| |
| Names | |
|---|---|
|
IUPAC name
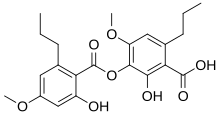
2-Hydroxy-3-(2-hydroxy-4-methoxy-6-propylbenzoyl)oxy-4-methoxy-6-propylbenzoic acid
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C22H26O8 | |
| Molar mass | 418.442 g·mol−1 |
| Appearance | Rectangular prisms or rhombic plates |
| Melting point | 150–151 °C (302–304 °F; 423–424 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Sekikaic acid is an organic compound in the structural class of chemicals known as depsides. It is found in some lichens. First isolated from Ramalina sekika, it is a fairly common lichen product in Ramalina and Cladonia, both genera of lichen-forming fungi. [1] The species epithet of the powdery lichen Lepraria sekikaica refers to the presence of this substance—a rarity in genus Lepraria. [2]
Properties
In its purified form, sekikaic acid exists as colourless rectangular prisms or rhombic plates. Its molecular formula is C22H2608. It has a melting point of 150–151 °C (302–304 °F). An ethanolic solution of sekikaic acid reacts with iron(III) chloride to produce a violet colour. Its ultraviolet spectrum has three peaks of maximum absorption (λmax) at 219, 263, and 303 nm. [3]
Sekikaic acid has been demonstrated to have several biological activities in laboratory experiments. These include antioxidant activity, [4] inhibition of the enzymes α-glucosidase and α-amylase, hypoglycemic activity, and lipid-lowering activity. [5] It also has antiviral activity against Respiratory syncytial virus, even more so than the standard antiviral medication ribavirin. [6]
Related compounds
The sekikaic acid chemosyndrome contains similar compounds that are metabolically related to sekikaic acid. It comprises sekikaic acid as the major compound, and 4'O-demethylsekikaic and homosekikaic acids as satellite metabolites. [7]
References
- ^ Culberson, Chicita (1970). "Supplement to "Chemical and Botanical Guide to Lichen Products"". The Bryologist. 73 (2): 177–377. doi: 10.2307/3241261. JSTOR 3241261.
- ^ Lumbsch, H.T.; Ahti, T.; Altermann, S.; De Paz, G.A.; Aptroot, A.; Arup, U.; et al. (2011). "One hundred new species of lichenized fungi: a signature of undiscovered global diversity". Phytotaxa. 18 (1): 81–82. doi: 10.11646/phytotaxa.18.1.1. hdl: 11336/4198.
- ^ Huneck, Siegfried (1996). Identification of Lichen Substances. Berlin, Heidelberg: Springer Berlin Heidelberg. pp. 49, 115, 296–297. ISBN 978-3-642-85245-9. OCLC 851387266.
- ^ Sisodia, R.; Geol, M.; Verma, S.; Rani, A.; Dureja, P. (2013). "Antibacterial and antioxidant activity of lichen species Ramalina roesleri". Natural Product Research. 27 (23): 2235–2239. doi: 10.1080/14786419.2013.811410. PMID 23822758.
- ^ Tatipamula, Vinay Bharadwaj; Annam, Satya Sowbhagya Priya; Nguyen, Ha Thi; Polimati, Haritha; Yejella, Rajendra Prasad (2020). "Sekikaic acid modulates pancreatic β-cells in streptozotocin-induced type 2 diabetic rats by inhibiting digestive enzymes". Natural Product Research. 35 (23): 5420–5424. doi: 10.1080/14786419.2020.1775226. PMID 32498563.
- ^ Lai, Daowan; Odimegwu, Damian; Esimone, Charles; Grunwald, Thomas; Proksch, Peter (2013). "Phenolic compounds with in vitro activity against Respiratory Syncytial Virus from the Nigerian lichen Ramalina farinacea". Planta Medica. 79 (15): 1440–1446. doi: 10.1055/s-0033-1350711. PMID 23970423.
- ^ Cordeiro, Lucimara M.C.; Iacomini, Marcello; Stocker-Wörgötter, Elfie (2004). "Culture studies and secondary compounds of six Ramalina species". Mycological Research. 108 (5): 489–497. doi: 10.1017/s0953756204009402. PMID 15230001.

