The plum pudding model is one of several historical scientific models of the atom. It was first proposed by J. J. Thomson in 1904 [1] after he discovered the electron in 1897, but before Ernest Rutherford discovered the atomic nucleus in 1911. The model tried to account for two properties of atoms then known: that there are electrons and that atoms have no net electric charge. Logically there had to be a commensurate quantity of positive charge to balance out the negative charge of the electrons, but having no clue as to the source of this positive charge, Thomson tentatively proposed it was everywhere in the atom, the atom being in the shape of a sphere. This was the mathematically simplest model. [2]
Thomson's model is popularly referred to as the plum pudding model in that the electrons are like raisins embedded in a pudding. Neither Thomson nor his colleagues used this analogy. Whoever coined the nickname mistakenly likened the positive charge to a solid whereas Thomson likened it to a liquid since he thought the electrons moved around in it. [3]
Overview
It had been known for many years that atoms contain negatively charged subatomic particles. Thomson called them "corpuscles" ( particles), but they were more commonly called "electrons", the name G. J. Stoney had coined for the " fundamental unit quantity of electricity" in 1891. [4] It had also been known for many years that atoms have no net electric charge. Thomson held that atoms must also contain some positive charge that cancels out the negative charge of their electrons. [5] [6] Thomson published his proposed model in the March 1904 edition of the Philosophical Magazine, the leading British science journal of the day. In Thomson's view:
... the atoms of the elements consist of a number of negatively electrified corpuscles enclosed in a sphere of uniform positive electrification, ... [7]
Thomson's model was the first to assign a specific inner structure to an atom, though his original description did not include mathematical formulas. [8] [9] He had followed the work of William Thomson (later Lord Kelvin) who had written a paper proposing a vortex atom in 1867, [10] J.J. Thomson abandoned his 1890 "nebular atom" hypothesis, based on the vortex theory of the atom, in which atoms were composed of immaterial vortices and suggested there were similarities between the arrangement of vortices and periodic regularity found among the chemical elements. [11] Thomson based his atomic model on known experimental evidence of the day, and in fact, followed Lord Kelvin's lead again as Kelvin had proposed a positive sphere atom a year earlier. [12] [13] Thomson's proposal, based on Kelvin's model of a positive volume charge, served to guide future experiments.
The main objective of Thomson's model after its initial publication was to account for the electrically neutral and chemically varied state of the atom. [7] Electron orbits were stable under classical mechanics. When an electron moves away from the center of the positively charged sphere it is subjected to a greater net positive inward force due to the presence of more positive charge inside its orbit (see Gauss's law). Electrons were free to rotate in rings that were further stabilized by interactions among the electrons, and spectroscopic measurements were meant to account for energy differences associated with different electron rings. As for the properties of matter, Thomson believed they arose from electrical effects. He further emphasized the need of a theory to help picture the physical and chemical aspects of an atom using the theory of corpuscles and positive charge. [14] Thomson attempted unsuccessfully to reshape his model to account for some of the major spectral lines experimentally known for several elements. [15] After the scientific discovery of radioactivity, Thomson decided to address it in his model by stating:
... we must face the problem of the constitution of the atom, and see if we can imagine a model which has in it the potentiality of explaining the remarkable properties shown by radio-active substances ... [16]
Thomson's model changed over the course of its initial publication, finally becoming a model with much more mobility containing electrons revolving in the dense field of positive charge rather than a static structure. Despite this, the colloquial nickname "plum pudding" was soon attributed to Thomson's model as the distribution of electrons within its positively charged region of space reminded many scientists of raisins, then called "plums", in the common English dessert, plum pudding. [8]
In 1909, Hans Geiger and Ernest Marsden conducted experiments where alpha particles were fired through thin sheets of gold. Their professor, Ernest Rutherford, expected to find results consistent with Thomson's atomic model. However, when the results were published in 1911, they instead implied the presence of a very small nucleus of positive charge at the center of each gold atom. [17] This led to the development of the Rutherford model of the atom. [18] Immediately after Rutherford published his results, Antonius van den Broek made the intuitive proposal that the atomic number of an atom is the total number of units of charge present in its nucleus. Henry Moseley's 1913 experiments (see Moseley's law) provided the necessary evidence to support Van den Broek's proposal. The effective nuclear charge was found to be consistent with the atomic number (Moseley found only one unit of charge difference). This work culminated in the solar-system-like Bohr model of the atom in the same year, in which a nucleus containing an atomic number of positive charges is surrounded by an equal number of electrons in orbital shells. As Thomson's model guided Rutherford's experiments, Bohr's model guided Moseley's research. The Bohr model was elaborated upon during the time of the " old quantum theory", and then subsumed by the full-fledged development of quantum mechanics. [19] [20]
Related scientific problems
As an important example of a scientific model, the plum pudding model has motivated and guided several related scientific problems.
