| |
| |
| Names | |
|---|---|
|
IUPAC name
Scandium trifluoromethanesulfonate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.157.499 |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C3F9O9S3Sc | |
| Molar mass | 492.16 g/mol |
| Hazards | |
| Safety data sheet (SDS) | Oxford MSDS |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Scandium trifluoromethanesulfonate, commonly called scandium triflate, is a
chemical compound with formula Sc(SO3CF3)3, a
salt consisting of
scandium
cations Sc3+ and
triflate SO
3CF−
3
anions.
Scandium triflate is used as a reagent in organic chemistry as a Lewis acid. [1] Compared to other Lewis acids, this reagent is stable towards water and can often be used in organic reactions as a true catalyst rather than one used in stoichiometric amounts. The compound is prepared by reaction of scandium oxide with trifluoromethanesulfonic acid.
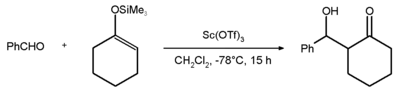
An example of the scientific use of scandium triflate is the Mukaiyama aldol addition reaction between benzaldehyde and the silyl enol ether of cyclohexanone with an 81% yield. [2]
See also
References
- ^ Deborah Longbottom (1999). "SYNLETT Spotlight 12: Scandium Triflate". Synlett. 1999 (12): 2023. doi: 10.1055/s-1999-5997.
- ^ S. Kobayashi (1999). "Scandium Triflate in Organic Synthesis". Eur. J. Org. Chem. 1999: 15–27. doi: 10.1002/(SICI)1099-0690(199901)1999:1<15::AID-EJOC15>3.0.CO;2-B. Archived from the original on 2013-01-05.

| |

| |
| Names | |
|---|---|
|
IUPAC name
Scandium trifluoromethanesulfonate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.157.499 |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C3F9O9S3Sc | |
| Molar mass | 492.16 g/mol |
| Hazards | |
| Safety data sheet (SDS) | Oxford MSDS |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Scandium trifluoromethanesulfonate, commonly called scandium triflate, is a
chemical compound with formula Sc(SO3CF3)3, a
salt consisting of
scandium
cations Sc3+ and
triflate SO
3CF−
3
anions.
Scandium triflate is used as a reagent in organic chemistry as a Lewis acid. [1] Compared to other Lewis acids, this reagent is stable towards water and can often be used in organic reactions as a true catalyst rather than one used in stoichiometric amounts. The compound is prepared by reaction of scandium oxide with trifluoromethanesulfonic acid.
An example of the scientific use of scandium triflate is the Mukaiyama aldol addition reaction between benzaldehyde and the silyl enol ether of cyclohexanone with an 81% yield. [2]
See also
References
- ^ Deborah Longbottom (1999). "SYNLETT Spotlight 12: Scandium Triflate". Synlett. 1999 (12): 2023. doi: 10.1055/s-1999-5997.
- ^ S. Kobayashi (1999). "Scandium Triflate in Organic Synthesis". Eur. J. Org. Chem. 1999: 15–27. doi: 10.1002/(SICI)1099-0690(199901)1999:1<15::AID-EJOC15>3.0.CO;2-B. Archived from the original on 2013-01-05.