| |
| Names | |
|---|---|
|
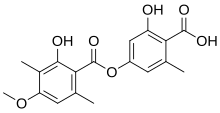
IUPAC name
2-hydroxy-4-(2-hydroxy-4-methoxy-3,6-dimethylbenzoyl)oxy-6-methylbenzoic acid
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C18H18O7 | |
| Molar mass | 346.335 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Ramalic acid is an organic compound with the molecular formula C18H18O7. [1] [2] Ramalic acid occurs as a secondary metabolite in some lichens like Ramalina pollinaria wherfrom ramalic acid has its name. [3] [4] [5] [6] Ramalic acid can be used as a dye. [6]
- ^ Abderhalden, Emil. Handbuch der biologischen arbeitsmethoden: Unter mitarbeit von zahlreichen fachmännern herausgegeben (in German). Urban & Schwarzenberg. p. 413.
- ^ "Ramalic acid". PubChem.
- ^ Jaeck, Andreas. "Ramalsäure". www.internetchemie.info.
- ^ Zopf, Wilhelm (18 May 2017). Die Flechtenstoffe (in German). BoD – Books on Demand. p. 167. ISBN 978-9925-0-7805-9.
- ^ Handbuch der chemischen Technologie: in Verbindung mit mehreren Gelehrten und Technikern (in German). F. Vieweg. p. 159.
- ^
a
b Hoppe, Heinz A. (31 December 1958). "Drogenkunde: Handbuch der pflanzlichen und tierischen Rohstoffe".
doi:
10.1515/9783111643809-020.
{{ cite journal}}: Cite journal requires|journal=( help)
- Koller, Georg; Pfeiffer, Gerhard (February 1933). "Über die Umbilikarsäure und die Ramalsäure". Monatshefte für Chemie. 62 (1–2): 241–251. doi: 10.1007/BF01518490. S2CID 105733398.

| |
| Names | |
|---|---|
|
IUPAC name
2-hydroxy-4-(2-hydroxy-4-methoxy-3,6-dimethylbenzoyl)oxy-6-methylbenzoic acid
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C18H18O7 | |
| Molar mass | 346.335 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Ramalic acid is an organic compound with the molecular formula C18H18O7. [1] [2] Ramalic acid occurs as a secondary metabolite in some lichens like Ramalina pollinaria wherfrom ramalic acid has its name. [3] [4] [5] [6] Ramalic acid can be used as a dye. [6]
- ^ Abderhalden, Emil. Handbuch der biologischen arbeitsmethoden: Unter mitarbeit von zahlreichen fachmännern herausgegeben (in German). Urban & Schwarzenberg. p. 413.
- ^ "Ramalic acid". PubChem.
- ^ Jaeck, Andreas. "Ramalsäure". www.internetchemie.info.
- ^ Zopf, Wilhelm (18 May 2017). Die Flechtenstoffe (in German). BoD – Books on Demand. p. 167. ISBN 978-9925-0-7805-9.
- ^ Handbuch der chemischen Technologie: in Verbindung mit mehreren Gelehrten und Technikern (in German). F. Vieweg. p. 159.
- ^
a
b Hoppe, Heinz A. (31 December 1958). "Drogenkunde: Handbuch der pflanzlichen und tierischen Rohstoffe".
doi:
10.1515/9783111643809-020.
{{ cite journal}}: Cite journal requires|journal=( help)
- Koller, Georg; Pfeiffer, Gerhard (February 1933). "Über die Umbilikarsäure und die Ramalsäure". Monatshefte für Chemie. 62 (1–2): 241–251. doi: 10.1007/BF01518490. S2CID 105733398.