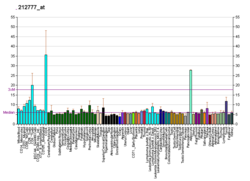
Son of sevenless homolog 1 is a protein that in humans is encoded by the SOS1 gene. [5] [6]
Function
SOS1 is a guanine nucleotide exchange factor (GEF) which interacts with Ras proteins to phosphorylate GDP into GTP, or from an inactive state to an active state to signal cell proliferation. RAS genes (e.g., MIM 190020) encode membrane-bound guanine nucleotide-binding proteins that function in the transduction of signals that control cell growth and differentiation. Binding of GTP activates RAS proteins, and subsequent hydrolysis of the bound GTP to GDP and phosphate inactivates signaling by these proteins. GTP binding can be catalyzed by guanine nucleotide exchange factors for RAS, and GTP hydrolysis can be accelerated by GTPase-activating proteins (GAPs). The first exchange factor to be identified for RAS was the S. cerevisiae Cdc25 gene product (not to be confused with the S. pombe Cdc25). Genetic analysis indicated that CDC25 is essential for activation of RAS proteins. In Drosophila, the protein encoded by the 'son of sevenless' gene (Sos) contains a domain that shows sequence similarity with the catalytic domain of Cdc25. Sos may act as a positive regulator of RAS by promoting guanine nucleotide exchange. [7]
Clinical significance
Recent studies also show that mutations in Sos1 can cause Noonan syndrome [8] and hereditary gingival fibromatosis type 1. [9] Noonan syndrome has also been shown to be caused by mutations in KRAS and PTPN11 genes. [10] activators of the MAP kinase pathway.
Inhibitors and activators
In 2019, the first SOS1 inhibitor, BAY-293, [11] was published which met the quality criteria for a 'Donated Chemical Probe' as defined by the Structural Genomics Consortium. [12] Shortly after, the discovery of BI-3406 [13] [14] was published.
In 2018, Fesik et al. reported the discovery of benzimidazole-derived SOS1 activators [15] (e.g. VUBI1).
Interactions
SOS1 has been shown to interact with:
- ABI1, [16]
- BCR gene, [17] [18]
- CRK, [19]
- EPS8, [16] [20]
- Epidermal growth factor receptor, [21] [22] [23]
- FRS2, [24] [25] [26]
- Grb2, [17] [19] [21] [24] [27] [28] [29] [30] [31] [32] [33] [34] [35] [36] [37] [38] [39] [40] [41] [42] [43]
- HRAS, [44] [45]
- ITSN1, [37]
- MUC1, [28] [46]
- NCK1, [19] [47] [48] [49]
- PLCG1, [50] [51]
- PTPN11, [27] [52]
- SH3KBP1, [53] and
- SHC1. [22] [25] [27] [40] and
See also
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000115904 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000024241 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Webb GC, Jenkins NA, Largaespada DA, Copeland NG, Fernandez CS, Bowtell DD (October 1993). "Mammalian homologues of the Drosophila Son of sevenless gene map to murine chromosomes 17 and 12 and to human chromosomes 2 and 14, respectively". Genomics. 18 (1): 14–19. doi: 10.1006/geno.1993.1421. PMID 8276400.
- ^ Xiao S, Wang X, Qu B, Yang M, Liu G, Bu L, et al. (September 2000). "Refinement of the locus for autosomal dominant hereditary gingival fibromatosis (GINGF) to a 3.8-cM region on 2p21". Genomics. 68 (3): 247–252. doi: 10.1006/geno.2000.6285. PMID 10995566.
- ^ "Entrez Gene: SOS1 son of sevenless homolog 1 (Drosophila)".
- ^ Roberts AE, Araki T, Swanson KD, Montgomery KT, Schiripo TA, Joshi VA, et al. (January 2007). "Germline gain-of-function mutations in SOS1 cause Noonan syndrome". Nature Genetics. 39 (1): 70–74. doi: 10.1038/ng1926. PMID 17143285. S2CID 10222262.
- ^ Hart TC, Zhang Y, Gorry MC, Hart PS, Cooper M, Marazita ML, et al. (April 2002). "A mutation in the SOS1 gene causes hereditary gingival fibromatosis type 1". American Journal of Human Genetics. 70 (4): 943–954. doi: 10.1086/339689. PMC 379122. PMID 11868160.
- ^ Tartaglia M, Mehler EL, Goldberg R, Zampino G, Brunner HG, Kremer H, et al. (December 2001). "Mutations in PTPN11, encoding the protein tyrosine phosphatase SHP-2, cause Noonan syndrome". Nature Genetics. 29 (4): 465–468. doi: 10.1038/ng772. PMID 11704759. S2CID 14627986.
- ^ Hillig RC, Sautier B, Schroeder J, Moosmayer D, Hilpmann A, Stegmann CM, et al. (February 2019). "Discovery of potent SOS1 inhibitors that block RAS activation via disruption of the RAS-SOS1 interaction". Proceedings of the National Academy of Sciences of the United States of America. 116 (7): 2551–2560. Bibcode: 2019PNAS..116.2551H. doi: 10.1073/pnas.1812963116. PMC 6377443. PMID 30683722.
- ^ "Donated Chemical Probes". 12 June 2018. Retrieved July 31, 2023.
- ^ Hofmann MH, Gmachl M, Ramharter J, Savarese F, Gerlach D, Marszalek JR, et al. (January 2021). "BI-3406, a Potent and Selective SOS1-KRAS Interaction Inhibitor, Is Effective in KRAS-Driven Cancers through Combined MEK Inhibition". Cancer Discovery. 11 (1): 142–157. doi: 10.1158/2159-8290.CD-20-0142. PMC 7892644. PMID 32816843.
- ^ Kessler D, Gerlach D, Kraut N, McConnell DB (June 2021). "Targeting Son of Sevenless 1: The pacemaker of KRAS". Current Opinion in Chemical Biology. 62: 109–118. doi: 10.1016/j.cbpa.2021.02.014. PMID 33848766.
- ^ Hodges TR, Abbott JR, Little AJ, Sarkar D, Salovich JM, Howes JE, et al. (October 2018). "Discovery and Structure-Based Optimization of Benzimidazole-Derived Activators of SOS1-Mediated Nucleotide Exchange on RAS". Journal of Medicinal Chemistry. 61 (19): 8875–8894. doi: 10.1021/acs.jmedchem.8b01108. PMC 8314423. PMID 30205005.
- ^ a b Scita G, Nordstrom J, Carbone R, Tenca P, Giardina G, Gutkind S, et al. (September 1999). "EPS8 and E3B1 transduce signals from Ras to Rac". Nature. 401 (6750): 290–293. Bibcode: 1999Natur.401..290S. doi: 10.1038/45822. PMID 10499589. S2CID 205045006.
- ^ a b Puil L, Liu J, Gish G, Mbamalu G, Bowtell D, Pelicci PG, et al. (February 1994). "Bcr-Abl oncoproteins bind directly to activators of the Ras signalling pathway". The EMBO Journal. 13 (4): 764–773. doi: 10.1002/j.1460-2075.1994.tb06319.x. PMC 394874. PMID 8112292.
