide clusters, [2] so that it can be regarded as the carboranyl version of Cp2Co+.
The countercation of dicarbollylcobaltate(III) could be Na+, [3][ failed verification] Cs+, [4] H+, [5] NH4+, [6] and other transition and main group metals. Among them, Na+ is the most commonly used cation.
Synthesis
Sodium dicarbollylcobaltate could be synthesized from o-carborane. Lewis bases like butylamine and sodium hydroxide could attack the boron that is connected by two carbon atoms and subsequently remove this boron atom from the carborane skeleton, affording sodium 7,8-dicarbaundecaborate(1-).

Then, two equivalents of sodium 7,8-dicarbaundecaborate could react with cobalt acetate tetrahydrate at high temperature, leading the formation of one equivlent of sodium acetate and one equivlent of sodium dicarbollylcobaltate.

The synthesis of sodium dicarbollylcobaltate could also be accomplished by ion exchange. First, the aqueous solution of the cesium salt could be acidified by sulfuric acid and be extracted in organic phase. Then using sodium carbonate as sodium source to react with acidified dicarbollylcobaltate to get the sodium dicarbaundecaborate. [2] [7] [8] [9][ failed verification]

Reactivity
The reactivity of the B-H bond in dicarbollylcobaltate is similar to that in o-carborane compounds. Until now, dicarbollylcobaltate anion has mostly been used as a spectator ion in the fields of catalytic chemistry, structural chemistry, and supramolecular chemistry. However, there is an example of the functionalization of the dicarbollylcobaltate anion. [10]
The preparation of iodonium zwitterions of dicarbollylcobaltate could be accomplished by the reaction of dicarbollylcobaltate with ArI(OAc)2 (Ar = Ph and 4-MeOC6H4) in 70% aqueous AcOH or CF3COOH.

The iodonium zwitterions could subsequently transform to other functionalized dicarbollylcobaltate by adding the appropriate nucleophiles. The reaction of the iodonium zwitterions with Me2NCHS and NEt4+CN− lead to corresponding closo-borate derivatives.

Applications
Dicarbollylcobaltate anion is stable enough in common organic solvent and water. Because of the uniqueness of its hydridic B-H vertices and charge delocalization, the [CoD]− anion exhibits surface activity and has an amphiphilic character. [11] [12] [CoD]- could be also used in radioactive ion extraction. [13] Due to the abundant boron atoms of [CoD]−, it also could be used in biomedical chemistry. [12] ( Boron neutron capture therapy is an important targeted radiation cancer therapy) Besides, because [CoD]− accumulates at interfaces and is soluble in both oil and water, [CoD]− could be also used in self-assembly in water. [14]
Structural analysis
The 1,2-B9C2H112- ion could be regarded as a ligand donating 6 electrons. The two carbon atoms and three boron atoms have approximately sp3 orbitals directed at the vacant 3-vertex. Therefore, with cobalt(III) as the metal center and a negative charge, the ion satisfies the 18-electron rule.