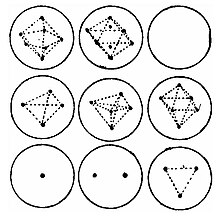
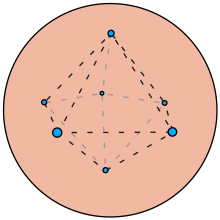
Mathematical Thomson problem
A particularly useful mathematics problem related to the plum pudding model is the optimal distribution of equal point charges on a unit sphere, called the Thomson problem. The Thomson problem is a natural consequence of the plum pudding model in the absence of its uniform positive background charge. [21] [22]
Origin of the nickname

The first known writer to compare Thomson's model to a plum pudding was an anonymous reporter who wrote an article for the British pharmaceutical magazine The Chemist and Druggist in August 1906.
While the negative electricity is concentrated on the extremely small corpuscle, the positive electricity is distributed throughout a considerable volume. An atom would thus consist of minute specks, the negative corpuscles, swimming about in a sphere of positive electrification, like raisins in a parsimonious plum-pudding, units of negative electricity being attracted toward the centre, while at the same time repelling each other. [23]
The analogy was never used by Thomson nor his colleagues. It seems to have been used by popular science writers to make the concept easier to understand for the layman. [8]
References
- ^ "Plum Pudding Model". Universe Today. 27 August 2009. Retrieved 19 December 2015.
- ^ J. J. Thomson (1907). The Corpuscular Theory of Matter, p. 103: "In default of exact knowledge of the nature of the way in which positive electricity occurs in the atom, we shall consider a case in which the positive electricity is distributed in the way most amenable to mathematical calculation, i.e., when it occurs as a sphere of uniform density, throughout which the corpuscles are distributed."
-
^ J. J. Thomson, in a letter to
Oliver Lodge dated 11 April 1904, quoted in Davis & Falconer (1997):
"With regard to positive electrification I have been in the habit of using the crude analogy of a liquid with a certain amount of cohesion, enough to keep it from flying to bits under its own repulsion. I have however always tried to keep the physical conception of the positive electricity in the background because I have always had hopes (not yet realised) of being able to do without positive electrification as a separate entity and to replace it by some property of the corpuscles."
- ^ O'Hara, J. G. (March 1975). "George Johnstone Stoney, F.R.S., and the Concept of the Electron". Notes and Records of the Royal Society of London. 29 (2): 265–276. doi: 10.1098/rsnr.1975.0018. JSTOR 531468. S2CID 145353314.
- ^ "Discovery of the electron and nucleus (article)". Khan Academy. Retrieved 9 February 2021.
- ^ Alviar-Agnew, Marissa; Agnew, Henry (4 April 2016). "4.3: The Nuclear Atom". Introductory Chemistry. LibreTexts. Retrieved 9 February 2021.
- ^ a b Thomson, J. J. (March 1904). "On the Structure of the Atom: an Investigation of the Stability and Periods of Oscillation of a number of Corpuscles arranged at equal intervals around the Circumference of a Circle; with Application of the Results to the Theory of Atomic Structure". Philosophical Magazine. Sixth series. 7 (39): 237–265. doi: 10.1080/14786440409463107. Archived (PDF) from the original on 2022-10-09.
- ^ a b c Giora Hon; Bernard R. Goldstein (6 September 2013). "J. J. Thomson's plum-pudding atomic model: The making of a scientific myth". Annalen der Physik. 525 (8–9): A129–A133. doi: 10.1002/andp.201300732.