- ^ Maru Y, Peters KL, Afar DE, Shibuya M, Witte ON, Smithgall TE (February 1995). "Tyrosine phosphorylation of BCR by FPS/FES protein-tyrosine kinases induces association of BCR with GRB-2/SOS". Molecular and Cellular Biology. 15 (2): 835–842. doi: 10.1128/MCB.15.2.835. PMC 231961. PMID 7529874.
- ^ a b c Okada S, Pessin JE (October 1996). "Interactions between Src homology (SH) 2/SH3 adapter proteins and the guanylnucleotide exchange factor SOS are differentially regulated by insulin and epidermal growth factor". The Journal of Biological Chemistry. 271 (41): 25533–25538. doi: 10.1074/jbc.271.41.25533. PMID 8810325.
- ^ Offenhäuser N, Borgonovo A, Disanza A, Romano P, Ponzanelli I, Iannolo G, et al. (January 2004). "The eps8 family of proteins links growth factor stimulation to actin reorganization generating functional redundancy in the Ras/Rac pathway". Molecular Biology of the Cell. 15 (1): 91–98. doi: 10.1091/mbc.E03-06-0427. PMC 307530. PMID 14565974.
- ^ a b Buday L, Egan SE, Rodriguez Viciana P, Cantrell DA, Downward J (March 1994). "A complex of Grb2 adaptor protein, Sos exchange factor, and a 36-kDa membrane-bound tyrosine phosphoprotein is implicated in ras activation in T cells". The Journal of Biological Chemistry. 269 (12): 9019–9023. doi: 10.1016/S0021-9258(17)37070-9. PMID 7510700.
- ^ a b Qian X, Esteban L, Vass WC, Upadhyaya C, Papageorge AG, Yienger K, et al. (February 2000). "The Sos1 and Sos2 Ras-specific exchange factors: differences in placental expression and signaling properties". The EMBO Journal. 19 (4): 642–654. doi: 10.1093/emboj/19.4.642. PMC 305602. PMID 10675333.
- ^ Qian X, Vass WC, Papageorge AG, Anborgh PH, Lowy DR (February 1998). "N terminus of Sos1 Ras exchange factor: critical roles for the Dbl and pleckstrin homology domains". Molecular and Cellular Biology. 18 (2): 771–778. doi: 10.1128/mcb.18.2.771. PMC 108788. PMID 9447973.
- ^ a b Wong A, Lamothe B, Lee A, Schlessinger J, Lax I (May 2002). "FRS2 alpha attenuates FGF receptor signaling by Grb2-mediated recruitment of the ubiquitin ligase Cbl". Proceedings of the National Academy of Sciences of the United States of America. 99 (10): 6684–6689. Bibcode: 2002PNAS...99.6684W. doi: 10.1073/pnas.052138899. PMC 124463. PMID 11997436.
- ^ a b Foehr ED, Tatavos A, Tanabe E, Raffioni S, Goetz S, Dimarco E, et al. (May 2000). "Discoidin domain receptor 1 (DDR1) signaling in PC12 cells: activation of juxtamembrane domains in PDGFR/DDR/TrkA chimeric receptors". FASEB Journal. 14 (7): 973–981. doi: 10.1096/fasebj.14.7.973. PMID 10783152. S2CID 43317719.
- ^ Kouhara H, Hadari YR, Spivak-Kroizman T, Schilling J, Bar-Sagi D, Lax I, Schlessinger J (May 1997). "A lipid-anchored Grb2-binding protein that links FGF-receptor activation to the Ras/MAPK signaling pathway". Cell. 89 (5): 693–702. doi: 10.1016/s0092-8674(00)80252-4. PMID 9182757. S2CID 2187363.
- ^ a b c Chin H, Saito T, Arai A, Yamamoto K, Kamiyama R, Miyasaka N, Miura O (October 1997). "Erythropoietin and IL-3 induce tyrosine phosphorylation of CrkL and its association with Shc, SHP-2, and Cbl in hematopoietic cells". Biochemical and Biophysical Research Communications. 239 (2): 412–417. doi: 10.1006/bbrc.1997.7480. PMID 9344843.
- ^ a b Pandey P, Kharbanda S, Kufe D (September 1995). "Association of the DF3/MUC1 breast cancer antigen with Grb2 and the Sos/Ras exchange protein". Cancer Research. 55 (18): 4000–4003. PMID 7664271.
- ^ Blagoev B, Kratchmarova I, Ong SE, Nielsen M, Foster LJ, Mann M (March 2003). "A proteomics strategy to elucidate functional protein-protein interactions applied to EGF signaling". Nature Biotechnology. 21 (3): 315–318. doi: 10.1038/nbt790. PMID 12577067. S2CID 26838266.
- ^ Liu SK, McGlade CJ (December 1998). "Gads is a novel SH2 and SH3 domain-containing adaptor protein that binds to tyrosine-phosphorylated Shc". Oncogene. 17 (24): 3073–3082. doi: 10.1038/sj.onc.1202337. PMID 9872323.
- ^ Oneyama C, Nakano H, Sharma SV (March 2002). "UCS15A, a novel small molecule, SH3 domain-mediated protein-protein interaction blocking drug". Oncogene. 21 (13): 2037–2050. doi: 10.1038/sj.onc.1205271. PMID 11960376.
- ^ Robinson A, Gibbins J, Rodríguez-Liñares B, Finan PM, Wilson L, Kellie S, et al. (July 1996). "Characterization of Grb2-binding proteins in human platelets activated by Fc gamma RIIA cross-linking". Blood. 88 (2): 522–530. doi: 10.1182/blood.V88.2.522.bloodjournal882522. PMID 8695800.
- ^ Li N, Batzer A, Daly R, Yajnik V, Skolnik E, Chardin P, et al. (May 1993). "Guanine-nucleotide-releasing factor hSos1 binds to Grb2 and links receptor tyrosine kinases to Ras signalling". Nature. 363 (6424): 85–88. Bibcode: 1993Natur.363...85L. doi: 10.1038/363085a0. PMID 8479541. S2CID 4323174.
- ^ Reif K, Buday L, Downward J, Cantrell DA (May 1994). "SH3 domains of the adapter molecule Grb2 complex with two proteins in T cells: the guanine nucleotide exchange protein Sos and a 75-kDa protein that is a substrate for T cell antigen receptor-activated tyrosine kinases". The Journal of Biological Chemistry. 269 (19): 14081–14087. doi: 10.1016/S0021-9258(17)36757-1. PMID 8188688.
- ^ D'Angelo G, Martini JF, Iiri T, Fantl WJ, Martial J, Weiner RI (May 1999). "16K human prolactin inhibits vascular endothelial growth factor-induced activation of Ras in capillary endothelial cells". Molecular Endocrinology. 13 (5): 692–704. doi: 10.1210/mend.13.5.0280. PMID 10319320.
