 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
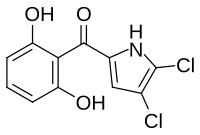
| Formula | C11H7Cl2NO3 |
| Molar mass | 272.08 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Pyoluteorin is a natural antibiotic that is biosynthesized from a hybrid nonribosomal peptide synthetase (NRPS) and polyketide synthase (PKS) pathway. [1] Pyoluteorin was first isolated in the 1950s from Pseudomonas aeruginosa strains T359 and IFO 3455 [2] and was found to be toxic against oomycetes, bacteria, fungi, and against certain plants. [3] Pyoluteorin is most notable for its toxicity against the oomycete Pythium ultimum, [4] which is a plant pathogen that causes a global loss in agriculture. Currently, pyoluteorin derivatives are being studied as an Mcl-1 antagonist in order to target cancers that have elevated Mcl-1 levels. [5]
Biosynthesis
Pyoluteorin is synthesized from an NRPS/PKS hybrid pathway. The resorcinol ring is derived from a type I PKS [6] [7] while the dichloropyrrole[ clarification needed] moiety is derived from a type II NRPS. [8] Pyoluteorin biosynthesis begins with the activation of L- proline to prolyl-AMP by the adenylation domain PltF. With prolyl-AMP still in the active site, the active form of the peptidyl carrier protein PltL binds to PltF. Then PltF catalyzes the aminoacylation of PltL by attaching L-proline to the thiol of the 4’phosphopantetheine arm of PltL. [9] Next, the dehydrogenase PltE desaturates the prolyl moiety on PltL to create pyrrolyl-PltL. The halogenation domain PltA then dichlorinates the pyrrole moiety first at position 5 and then at position 4 in a FADH2 dependent manner. [10] The dichloropyrroyl residue is then transferred to the type I PKS PltB and PltC, however, the mechanism of transfer is unknown. The addition of 3 malonyl-CoA monomers, cyclization, and release by the thioesterase PltG gives pyoluteorin.

References
- ^ Gross H, Loper JE (November 2009). "Genomics of secondary metabolite production by Pseudomonas spp". Natural Product Reports. 26 (11): 1408–46. doi: 10.1039/b817075b. PMID 19844639.
- ^ Takeda R (1958). "Structure of a new antibiotic, pyoluteorin". Journal of the American Chemical Society. 80 (17): 4749–4750. doi: 10.1021/ja01550a093.
- ^ Maurhofer M (September 10, 1991). "Influence of Enhanced Antibiotic Production in Pseudomonas fluorescens Strain CHA0 on its Disease Suppressive Capacity". Phytopathology. 82 (2): 190–195. doi: 10.1094/Phyto-82-190.
- ^ Howell CR (January 16, 1980). "Suppression of Pythium ultimum-induced damping-off of cotton seedlings by pseudomonas fluorescens and its antibiotic, pyoluteorin". Phytopathology. 70 (8): 712–715. doi: 10.1094/Phyto-70-712.
- ^ Doi K (October 2014). "Characterization of pyoluteorin derivatives as Mcl-1 antagonists". Cancer Research. 74 (19): 1805. doi: 10.1158/1538-7445.AM2014-1805.
- ^ Cuppels DA (January 15, 1986). "Biosynthesis of Pyoluteorin: A Mixed Polyketide-Tricarboxylic Acid Cycle Origin Demonstrated by [l,2-13C2]Acetate Incorporation". Zeitschrift für Naturforschung C. 41 (5–6): 532–536. doi: 10.1515/znc-1986-5-607.
- ^ Nowak-Thompson B, Gould SJ, Loper JE (December 1997). "Identification and sequence analysis of the genes encoding a polyketide synthase required for pyoluteorin biosynthesis in Pseudomonas fluorescens Pf-5". Gene. 204 (1–2): 17–24. doi: 10.1016/S0378-1119(97)00501-5. PMID 9434161.
- ^ Nowak-Thompson B, Chaney N, Wing JS, Gould SJ, Loper JE (April 1999). "Characterization of the pyoluteorin biosynthetic gene cluster of Pseudomonas fluorescens Pf-5". Journal of Bacteriology. 181 (7): 2166–74. doi: 10.1128/JB.181.7.2166-2174.1999. PMC 93630. PMID 10094695.
- ^ Thomas MG, Burkart MD, Walsh CT (February 2002). "Conversion of L-proline to pyrrolyl-2-carboxyl-S-PCP during undecylprodigiosin and pyoluteorin biosynthesis". Chemistry & Biology. 9 (2): 171–84. doi: 10.1016/S1074-5521(02)00100-X. PMID 11880032.
- ^ Dorrestein PC, Yeh E, Garneau-Tsodikova S, Kelleher NL, Walsh CT (September 2005). "Dichlorination of a pyrrolyl-S-carrier protein by FADH2-dependent halogenase PltA during pyoluteorin biosynthesis". Proceedings of the National Academy of Sciences of the United States of America. 102 (39): 13843–8. Bibcode: 2005PNAS..10213843D. doi: 10.1073/pnas.0506964102. PMC 1236592. PMID 16162666.