- ^ Thomson, J. J. (December 1899). "On the masses of the ions in gases at low pressures". The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science. 48 (295): 547–567. doi: 10.1080/14786449908621447.
- ^ Thomson, William (1869). "On Vortex Atoms". Proceedings of the Royal Society of Edinburgh. 6: 94–105. doi: 10.1017/S0370164600045430.
- ^ Kragh, Helge (2002). Quantum Generations: A History of Physics in the Twentieth Century (Reprint ed.). Princeton University Press. pp. 43–45. ISBN 978-0691095523.
- ^ Models of the Atom, Michael Fowler, University of Virginia https://galileo.phys.virginia.edu/classes/252/more_atoms.html#Plum%20Pudding
- ^ Kumar, Manjit, Quantum Einstein, Bohr and the Great Debate, ISBN 978-0393339888, 2008.
- ^ E. R. (1908). "The Corpuscular Theory of Matter". Nature. 77 (2005): 505–506. Bibcode: 1908Natur..77..505R. doi: 10.1038/077505a0. S2CID 36356538.
- ^ Models of the Atom, Michael Fowler, University of Virginia https://galileo.phys.virginia.edu/classes/252/more_atoms.html#Plum%20Pudding
- ^ Thomson, J. J. (1904). Electricity and Matter. Mrs. Hepsa Ely Silliman Memorial Lectures. New Haven: Yale University Press. ISBN 978-0-686-83533-2.
- ^ Angelo, Joseph A. (2004). Nuclear Technology. Greenwood Publishing. p. 110. ISBN 978-1-57356-336-9.
- ^ Heilbron, John L. (2013). "The path to the quantum atom". Nature. 498 (7452): 27–30. doi: 10.1038/498027a. PMID 23739408. S2CID 4355108.
- ^ Hentschel, Klaus (2009). "Zeeman Effect". In Greenberger, Daniel; Hentschel, Klaus; Weinert, Friedel (eds.). Compendium of Quantum Physics. Berlin, Heidelberg: Springer. pp. 862–864. doi: 10.1007/978-3-540-70626-7_241. ISBN 978-3-540-70622-9.
- ^ Eckert, Michael (April 2014). "How Sommerfeld extended Bohr's model of the atom (1913–1916)". The European Physical Journal H. 39 (2): 141–156. Bibcode: 2014EPJH...39..141E. doi: 10.1140/epjh/e2013-40052-4. S2CID 256006474.
- ^ Levin, Y.; Arenzon, J. J. (2003). "Why charges go to the Surface: A generalized Thomson Problem". Europhys. Lett. 63 (3): 415–418. arXiv: cond-mat/0302524. Bibcode: 2003EL.....63..415L. doi: 10.1209/epl/i2003-00546-1. S2CID 250764497.
-
^ Roth, J. (2007-10-24).
"Description of a highly symmetric polytope observed in Thomson's problem of charges on a hypersphere". Physical Review E. 76 (4): 047702.
Bibcode:
2007PhRvE..76d7702R.
doi:
10.1103/PhysRevE.76.047702.
ISSN
1539-3755.
PMID
17995142.
Although Thomson's model has been outdated for a long time by quantum mechanics, his problem of placing charges on a sphere is still noteworthy.
- ^ "What is Matter?". The Chemist and Druggist. 69 (8): 329–330. 25 August 1906.
Bibliography
- J. J. Thomson (1907). The Corpuscular Theory of Matter. Charles Scribner's Sons.