- ^ Nel AE, Gupta S, Lee L, Ledbetter JA, Kanner SB (August 1995). "Ligation of the T-cell antigen receptor (TCR) induces association of hSos1, ZAP-70, phospholipase C-gamma 1, and other phosphoproteins with Grb2 and the zeta-chain of the TCR". The Journal of Biological Chemistry. 270 (31): 18428–18436. doi: 10.1074/jbc.270.31.18428. PMID 7629168.
- ^ a b Tong XK, Hussain NK, de Heuvel E, Kurakin A, Abi-Jaoude E, Quinn CC, et al. (March 2000). "The endocytic protein intersectin is a major binding partner for the Ras exchange factor mSos1 in rat brain". The EMBO Journal. 19 (6): 1263–1271. doi: 10.1093/emboj/19.6.1263. PMC 305667. PMID 10716926.
- ^ Park RK, Izadi KD, Deo YM, Durden DL (September 1999). "Role of Src in the modulation of multiple adaptor proteins in FcalphaRI oxidant signaling". Blood. 94 (6): 2112–2120. doi: 10.1182/blood.V94.6.2112. PMID 10477741.
- ^ Germani A, Romero F, Houlard M, Camonis J, Gisselbrecht S, Fischer S, Varin-Blank N (May 1999). "hSiah2 is a new Vav binding protein which inhibits Vav-mediated signaling pathways". Molecular and Cellular Biology. 19 (5): 3798–3807. doi: 10.1128/MCB.19.5.3798. PMC 84217. PMID 10207103.
- ^ a b Park RK, Erdreich-Epstein A, Liu M, Izadi KD, Durden DL (December 1999). "High affinity IgG receptor activation of Src family kinases is required for modulation of the Shc-Grb2-Sos complex and the downstream activation of the nicotinamide adenine dinucleotide phosphate (reduced) oxidase". Journal of Immunology. 163 (11): 6023–6034. doi: 10.4049/jimmunol.163.11.6023. PMID 10570290. S2CID 36719981.
- ^ Saci A, Liu WQ, Vidal M, Garbay C, Rendu F, Bachelot-Loza C (May 2002). "Differential effect of the inhibition of Grb2-SH3 interactions in platelet activation induced by thrombin and by Fc receptor engagement". The Biochemical Journal. 363 (Pt 3): 717–725. doi: 10.1042/0264-6021:3630717. PMC 1222524. PMID 11964172.
- ^ Sastry L, Cao T, King CR (January 1997). "Multiple Grb2-protein complexes in human cancer cells". International Journal of Cancer. 70 (2): 208–213. doi: 10.1002/(sici)1097-0215(19970117)70:2<208::aid-ijc12>3.0.co;2-e. PMID 9009162. S2CID 10317185.
- ^ Spivak-Kroizman T, Mohammadi M, Hu P, Jaye M, Schlessinger J, Lax I (May 1994). "Point mutation in the fibroblast growth factor receptor eliminates phosphatidylinositol hydrolysis without affecting neuronal differentiation of PC12 cells". The Journal of Biological Chemistry. 269 (20): 14419–14423. doi: 10.1016/S0021-9258(17)36639-5. PMID 7514169.
- ^ Margarit SM, Sondermann H, Hall BE, Nagar B, Hoelz A, Pirruccello M, et al. (March 2003). "Structural evidence for feedback activation by Ras.GTP of the Ras-specific nucleotide exchange factor SOS". Cell. 112 (5): 685–695. doi: 10.1016/s0092-8674(03)00149-1. PMID 12628188. S2CID 11576361.
- ^ Boriack-Sjodin PA, Margarit SM, Bar-Sagi D, Kuriyan J (July 1998). "The structural basis of the activation of Ras by Sos". Nature. 394 (6691): 337–343. Bibcode: 1998Natur.394..337B. doi: 10.1038/28548. PMID 9690470. S2CID 204998911.
- ^ Schroeder JA, Thompson MC, Gardner MM, Gendler SJ (April 2001). "Transgenic MUC1 interacts with epidermal growth factor receptor and correlates with mitogen-activated protein kinase activation in the mouse mammary gland". The Journal of Biological Chemistry. 276 (16): 13057–13064. doi: 10.1074/jbc.M011248200. PMID 11278868.
- ^ Wunderlich L, Faragó A, Buday L (January 1999). "Characterization of interactions of Nck with Sos and dynamin". Cellular Signalling. 11 (1): 25–29. doi: 10.1016/s0898-6568(98)00027-8. PMID 10206341.
- ^ Hu Q, Milfay D, Williams LT (March 1995). "Binding of NCK to SOS and activation of ras-dependent gene expression". Molecular and Cellular Biology. 15 (3): 1169–1174. doi: 10.1128/MCB.15.3.1169. PMC 230339. PMID 7862111.
- ^ Braverman LE, Quilliam LA (February 1999). "Identification of Grb4/Nckbeta, a src homology 2 and 3 domain-containing adapter protein having similar binding and biological properties to Nck". The Journal of Biological Chemistry. 274 (9): 5542–5549. doi: 10.1074/jbc.274.9.5542. PMID 10026169.
- ^ Kim MJ, Chang JS, Park SK, Hwang JI, Ryu SH, Suh PG (July 2000). "Direct interaction of SOS1 Ras exchange protein with the SH3 domain of phospholipase C-gamma1". Biochemistry. 39 (29): 8674–8682. doi: 10.1021/bi992558t. PMID 10913276.
- ^ Scholler JK, Perez-Villar JJ, O'Day K, Kanner SB (August 2000). "Engagement of the T lymphocyte antigen receptor regulates association of son-of-sevenless homologues with the SH3 domain of phospholipase Cgamma1". European Journal of Immunology. 30 (8): 2378–2387. doi: 10.1002/1521-4141(2000)30:8<2378::AID-IMMU2378>3.0.CO;2-E. PMID 10940929.
- ^ Hadari YR, Kouhara H, Lax I, Schlessinger J (July 1998). "Binding of Shp2 tyrosine phosphatase to FRS2 is essential for fibroblast growth factor-induced PC12 cell differentiation". Molecular and Cellular Biology. 18 (7): 3966–3973. doi: 10.1128/MCB.18.7.3966. PMC 108981. PMID 9632781.
- ^ Watanabe S, Take H, Takeda K, Yu ZX, Iwata N, Kajigaya S (November 2000). "Characterization of the CIN85 adaptor protein and identification of components involved in CIN85 complexes". Biochemical and Biophysical Research Communications. 278 (1): 167–174. doi: 10.1006/bbrc.2000.3760. PMID 11071869.
Further reading
- Lioubin MN, Myles GM, Carlberg K, Bowtell D, Rohrschneider LR (September 1994). "Shc, Grb2, Sos1, and a 150-kilodalton tyrosine-phosphorylated protein form complexes with Fms in hematopoietic cells". Molecular and Cellular Biology. 14 (9): 5682–5691. doi: 10.1128/mcb.14.9.5682. PMC 359093. PMID 7520523.