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C11H7Cl2NO3 |
| Molar mass | 272.08 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Pyoluteorin is a natural antibiotic that is biosynthesized from a hybrid nonribosomal peptide synthetase (NRPS) and polyketide synthase (PKS) pathway. [1] Pyoluteorin was first isolated in the 1950s from Pseudomonas aeruginosa strains T359 and IFO 3455 [2] and was found to be toxic against oomycetes, bacteria, fungi, and against certain plants. [3] Pyoluteorin is most notable for its toxicity against the oomycete Pythium ultimum, [4] which is a plant pathogen that causes a global loss in agriculture. Currently, pyoluteorin derivatives are being studied as an Mcl-1 antagonist in order to target cancers that have elevated Mcl-1 levels. [5]
Biosynthesis
Pyoluteorin is synthesized from an NRPS/PKS hybrid pathway. The resorcinol ring is derived from a type I PKS [6] [7] while the dichloropyrrole[ clarification needed] moiety is derived from a type II NRPS. [8] Pyoluteorin biosynthesis begins with the activation of L- proline to prolyl-AMP by the adenylation domain PltF. With prolyl-AMP still in the active site, the active form of the peptidyl carrier protein PltL binds to PltF. Then PltF catalyzes the aminoacylation of PltL by attaching L-proline to the thiol of the 4’phosphopantetheine arm of PltL. [9] Next, the dehydrogenase PltE desaturates the prolyl moiety on PltL to create pyrrolyl-PltL. The halogenation domain PltA then dichlorinates the pyrrole moiety first at position 5 and then at position 4 in a FADH2 dependent manner. [10] The dichloropyrroyl residue is then transferred to the type I PKS PltB and PltC, however, the mechanism of transfer is unknown. The addition of 3 malonyl-CoA monomers, cyclization, and release by the thioesterase PltG gives pyoluteorin.

References
- ^ Gross H, Loper JE (November 2009). "Genomics of secondary metabolite production by Pseudomonas spp". Natural Product Reports. 26 (11): 1408–46. doi: 10.1039/b817075b. PMID 19844639.
- ^ Takeda R (1958). "Structure of a new antibiotic, pyoluteorin". Journal of the American Chemical Society. 80 (17): 4749–4750. doi: 10.1021/ja01550a093.
- ^ Maurhofer M (September 10, 1991). "Influence of Enhanced Antibiotic Production in Pseudomonas fluorescens Strain CHA0 on its Disease Suppressive Capacity". Phytopathology. 82 (2): 190–195. doi: 10.1094/Phyto-82-190.
- ^ Howell CR (January 16, 1980). "Suppression of Pythium ultimum-induced damping-off of cotton seedlings by pseudomonas fluorescens and its antibiotic, pyoluteorin". Phytopathology. 70 (8): 712–715. doi: 10.1094/Phyto-70-712.
- ^ Doi K (October 2014). "Characterization of pyoluteorin derivatives as Mcl-1 antagonists". Cancer Research. 74 (19): 1805. doi: 10.1158/1538-7445.AM2014-1805.
- ^ Cuppels DA (January 15, 1986). "Biosynthesis of Pyoluteorin: A Mixed Polyketide-Tricarboxylic Acid Cycle Origin Demonstrated by [l,2-13C2]Acetate Incorporation". Zeitschrift für Naturforschung C. 41 (5–6): 532–536. doi: 10.1515/znc-1986-5-607.
- ^ Nowak-Thompson B, Gould SJ, Loper JE (December 1997). "Identification and sequence analysis of the genes encoding a polyketide synthase required for pyoluteorin biosynthesis in Pseudomonas fluorescens Pf-5". Gene. 204 (1–2): 17–24. doi: 10.1016/S0378-1119(97)00501-5. PMID 9434161.
- ^ Nowak-Thompson B, Chaney N, Wing JS, Gould SJ, Loper JE (April 1999). "Characterization of the pyoluteorin biosynthetic gene cluster of Pseudomonas fluorescens Pf-5". Journal of Bacteriology. 181 (7): 2166–74. doi: 10.1128/JB.181.7.2166-2174.1999. PMC 93630. PMID 10094695.
- ^ Thomas MG, Burkart MD, Walsh CT (February 2002). "Conversion of L-proline to pyrrolyl-2-carboxyl-S-PCP during undecylprodigiosin and pyoluteorin biosynthesis". Chemistry & Biology. 9 (2): 171–84. doi: 10.1016/S1074-5521(02)00100-X. PMID 11880032.
- ^ Dorrestein PC, Yeh E, Garneau-Tsodikova S, Kelleher NL, Walsh CT (September 2005). "Dichlorination of a pyrrolyl-S-carrier protein by FADH2-dependent halogenase PltA during pyoluteorin biosynthesis". Proceedings of the National Academy of Sciences of the United States of America. 102 (39): 13843–8. Bibcode: 2005PNAS..10213843D. doi: 10.1073/pnas.0506964102. PMC 1236592. PMID 16162666.