References
- ^ "Sodium cobalticarborane". pubchem.ncbi.nlm.nih.gov.
- ^ a b c Hawthorne, M. Frederick; Young, Donald Clifford; Andrews, Timothy D.; Howe, David V.; Pilling, Richard L.; Pitts, A. Denise; Reintjes, Marten; Warren, Leslie F.; Wegner, Patrick A. (February 1968). ".pi.-Dicarbollyl derivatives of the transition metals. Metallocene analogs". Journal of the American Chemical Society. 90 (4): 879–896. doi: 10.1021/ja01006a008. ISSN 0002-7863.
- ^ Schweiger, Manuela; Seidel, S. Russell; Arif, Atta M.; Stang, Peter J. (2002-05-01). "Solution and Solid State Studies of a Triangle−Square Equilibrium: Anion-Induced Selective Crystallization in Supramolecular Self-Assembly". Inorganic Chemistry. 41 (9): 2556–2559. doi: 10.1021/ic0112692. ISSN 0020-1669. PMID 11978126.
- ^ González-Cardoso, Patricia; Stoica, Anca-Iulia; Farràs, Pau; Pepiol, Ariadna; Viñas, Clara; Teixidor, Francesc (2010-06-11). "Additive Tuning of Redox Potential in Metallacarboranes by Sequential Halogen Substitution". Chemistry - A European Journal. 16 (22): 6660–6665. doi: 10.1002/chem.200902558. hdl: 10379/5951. PMID 20411531.
- ^ Tarrés, Màrius; Arderiu, Víctor S.; Zaulet, Adnana; Viñas, Clara; Biani, Fabrizia Fabrizi de; Teixidor, Francesc (2015-06-23). "How to get the desired reduction voltage in a single framework! Metallacarborane as an optimal probe for sequential voltage tuning". Dalton Transactions. 44 (26): 11690–11695. doi: 10.1039/C5DT01464F. hdl: 11365/983458. ISSN 1477-9234. PMID 26054738.
- ^ Makrlík, E. (2002-08-01). "Solvent extraction of barium into nitrobenzene by using ammonium dicarbollylcobaltate in the presence of 15-crown-5". Journal of Radioanalytical and Nuclear Chemistry. 253 (2): 327–329. doi: 10.1023/A:1019678615764. ISSN 1588-2780. S2CID 93764779.
- ^ Rak, Jakub; Jakubek, Milan; Kaplánek, Robert; Král, Vladimír (2012-04-02). "Low-Melting Salts Based on a Glycolated Cobalt Bis(dicarbollide) Anion". Inorganic Chemistry. 51 (7): 4099–4107. doi: 10.1021/ic2023665. ISSN 0020-1669. PMID 22432551.
- ^ Guerrero, Isabel; Viñas, Clara; Romero, Isabel; Teixidor, Francesc (2021-12-13). "A stand-alone cobalt bis(dicarbollide) photoredox catalyst epoxidates alkenes in water at extremely low catalyst load". Green Chemistry. 23 (24): 10123–10131. doi: 10.1039/D1GC03119H. hdl: 10256/20224. ISSN 1463-9270. S2CID 244096222.
- ^ Vrbata, David; Ďorďovič, Vladimír; Seitsonen, Jani; Ruokolainen, Janne; Janoušková, Olga; Uchman, Mariusz; Matějíček, Pavel (2019-03-05). "Preparation of membrane-mimicking lamellar structures by molecular confinement of hybrid nanocomposites". Chemical Communications. 55 (20): 2900–2903. doi: 10.1039/C8CC09399G. ISSN 1364-548X. PMID 30698594. S2CID 206128749.
- ^ Kaszyński, Piotr; Ringstrand, Bryan (2015-05-26). "Functionalization of closo -Borates via Iodonium Zwitterions". Angewandte Chemie International Edition. 54 (22): 6576–6581. doi: 10.1002/anie.201411858. PMID 25877873.
- ^ Chevrot, G.; Schurhammer, R.; Wipff, G. (2006-04-22). "Surfactant Behavior of "Ellipsoidal" Dicarbollide Anions: A Molecular Dynamics Study". The Journal of Physical Chemistry B. 110 (19): 9488–9498. doi: 10.1021/jp060930q. ISSN 1520-6106. PMID 16686495.
- ^ a b Gassin, Pierre-Marie; Girard, Luc; Martin-Gassin, Gaelle; Brusselle, Damien; Jonchère, Alban; Diat, Olivier; Viñas, Clara; Teixidor, Francesc; Bauduin, Pierre (2015-02-16). "Surface Activity and Molecular Organization of Metallacarboranes at the Air–Water Interface Revealed by Nonlinear Optics". Langmuir. 31 (8): 2297–2303. doi: 10.1021/acs.langmuir.5b00125. ISSN 0743-7463. PMID 25644035.
- ^ Hosmane, Narayan S., ed. (2016-04-19). Boron Science. CRC Press. doi: 10.1201/b11199. ISBN 978-0-429-06273-5.
- ^ Uchman, Mariusz; Ďorďovič, Vladimír; Tošner, Zdeněk; Matějíček, Pavel (2015-11-16). "Classical Amphiphilic Behavior of Nonclassical Amphiphiles: A Comparison of Metallacarborane Self-Assembly with SDS Micellization". Angewandte Chemie International Edition. 54 (47): 14113–14117. doi: 10.1002/anie.201506545. PMID 26425966.