- Nel AE, Gupta S, Lee L, Ledbetter JA, Kanner SB (August 1995). "Ligation of the T-cell antigen receptor (TCR) induces association of hSos1, ZAP-70, phospholipase C-gamma 1, and other phosphoproteins with Grb2 and the zeta-chain of the TCR". The Journal of Biological Chemistry. 270 (31): 18428–18436. doi: 10.1074/jbc.270.31.18428. PMID 7629168.
- Pandey P, Kharbanda S, Kufe D (September 1995). "Association of the DF3/MUC1 breast cancer antigen with Grb2 and the Sos/Ras exchange protein". Cancer Research. 55 (18): 4000–4003. PMID 7664271.
- Hu Q, Milfay D, Williams LT (March 1995). "Binding of NCK to SOS and activation of ras-dependent gene expression". Molecular and Cellular Biology. 15 (3): 1169–1174. doi: 10.1128/MCB.15.3.1169. PMC 230339. PMID 7862111.
- Puil L, Liu J, Gish G, Mbamalu G, Bowtell D, Pelicci PG, et al. (February 1994). "Bcr-Abl oncoproteins bind directly to activators of the Ras signalling pathway". The EMBO Journal. 13 (4): 764–773. doi: 10.1002/j.1460-2075.1994.tb06319.x. PMC 394874. PMID 8112292.
- Li N, Batzer A, Daly R, Yajnik V, Skolnik E, Chardin P, et al. (May 1993). "Guanine-nucleotide-releasing factor hSos1 binds to Grb2 and links receptor tyrosine kinases to Ras signalling". Nature. 363 (6424): 85–88. Bibcode: 1993Natur.363...85L. doi: 10.1038/363085a0. PMID 8479541. S2CID 4323174.
- Chardin P, Camonis JH, Gale NW, van Aelst L, Schlessinger J, Wigler MH, Bar-Sagi D (May 1993). "Human Sos1: a guanine nucleotide exchange factor for Ras that binds to GRB2". Science. 260 (5112): 1338–1343. Bibcode: 1993Sci...260.1338C. doi: 10.1126/science.8493579. PMID 8493579.
- Sadoshima J, Izumo S (February 1996). "The heterotrimeric G q protein-coupled angiotensin II receptor activates p21 ras via the tyrosine kinase-Shc-Grb2-Sos pathway in cardiac myocytes". The EMBO Journal. 15 (4): 775–787. doi: 10.1002/j.1460-2075.1996.tb00413.x. PMC 450276. PMID 8631299.
- Feng GS, Ouyang YB, Hu DP, Shi ZQ, Gentz R, Ni J (May 1996). "Grap is a novel SH3-SH2-SH3 adaptor protein that couples tyrosine kinases to the Ras pathway". The Journal of Biological Chemistry. 271 (21): 12129–12132. doi: 10.1074/jbc.271.21.12129. PMID 8647802.
- Okada S, Pessin JE (October 1996). "Interactions between Src homology (SH) 2/SH3 adapter proteins and the guanylnucleotide exchange factor SOS are differentially regulated by insulin and epidermal growth factor". The Journal of Biological Chemistry. 271 (41): 25533–25538. doi: 10.1074/jbc.271.41.25533. PMID 8810325.
- Corbalan-Garcia S, Yang SS, Degenhardt KR, Bar-Sagi D (October 1996). "Identification of the mitogen-activated protein kinase phosphorylation sites on human Sos1 that regulate interaction with Grb2". Molecular and Cellular Biology. 16 (10): 5674–5682. doi: 10.1128/MCB.16.10.5674. PMC 231567. PMID 8816480.
- Sattler M, Salgia R, Shrikhande G, Verma S, Uemura N, Law SF, et al. (May 1997). "Differential signaling after beta1 integrin ligation is mediated through binding of CRKL to p120(CBL) and p110(HEF1)". The Journal of Biological Chemistry. 272 (22): 14320–14326. doi: 10.1074/jbc.272.22.14320. hdl: 20.500.12613/9173. PMID 9162067.
- Leprince C, Romero F, Cussac D, Vayssiere B, Berger R, Tavitian A, Camonis JH (June 1997). "A new member of the amphiphysin family connecting endocytosis and signal transduction pathways". The Journal of Biological Chemistry. 272 (24): 15101–15105. doi: 10.1074/jbc.272.24.15101. PMID 9182529.
- Kouhara H, Hadari YR, Spivak-Kroizman T, Schilling J, Bar-Sagi D, Lax I, Schlessinger J (May 1997). "A lipid-anchored Grb2-binding protein that links FGF-receptor activation to the Ras/MAPK signaling pathway". Cell. 89 (5): 693–702. doi: 10.1016/S0092-8674(00)80252-4. PMID 9182757. S2CID 2187363.
- Chin H, Saito T, Arai A, Yamamoto K, Kamiyama R, Miyasaka N, Miura O (October 1997). "Erythropoietin and IL-3 induce tyrosine phosphorylation of CrkL and its association with Shc, SHP-2, and Cbl in hematopoietic cells". Biochemical and Biophysical Research Communications. 239 (2): 412–417. doi: 10.1006/bbrc.1997.7480. PMID 9344843.
- Zheng J, Chen RH, Corblan-Garcia S, Cahill SM, Bar-Sagi D, Cowburn D (November 1997). "The solution structure of the pleckstrin homology domain of human SOS1. A possible structural role for the sequential association of diffuse B cell lymphoma and pleckstrin homology domains". The Journal of Biological Chemistry. 272 (48): 30340–30344. doi: 10.1074/jbc.272.48.30340. PMID 9374522.
- Li S, Kim M, Hu YL, Jalali S, Schlaepfer DD, Hunter T, et al. (November 1997). "Fluid shear stress activation of focal adhesion kinase. Linking to mitogen-activated protein kinases". The Journal of Biological Chemistry. 272 (48): 30455–30462. doi: 10.1074/jbc.272.48.30455. PMID 9374537.
- Qian X, Vass WC, Papageorge AG, Anborgh PH, Lowy DR (February 1998). "N terminus of Sos1 Ras exchange factor: critical roles for the Dbl and pleckstrin homology domains". Molecular and Cellular Biology. 18 (2): 771–778. doi: 10.1128/mcb.18.2.771. PMC 108788. PMID 9447973.
- Curto M, Frankel P, Carrero A, Foster DA (February 1998). "Novel recruitment of Shc, Grb2, and Sos by fibroblast growth factor receptor-1 in v-Src-transformed cells". Biochemical and Biophysical Research Communications. 243 (2): 555–560. doi: 10.1006/bbrc.1997.7982. PMID 9480847.