The plum pudding model is one of several historical scientific models of the atom. It was first proposed by J. J. Thomson in 1904 [1] after he discovered the electron in 1897, but before Ernest Rutherford discovered the atomic nucleus in 1911. The model tried to account for two properties of atoms then known: that there are electrons and that atoms have no net electric charge. Logically there had to be a commensurate quantity of positive charge to balance out the negative charge of the electrons, but having no clue as to the source of this positive charge, Thomson tentatively proposed it was everywhere in the atom, the atom being in the shape of a sphere. This was the mathematically simplest model. [2]
Thomson's model is popularly referred to as the plum pudding model in that the electrons are like raisins embedded in a pudding. Neither Thomson nor his colleagues used this analogy. Whoever coined the nickname mistakenly likened the positive charge to a solid whereas Thomson likened it to a liquid since he thought the electrons moved around in it. [3]
Overview
It had been known for many years that atoms contain negatively charged subatomic particles. Thomson called them "corpuscles" ( particles), but they were more commonly called "electrons", the name G. J. Stoney had coined for the " fundamental unit quantity of electricity" in 1891. [4] It had also been known for many years that atoms have no net electric charge. Thomson held that atoms must also contain some positive charge that cancels out the negative charge of their electrons. [5] [6] Thomson published his proposed model in the March 1904 edition of the Philosophical Magazine, the leading British science journal of the day. In Thomson's view:
... the atoms of the elements consist of a number of negatively electrified corpuscles enclosed in a sphere of uniform positive electrification, ... [7]
Thomson's model was the first to assign a specific inner structure to an atom, though his original description did not include mathematical formulas. [8] [9] He had followed the work of William Thomson (later Lord Kelvin) who had written a paper proposing a vortex atom in 1867, [10] J.J. Thomson abandoned his 1890 "nebular atom" hypothesis, based on the vortex theory of the atom, in which atoms were composed of immaterial vortices and suggested there were similarities between the arrangement of vortices and periodic regularity found among the chemical elements. [11] Thomson based his atomic model on known experimental evidence of the day, and in fact, followed Lord Kelvin's lead again as Kelvin had proposed a positive sphere atom a year earlier. [12] [13] Thomson's proposal, based on Kelvin's model of a positive volume charge, served to guide future experiments.
The main objective of Thomson's model after its initial publication was to account for the electrically neutral and chemically varied state of the atom. [7] Electron orbits were stable under classical mechanics. When an electron moves away from the center of the positively charged sphere it is subjected to a greater net positive inward force due to the presence of more positive charge inside its orbit (see Gauss's law). Electrons were free to rotate in rings that were further stabilized by interactions among the electrons, and spectroscopic measurements were meant to account for energy differences associated with different electron rings. As for the properties of matter, Thomson believed they arose from electrical effects. He further emphasized the need of a theory to help picture the physical and chemical aspects of an atom using the theory of corpuscles and positive charge. [14] Thomson attempted unsuccessfully to reshape his model to account for some of the major spectral lines experimentally known for several elements. [15] After the scientific discovery of radioactivity, Thomson decided to address it in his model by stating:
... we must face the problem of the constitution of the atom, and see if we can imagine a model which has in it the potentiality of explaining the remarkable properties shown by radio-active substances ... [16]
Thomson's model changed over the course of its initial publication, finally becoming a model with much more mobility containing electrons revolving in the dense field of positive charge rather than a static structure. Despite this, the colloquial nickname "plum pudding" was soon attributed to Thomson's model as the distribution of electrons within its positively charged region of space reminded many scientists of raisins, then called "plums", in the common English dessert, plum pudding. [8]
In 1909, Hans Geiger and Ernest Marsden conducted experiments where alpha particles were fired through thin sheets of gold. Their professor, Ernest Rutherford, expected to find results consistent with Thomson's atomic model. However, when the results were published in 1911, they instead implied the presence of a very small nucleus of positive charge at the center of each gold atom. [17] This led to the development of the Rutherford model of the atom. [18] Immediately after Rutherford published his results, Antonius van den Broek made the intuitive proposal that the atomic number of an atom is the total number of units of charge present in its nucleus. Henry Moseley's 1913 experiments (see Moseley's law) provided the necessary evidence to support Van den Broek's proposal. The effective nuclear charge was found to be consistent with the atomic number (Moseley found only one unit of charge difference). This work culminated in the solar-system-like Bohr model of the atom in the same year, in which a nucleus containing an atomic number of positive charges is surrounded by an equal number of electrons in orbital shells. As Thomson's model guided Rutherford's experiments, Bohr's model guided Moseley's research. The Bohr model was elaborated upon during the time of the " old quantum theory", and then subsumed by the full-fledged development of quantum mechanics. [19] [20]
Related scientific problems
As an important example of a scientific model, the plum pudding model has motivated and guided several related scientific problems.