| |
| Names | |
|---|---|
|
IUPAC name
2-Hydroxy-3-(2-hydroxy-4-methoxy-6-propylbenzoyl)oxy-4-methoxy-6-propylbenzoic acid
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C22H26O8 | |
| Molar mass | 418.442 g·mol−1 |
| Appearance | Rectangular prisms or rhombic plates |
| Melting point | 150–151 °C (302–304 °F; 423–424 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Sekikaic acid is an organic compound in the structural class of chemicals known as depsides. It is found in some lichens. First isolated from Ramalina sekika, it is a fairly common lichen product in Ramalina and Cladonia, both genera of lichen-forming fungi. [1] The species epithet of the powdery lichen Lepraria sekikaica refers to the presence of this substance—a rarity in genus Lepraria. [2]
Properties
In its purified form, sekikaic acid exists as colourless rectangular prisms or rhombic plates. Its molecular formula is C22H2608. It has a melting point of 150–151 °C (302–304 °F). An ethanolic solution of sekikaic acid reacts with iron(III) chloride to produce a violet colour. Its ultraviolet spectrum has three peaks of maximum absorption (λmax) at 219, 263, and 303 nm. [3]
Sekikaic acid has been demonstrated to have several biological activities in laboratory experiments. These include antioxidant activity, [4] inhibition of the enzymes α-glucosidase and α-amylase, hypoglycemic activity, and lipid-lowering activity. [5] It also has antiviral activity against Respiratory syncytial virus, even more so than the standard antiviral medication ribavirin. [6]
Related compounds
The sekikaic acid chemosyndrome contains similar compounds that are metabolically related to sekikaic acid. It comprises sekikaic acid as the major compound, and 4'O-demethylsekikaic and homosekikaic acids as satellite metabolites. [7]
References
- ^ Culberson, Chicita (1970). "Supplement to "Chemical and Botanical Guide to Lichen Products"". The Bryologist. 73 (2): 177–377. doi: 10.2307/3241261. JSTOR 3241261.
- ^ Lumbsch, H.T.; Ahti, T.; Altermann, S.; De Paz, G.A.; Aptroot, A.; Arup, U.; et al. (2011). "One hundred new species of lichenized fungi: a signature of undiscovered global diversity". Phytotaxa. 18 (1): 81–82. doi: 10.11646/phytotaxa.18.1.1. hdl: 11336/4198.
- ^ Huneck, Siegfried (1996). Identification of Lichen Substances. Berlin, Heidelberg: Springer Berlin Heidelberg. pp. 49, 115, 296–297. ISBN 978-3-642-85245-9. OCLC 851387266.
- ^ Sisodia, R.; Geol, M.; Verma, S.; Rani, A.; Dureja, P. (2013). "Antibacterial and antioxidant activity of lichen species Ramalina roesleri". Natural Product Research. 27 (23): 2235–2239. doi: 10.1080/14786419.2013.811410. PMID 23822758.
- ^ Tatipamula, Vinay Bharadwaj; Annam, Satya Sowbhagya Priya; Nguyen, Ha Thi; Polimati, Haritha; Yejella, Rajendra Prasad (2020). "Sekikaic acid modulates pancreatic β-cells in streptozotocin-induced type 2 diabetic rats by inhibiting digestive enzymes". Natural Product Research. 35 (23): 5420–5424. doi: 10.1080/14786419.2020.1775226. PMID 32498563.
- ^ Lai, Daowan; Odimegwu, Damian; Esimone, Charles; Grunwald, Thomas; Proksch, Peter (2013). "Phenolic compounds with in vitro activity against Respiratory Syncytial Virus from the Nigerian lichen Ramalina farinacea". Planta Medica. 79 (15): 1440–1446. doi: 10.1055/s-0033-1350711. PMID 23970423.
- ^ Cordeiro, Lucimara M.C.; Iacomini, Marcello; Stocker-Wörgötter, Elfie (2004). "Culture studies and secondary compounds of six Ramalina species". Mycological Research. 108 (5): 489–497. doi: 10.1017/s0953756204009402. PMID 15230001.