External links
- GeneReviews/NCBI/NIH/UW entry on Noonan syndrome
- http://www.noonansyndrome.org
- http://ghr.nlm.nih.gov/gene=sos1
This article incorporates text from the United States National Library of Medicine, which is in the public domain.
| SOS1 | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | SOS1, GF1, GGF1, GINGF, HGF, NS4, SOS Ras/Rac guanine nucleotide exchange factor 1, SOS-1 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 182530; MGI: 98354; HomoloGene: 4117; GeneCards: SOS1; OMA: SOS1 - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Son of sevenless homolog 1 is a protein that in humans is encoded by the SOS1 gene. [5] [6]
Function
SOS1 is a guanine nucleotide exchange factor (GEF) which interacts with Ras proteins to phosphorylate GDP into GTP, or from an inactive state to an active state to signal cell proliferation. RAS genes (e.g., MIM 190020) encode membrane-bound guanine nucleotide-binding proteins that function in the transduction of signals that control cell growth and differentiation. Binding of GTP activates RAS proteins, and subsequent hydrolysis of the bound GTP to GDP and phosphate inactivates signaling by these proteins. GTP binding can be catalyzed by guanine nucleotide exchange factors for RAS, and GTP hydrolysis can be accelerated by GTPase-activating proteins (GAPs). The first exchange factor to be identified for RAS was the S. cerevisiae Cdc25 gene product (not to be confused with the S. pombe Cdc25). Genetic analysis indicated that CDC25 is essential for activation of RAS proteins. In Drosophila, the protein encoded by the 'son of sevenless' gene (Sos) contains a domain that shows sequence similarity with the catalytic domain of Cdc25. Sos may act as a positive regulator of RAS by promoting guanine nucleotide exchange. [7]
Clinical significance
Recent studies also show that mutations in Sos1 can cause Noonan syndrome [8] and hereditary gingival fibromatosis type 1. [9] Noonan syndrome has also been shown to be caused by mutations in KRAS and PTPN11 genes. [10] activators of the MAP kinase pathway.
Inhibitors and activators
In 2019, the first SOS1 inhibitor, BAY-293, [11] was published which met the quality criteria for a 'Donated Chemical Probe' as defined by the Structural Genomics Consortium. [12] Shortly after, the discovery of BI-3406 [13] [14] was published.
In 2018, Fesik et al. reported the discovery of benzimidazole-derived SOS1 activators [15] (e.g. VUBI1).
Interactions
SOS1 has been shown to interact with:
- ABI1, [16]
- BCR gene, [17] [18]
- CRK, [19]
- EPS8, [16] [20]
- Epidermal growth factor receptor, [21] [22] [23]
- FRS2, [24] [25] [26]
- Grb2, [17] [19] [21] [24] [27] [28] [29] [30] [31] [32] [33] [34] [35] [36] [37] [38] [39] [40] [41] [42] [43]
- HRAS, [44] [45]
- ITSN1, [37]
- MUC1, [28] [46]
- NCK1, [19] [47] [48] [49]
- PLCG1, [50] [51]
- PTPN11, [27] [52]
- SH3KBP1, [53] and
- SHC1. [22] [25] [27] [40] and
See also
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000115904 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000024241 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Webb GC, Jenkins NA, Largaespada DA, Copeland NG, Fernandez CS, Bowtell DD (October 1993). "Mammalian homologues of the Drosophila Son of sevenless gene map to murine chromosomes 17 and 12 and to human chromosomes 2 and 14, respectively". Genomics. 18 (1): 14–19. doi: 10.1006/geno.1993.1421. PMID 8276400.
- ^ Xiao S, Wang X, Qu B, Yang M, Liu G, Bu L, et al. (September 2000). "Refinement of the locus for autosomal dominant hereditary gingival fibromatosis (GINGF) to a 3.8-cM region on 2p21". Genomics. 68 (3): 247–252. doi: 10.1006/geno.2000.6285. PMID 10995566.
- ^ "Entrez Gene: SOS1 son of sevenless homolog 1 (Drosophila)".
- ^ Roberts AE, Araki T, Swanson KD, Montgomery KT, Schiripo TA, Joshi VA, et al. (January 2007). "Germline gain-of-function mutations in SOS1 cause Noonan syndrome". Nature Genetics. 39 (1): 70–74. doi: 10.1038/ng1926. PMID 17143285. S2CID 10222262.
- ^ Hart TC, Zhang Y, Gorry MC, Hart PS, Cooper M, Marazita ML, et al. (April 2002). "A mutation in the SOS1 gene causes hereditary gingival fibromatosis type 1". American Journal of Human Genetics. 70 (4): 943–954. doi: 10.1086/339689. PMC 379122. PMID 11868160.
- ^ Tartaglia M, Mehler EL, Goldberg R, Zampino G, Brunner HG, Kremer H, et al. (December 2001). "Mutations in PTPN11, encoding the protein tyrosine phosphatase SHP-2, cause Noonan syndrome". Nature Genetics. 29 (4): 465–468. doi: 10.1038/ng772. PMID 11704759. S2CID 14627986.
- ^ Hillig RC, Sautier B, Schroeder J, Moosmayer D, Hilpmann A, Stegmann CM, et al. (February 2019). "Discovery of potent SOS1 inhibitors that block RAS activation via disruption of the RAS-SOS1 interaction". Proceedings of the National Academy of Sciences of the United States of America. 116 (7): 2551–2560. Bibcode: 2019PNAS..116.2551H. doi: 10.1073/pnas.1812963116. PMC 6377443. PMID 30683722.
- ^ "Donated Chemical Probes". 12 June 2018. Retrieved July 31, 2023.
- ^ Hofmann MH, Gmachl M, Ramharter J, Savarese F, Gerlach D, Marszalek JR, et al. (January 2021). "BI-3406, a Potent and Selective SOS1-KRAS Interaction Inhibitor, Is Effective in KRAS-Driven Cancers through Combined MEK Inhibition". Cancer Discovery. 11 (1): 142–157. doi: 10.1158/2159-8290.CD-20-0142. PMC 7892644. PMID 32816843.
- ^ Kessler D, Gerlach D, Kraut N, McConnell DB (June 2021). "Targeting Son of Sevenless 1: The pacemaker of KRAS". Current Opinion in Chemical Biology. 62: 109–118. doi: 10.1016/j.cbpa.2021.02.014. PMID 33848766.
- ^ Hodges TR, Abbott JR, Little AJ, Sarkar D, Salovich JM, Howes JE, et al. (October 2018). "Discovery and Structure-Based Optimization of Benzimidazole-Derived Activators of SOS1-Mediated Nucleotide Exchange on RAS". Journal of Medicinal Chemistry. 61 (19): 8875–8894. doi: 10.1021/acs.jmedchem.8b01108. PMC 8314423. PMID 30205005.
- ^ a b Scita G, Nordstrom J, Carbone R, Tenca P, Giardina G, Gutkind S, et al. (September 1999). "EPS8 and E3B1 transduce signals from Ras to Rac". Nature. 401 (6750): 290–293. Bibcode: 1999Natur.401..290S. doi: 10.1038/45822. PMID 10499589. S2CID 205045006.
- ^ a b Puil L, Liu J, Gish G, Mbamalu G, Bowtell D, Pelicci PG, et al. (February 1994). "Bcr-Abl oncoproteins bind directly to activators of the Ras signalling pathway". The EMBO Journal. 13 (4): 764–773. doi: 10.1002/j.1460-2075.1994.tb06319.x. PMC 394874. PMID 8112292.
- ^ Maru Y, Peters KL, Afar DE, Shibuya M, Witte ON, Smithgall TE (February 1995). "Tyrosine phosphorylation of BCR by FPS/FES protein-tyrosine kinases induces association of BCR with GRB-2/SOS". Molecular and Cellular Biology. 15 (2): 835–842. doi: 10.1128/MCB.15.2.835. PMC 231961. PMID 7529874.
- ^ a b c Okada S, Pessin JE (October 1996). "Interactions between Src homology (SH) 2/SH3 adapter proteins and the guanylnucleotide exchange factor SOS are differentially regulated by insulin and epidermal growth factor". The Journal of Biological Chemistry. 271 (41): 25533–25538. doi: 10.1074/jbc.271.41.25533. PMID 8810325.
- ^ Offenhäuser N, Borgonovo A, Disanza A, Romano P, Ponzanelli I, Iannolo G, et al. (January 2004). "The eps8 family of proteins links growth factor stimulation to actin reorganization generating functional redundancy in the Ras/Rac pathway". Molecular Biology of the Cell. 15 (1): 91–98. doi: 10.1091/mbc.E03-06-0427. PMC 307530. PMID 14565974.
- ^ a b Buday L, Egan SE, Rodriguez Viciana P, Cantrell DA, Downward J (March 1994). "A complex of Grb2 adaptor protein, Sos exchange factor, and a 36-kDa membrane-bound tyrosine phosphoprotein is implicated in ras activation in T cells". The Journal of Biological Chemistry. 269 (12): 9019–9023. doi: 10.1016/S0021-9258(17)37070-9. PMID 7510700.
- ^ a b Qian X, Esteban L, Vass WC, Upadhyaya C, Papageorge AG, Yienger K, et al. (February 2000). "The Sos1 and Sos2 Ras-specific exchange factors: differences in placental expression and signaling properties". The EMBO Journal. 19 (4): 642–654. doi: 10.1093/emboj/19.4.642. PMC 305602. PMID 10675333.
- ^ Qian X, Vass WC, Papageorge AG, Anborgh PH, Lowy DR (February 1998). "N terminus of Sos1 Ras exchange factor: critical roles for the Dbl and pleckstrin homology domains". Molecular and Cellular Biology. 18 (2): 771–778. doi: 10.1128/mcb.18.2.771. PMC 108788. PMID 9447973.
- ^ a b Wong A, Lamothe B, Lee A, Schlessinger J, Lax I (May 2002). "FRS2 alpha attenuates FGF receptor signaling by Grb2-mediated recruitment of the ubiquitin ligase Cbl". Proceedings of the National Academy of Sciences of the United States of America. 99 (10): 6684–6689. Bibcode: 2002PNAS...99.6684W. doi: 10.1073/pnas.052138899. PMC 124463. PMID 11997436.
- ^ a b Foehr ED, Tatavos A, Tanabe E, Raffioni S, Goetz S, Dimarco E, et al. (May 2000). "Discoidin domain receptor 1 (DDR1) signaling in PC12 cells: activation of juxtamembrane domains in PDGFR/DDR/TrkA chimeric receptors". FASEB Journal. 14 (7): 973–981. doi: 10.1096/fasebj.14.7.973. PMID 10783152. S2CID 43317719.
- ^ Kouhara H, Hadari YR, Spivak-Kroizman T, Schilling J, Bar-Sagi D, Lax I, Schlessinger J (May 1997). "A lipid-anchored Grb2-binding protein that links FGF-receptor activation to the Ras/MAPK signaling pathway". Cell. 89 (5): 693–702. doi: 10.1016/s0092-8674(00)80252-4. PMID 9182757. S2CID 2187363.
- ^ a b c Chin H, Saito T, Arai A, Yamamoto K, Kamiyama R, Miyasaka N, Miura O (October 1997). "Erythropoietin and IL-3 induce tyrosine phosphorylation of CrkL and its association with Shc, SHP-2, and Cbl in hematopoietic cells". Biochemical and Biophysical Research Communications. 239 (2): 412–417. doi: 10.1006/bbrc.1997.7480. PMID 9344843.
- ^ a b Pandey P, Kharbanda S, Kufe D (September 1995). "Association of the DF3/MUC1 breast cancer antigen with Grb2 and the Sos/Ras exchange protein". Cancer Research. 55 (18): 4000–4003. PMID 7664271.
- ^ Blagoev B, Kratchmarova I, Ong SE, Nielsen M, Foster LJ, Mann M (March 2003). "A proteomics strategy to elucidate functional protein-protein interactions applied to EGF signaling". Nature Biotechnology. 21 (3): 315–318. doi: 10.1038/nbt790. PMID 12577067. S2CID 26838266.
- ^ Liu SK, McGlade CJ (December 1998). "Gads is a novel SH2 and SH3 domain-containing adaptor protein that binds to tyrosine-phosphorylated Shc". Oncogene. 17 (24): 3073–3082. doi: 10.1038/sj.onc.1202337. PMID 9872323.
- ^ Oneyama C, Nakano H, Sharma SV (March 2002). "UCS15A, a novel small molecule, SH3 domain-mediated protein-protein interaction blocking drug". Oncogene. 21 (13): 2037–2050. doi: 10.1038/sj.onc.1205271. PMID 11960376.
- ^ Robinson A, Gibbins J, Rodríguez-Liñares B, Finan PM, Wilson L, Kellie S, et al. (July 1996). "Characterization of Grb2-binding proteins in human platelets activated by Fc gamma RIIA cross-linking". Blood. 88 (2): 522–530. doi: 10.1182/blood.V88.2.522.bloodjournal882522. PMID 8695800.
- ^ Li N, Batzer A, Daly R, Yajnik V, Skolnik E, Chardin P, et al. (May 1993). "Guanine-nucleotide-releasing factor hSos1 binds to Grb2 and links receptor tyrosine kinases to Ras signalling". Nature. 363 (6424): 85–88. Bibcode: 1993Natur.363...85L. doi: 10.1038/363085a0. PMID 8479541. S2CID 4323174.
- ^ Reif K, Buday L, Downward J, Cantrell DA (May 1994). "SH3 domains of the adapter molecule Grb2 complex with two proteins in T cells: the guanine nucleotide exchange protein Sos and a 75-kDa protein that is a substrate for T cell antigen receptor-activated tyrosine kinases". The Journal of Biological Chemistry. 269 (19): 14081–14087. doi: 10.1016/S0021-9258(17)36757-1. PMID 8188688.
- ^ D'Angelo G, Martini JF, Iiri T, Fantl WJ, Martial J, Weiner RI (May 1999). "16K human prolactin inhibits vascular endothelial growth factor-induced activation of Ras in capillary endothelial cells". Molecular Endocrinology. 13 (5): 692–704. doi: 10.1210/mend.13.5.0280. PMID 10319320.
- ^ Nel AE, Gupta S, Lee L, Ledbetter JA, Kanner SB (August 1995). "Ligation of the T-cell antigen receptor (TCR) induces association of hSos1, ZAP-70, phospholipase C-gamma 1, and other phosphoproteins with Grb2 and the zeta-chain of the TCR". The Journal of Biological Chemistry. 270 (31): 18428–18436. doi: 10.1074/jbc.270.31.18428. PMID 7629168.
- ^ a b Tong XK, Hussain NK, de Heuvel E, Kurakin A, Abi-Jaoude E, Quinn CC, et al. (March 2000). "The endocytic protein intersectin is a major binding partner for the Ras exchange factor mSos1 in rat brain". The EMBO Journal. 19 (6): 1263–1271. doi: 10.1093/emboj/19.6.1263. PMC 305667. PMID 10716926.
- ^ Park RK, Izadi KD, Deo YM, Durden DL (September 1999). "Role of Src in the modulation of multiple adaptor proteins in FcalphaRI oxidant signaling". Blood. 94 (6): 2112–2120. doi: 10.1182/blood.V94.6.2112. PMID 10477741.
- ^ Germani A, Romero F, Houlard M, Camonis J, Gisselbrecht S, Fischer S, Varin-Blank N (May 1999). "hSiah2 is a new Vav binding protein which inhibits Vav-mediated signaling pathways". Molecular and Cellular Biology. 19 (5): 3798–3807. doi: 10.1128/MCB.19.5.3798. PMC 84217. PMID 10207103.
- ^ a b Park RK, Erdreich-Epstein A, Liu M, Izadi KD, Durden DL (December 1999). "High affinity IgG receptor activation of Src family kinases is required for modulation of the Shc-Grb2-Sos complex and the downstream activation of the nicotinamide adenine dinucleotide phosphate (reduced) oxidase". Journal of Immunology. 163 (11): 6023–6034. doi: 10.4049/jimmunol.163.11.6023. PMID 10570290. S2CID 36719981.
- ^ Saci A, Liu WQ, Vidal M, Garbay C, Rendu F, Bachelot-Loza C (May 2002). "Differential effect of the inhibition of Grb2-SH3 interactions in platelet activation induced by thrombin and by Fc receptor engagement". The Biochemical Journal. 363 (Pt 3): 717–725. doi: 10.1042/0264-6021:3630717. PMC 1222524. PMID 11964172.
- ^ Sastry L, Cao T, King CR (January 1997). "Multiple Grb2-protein complexes in human cancer cells". International Journal of Cancer. 70 (2): 208–213. doi: 10.1002/(sici)1097-0215(19970117)70:2<208::aid-ijc12>3.0.co;2-e. PMID 9009162. S2CID 10317185.
- ^ Spivak-Kroizman T, Mohammadi M, Hu P, Jaye M, Schlessinger J, Lax I (May 1994). "Point mutation in the fibroblast growth factor receptor eliminates phosphatidylinositol hydrolysis without affecting neuronal differentiation of PC12 cells". The Journal of Biological Chemistry. 269 (20): 14419–14423. doi: 10.1016/S0021-9258(17)36639-5. PMID 7514169.
- ^ Margarit SM, Sondermann H, Hall BE, Nagar B, Hoelz A, Pirruccello M, et al. (March 2003). "Structural evidence for feedback activation by Ras.GTP of the Ras-specific nucleotide exchange factor SOS". Cell. 112 (5): 685–695. doi: 10.1016/s0092-8674(03)00149-1. PMID 12628188. S2CID 11576361.
- ^ Boriack-Sjodin PA, Margarit SM, Bar-Sagi D, Kuriyan J (July 1998). "The structural basis of the activation of Ras by Sos". Nature. 394 (6691): 337–343. Bibcode: 1998Natur.394..337B. doi: 10.1038/28548. PMID 9690470. S2CID 204998911.
- ^ Schroeder JA, Thompson MC, Gardner MM, Gendler SJ (April 2001). "Transgenic MUC1 interacts with epidermal growth factor receptor and correlates with mitogen-activated protein kinase activation in the mouse mammary gland". The Journal of Biological Chemistry. 276 (16): 13057–13064. doi: 10.1074/jbc.M011248200. PMID 11278868.
- ^ Wunderlich L, Faragó A, Buday L (January 1999). "Characterization of interactions of Nck with Sos and dynamin". Cellular Signalling. 11 (1): 25–29. doi: 10.1016/s0898-6568(98)00027-8. PMID 10206341.
- ^ Hu Q, Milfay D, Williams LT (March 1995). "Binding of NCK to SOS and activation of ras-dependent gene expression". Molecular and Cellular Biology. 15 (3): 1169–1174. doi: 10.1128/MCB.15.3.1169. PMC 230339. PMID 7862111.
- ^ Braverman LE, Quilliam LA (February 1999). "Identification of Grb4/Nckbeta, a src homology 2 and 3 domain-containing adapter protein having similar binding and biological properties to Nck". The Journal of Biological Chemistry. 274 (9): 5542–5549. doi: 10.1074/jbc.274.9.5542. PMID 10026169.
- ^ Kim MJ, Chang JS, Park SK, Hwang JI, Ryu SH, Suh PG (July 2000). "Direct interaction of SOS1 Ras exchange protein with the SH3 domain of phospholipase C-gamma1". Biochemistry. 39 (29): 8674–8682. doi: 10.1021/bi992558t. PMID 10913276.
- ^ Scholler JK, Perez-Villar JJ, O'Day K, Kanner SB (August 2000). "Engagement of the T lymphocyte antigen receptor regulates association of son-of-sevenless homologues with the SH3 domain of phospholipase Cgamma1". European Journal of Immunology. 30 (8): 2378–2387. doi: 10.1002/1521-4141(2000)30:8<2378::AID-IMMU2378>3.0.CO;2-E. PMID 10940929.
- ^ Hadari YR, Kouhara H, Lax I, Schlessinger J (July 1998). "Binding of Shp2 tyrosine phosphatase to FRS2 is essential for fibroblast growth factor-induced PC12 cell differentiation". Molecular and Cellular Biology. 18 (7): 3966–3973. doi: 10.1128/MCB.18.7.3966. PMC 108981. PMID 9632781.
- ^ Watanabe S, Take H, Takeda K, Yu ZX, Iwata N, Kajigaya S (November 2000). "Characterization of the CIN85 adaptor protein and identification of components involved in CIN85 complexes". Biochemical and Biophysical Research Communications. 278 (1): 167–174. doi: 10.1006/bbrc.2000.3760. PMID 11071869.
Further reading
- Lioubin MN, Myles GM, Carlberg K, Bowtell D, Rohrschneider LR (September 1994). "Shc, Grb2, Sos1, and a 150-kilodalton tyrosine-phosphorylated protein form complexes with Fms in hematopoietic cells". Molecular and Cellular Biology. 14 (9): 5682–5691. doi: 10.1128/mcb.14.9.5682. PMC 359093. PMID 7520523.
- Nel AE, Gupta S, Lee L, Ledbetter JA, Kanner SB (August 1995). "Ligation of the T-cell antigen receptor (TCR) induces association of hSos1, ZAP-70, phospholipase C-gamma 1, and other phosphoproteins with Grb2 and the zeta-chain of the TCR". The Journal of Biological Chemistry. 270 (31): 18428–18436. doi: 10.1074/jbc.270.31.18428. PMID 7629168.
- Pandey P, Kharbanda S, Kufe D (September 1995). "Association of the DF3/MUC1 breast cancer antigen with Grb2 and the Sos/Ras exchange protein". Cancer Research. 55 (18): 4000–4003. PMID 7664271.
- Hu Q, Milfay D, Williams LT (March 1995). "Binding of NCK to SOS and activation of ras-dependent gene expression". Molecular and Cellular Biology. 15 (3): 1169–1174. doi: 10.1128/MCB.15.3.1169. PMC 230339. PMID 7862111.
- Puil L, Liu J, Gish G, Mbamalu G, Bowtell D, Pelicci PG, et al. (February 1994). "Bcr-Abl oncoproteins bind directly to activators of the Ras signalling pathway". The EMBO Journal. 13 (4): 764–773. doi: 10.1002/j.1460-2075.1994.tb06319.x. PMC 394874. PMID 8112292.
- Li N, Batzer A, Daly R, Yajnik V, Skolnik E, Chardin P, et al. (May 1993). "Guanine-nucleotide-releasing factor hSos1 binds to Grb2 and links receptor tyrosine kinases to Ras signalling". Nature. 363 (6424): 85–88. Bibcode: 1993Natur.363...85L. doi: 10.1038/363085a0. PMID 8479541. S2CID 4323174.
- Chardin P, Camonis JH, Gale NW, van Aelst L, Schlessinger J, Wigler MH, Bar-Sagi D (May 1993). "Human Sos1: a guanine nucleotide exchange factor for Ras that binds to GRB2". Science. 260 (5112): 1338–1343. Bibcode: 1993Sci...260.1338C. doi: 10.1126/science.8493579. PMID 8493579.
- Sadoshima J, Izumo S (February 1996). "The heterotrimeric G q protein-coupled angiotensin II receptor activates p21 ras via the tyrosine kinase-Shc-Grb2-Sos pathway in cardiac myocytes". The EMBO Journal. 15 (4): 775–787. doi: 10.1002/j.1460-2075.1996.tb00413.x. PMC 450276. PMID 8631299.
- Feng GS, Ouyang YB, Hu DP, Shi ZQ, Gentz R, Ni J (May 1996). "Grap is a novel SH3-SH2-SH3 adaptor protein that couples tyrosine kinases to the Ras pathway". The Journal of Biological Chemistry. 271 (21): 12129–12132. doi: 10.1074/jbc.271.21.12129. PMID 8647802.
- Okada S, Pessin JE (October 1996). "Interactions between Src homology (SH) 2/SH3 adapter proteins and the guanylnucleotide exchange factor SOS are differentially regulated by insulin and epidermal growth factor". The Journal of Biological Chemistry. 271 (41): 25533–25538. doi: 10.1074/jbc.271.41.25533. PMID 8810325.
- Corbalan-Garcia S, Yang SS, Degenhardt KR, Bar-Sagi D (October 1996). "Identification of the mitogen-activated protein kinase phosphorylation sites on human Sos1 that regulate interaction with Grb2". Molecular and Cellular Biology. 16 (10): 5674–5682. doi: 10.1128/MCB.16.10.5674. PMC 231567. PMID 8816480.
- Sattler M, Salgia R, Shrikhande G, Verma S, Uemura N, Law SF, et al. (May 1997). "Differential signaling after beta1 integrin ligation is mediated through binding of CRKL to p120(CBL) and p110(HEF1)". The Journal of Biological Chemistry. 272 (22): 14320–14326. doi: 10.1074/jbc.272.22.14320. hdl: 20.500.12613/9173. PMID 9162067.
- Leprince C, Romero F, Cussac D, Vayssiere B, Berger R, Tavitian A, Camonis JH (June 1997). "A new member of the amphiphysin family connecting endocytosis and signal transduction pathways". The Journal of Biological Chemistry. 272 (24): 15101–15105. doi: 10.1074/jbc.272.24.15101. PMID 9182529.
- Kouhara H, Hadari YR, Spivak-Kroizman T, Schilling J, Bar-Sagi D, Lax I, Schlessinger J (May 1997). "A lipid-anchored Grb2-binding protein that links FGF-receptor activation to the Ras/MAPK signaling pathway". Cell. 89 (5): 693–702. doi: 10.1016/S0092-8674(00)80252-4. PMID 9182757. S2CID 2187363.
- Chin H, Saito T, Arai A, Yamamoto K, Kamiyama R, Miyasaka N, Miura O (October 1997). "Erythropoietin and IL-3 induce tyrosine phosphorylation of CrkL and its association with Shc, SHP-2, and Cbl in hematopoietic cells". Biochemical and Biophysical Research Communications. 239 (2): 412–417. doi: 10.1006/bbrc.1997.7480. PMID 9344843.
- Zheng J, Chen RH, Corblan-Garcia S, Cahill SM, Bar-Sagi D, Cowburn D (November 1997). "The solution structure of the pleckstrin homology domain of human SOS1. A possible structural role for the sequential association of diffuse B cell lymphoma and pleckstrin homology domains". The Journal of Biological Chemistry. 272 (48): 30340–30344. doi: 10.1074/jbc.272.48.30340. PMID 9374522.
- Li S, Kim M, Hu YL, Jalali S, Schlaepfer DD, Hunter T, et al. (November 1997). "Fluid shear stress activation of focal adhesion kinase. Linking to mitogen-activated protein kinases". The Journal of Biological Chemistry. 272 (48): 30455–30462. doi: 10.1074/jbc.272.48.30455. PMID 9374537.
- Qian X, Vass WC, Papageorge AG, Anborgh PH, Lowy DR (February 1998). "N terminus of Sos1 Ras exchange factor: critical roles for the Dbl and pleckstrin homology domains". Molecular and Cellular Biology. 18 (2): 771–778. doi: 10.1128/mcb.18.2.771. PMC 108788. PMID 9447973.
- Curto M, Frankel P, Carrero A, Foster DA (February 1998). "Novel recruitment of Shc, Grb2, and Sos by fibroblast growth factor receptor-1 in v-Src-transformed cells". Biochemical and Biophysical Research Communications. 243 (2): 555–560. doi: 10.1006/bbrc.1997.7982. PMID 9480847.
External links
- GeneReviews/NCBI/NIH/UW entry on Noonan syndrome
- http://www.noonansyndrome.org
- http://ghr.nlm.nih.gov/gene=sos1
This article incorporates text from the United States National Library of Medicine, which is in the public domain.