Mathematical Thomson problem
A particularly useful mathematics problem related to the plum pudding model is the optimal distribution of equal point charges on a unit sphere, called the Thomson problem. The Thomson problem is a natural consequence of the plum pudding model in the absence of its uniform positive background charge. [21] [22]
Origin of the nickname

The first known writer to compare Thomson's model to a plum pudding was an anonymous reporter who wrote an article for the British pharmaceutical magazine The Chemist and Druggist in August 1906.
While the negative electricity is concentrated on the extremely small corpuscle, the positive electricity is distributed throughout a considerable volume. An atom would thus consist of minute specks, the negative corpuscles, swimming about in a sphere of positive electrification, like raisins in a parsimonious plum-pudding, units of negative electricity being attracted toward the centre, while at the same time repelling each other. [23]
The analogy was never used by Thomson nor his colleagues. It seems to have been used by popular science writers to make the concept easier to understand for the layman. [8]
References
- ^ "Plum Pudding Model". Universe Today. 27 August 2009. Retrieved 19 December 2015.
- ^ J. J. Thomson (1907). The Corpuscular Theory of Matter, p. 103: "In default of exact knowledge of the nature of the way in which positive electricity occurs in the atom, we shall consider a case in which the positive electricity is distributed in the way most amenable to mathematical calculation, i.e., when it occurs as a sphere of uniform density, throughout which the corpuscles are distributed."
-
^ J. J. Thomson, in a letter to
Oliver Lodge dated 11 April 1904, quoted in Davis & Falconer (1997):
"With regard to positive electrification I have been in the habit of using the crude analogy of a liquid with a certain amount of cohesion, enough to keep it from flying to bits under its own repulsion. I have however always tried to keep the physical conception of the positive electricity in the background because I have always had hopes (not yet realised) of being able to do without positive electrification as a separate entity and to replace it by some property of the corpuscles."
- ^ O'Hara, J. G. (March 1975). "George Johnstone Stoney, F.R.S., and the Concept of the Electron". Notes and Records of the Royal Society of London. 29 (2): 265–276. doi: 10.1098/rsnr.1975.0018. JSTOR 531468. S2CID 145353314.
- ^ "Discovery of the electron and nucleus (article)". Khan Academy. Retrieved 9 February 2021.
- ^ Alviar-Agnew, Marissa; Agnew, Henry (4 April 2016). "4.3: The Nuclear Atom". Introductory Chemistry. LibreTexts. Retrieved 9 February 2021.
- ^ a b Thomson, J. J. (March 1904). "On the Structure of the Atom: an Investigation of the Stability and Periods of Oscillation of a number of Corpuscles arranged at equal intervals around the Circumference of a Circle; with Application of the Results to the Theory of Atomic Structure". Philosophical Magazine. Sixth series. 7 (39): 237–265. doi: 10.1080/14786440409463107. Archived (PDF) from the original on 2022-10-09.
- ^ a b c Giora Hon; Bernard R. Goldstein (6 September 2013). "J. J. Thomson's plum-pudding atomic model: The making of a scientific myth". Annalen der Physik. 525 (8–9): A129–A133. doi: 10.1002/andp.201300732.
- ^ Thomson, J. J. (December 1899). "On the masses of the ions in gases at low pressures". The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science. 48 (295): 547–567. doi: 10.1080/14786449908621447.
- ^ Thomson, William (1869). "On Vortex Atoms". Proceedings of the Royal Society of Edinburgh. 6: 94–105. doi: 10.1017/S0370164600045430.
- ^ Kragh, Helge (2002). Quantum Generations: A History of Physics in the Twentieth Century (Reprint ed.). Princeton University Press. pp. 43–45. ISBN 978-0691095523.
- ^ Models of the Atom, Michael Fowler, University of Virginia https://galileo.phys.virginia.edu/classes/252/more_atoms.html#Plum%20Pudding
- ^ Kumar, Manjit, Quantum Einstein, Bohr and the Great Debate, ISBN 978-0393339888, 2008.
- ^ E. R. (1908). "The Corpuscular Theory of Matter". Nature. 77 (2005): 505–506. Bibcode: 1908Natur..77..505R. doi: 10.1038/077505a0. S2CID 36356538.
- ^ Models of the Atom, Michael Fowler, University of Virginia https://galileo.phys.virginia.edu/classes/252/more_atoms.html#Plum%20Pudding
- ^ Thomson, J. J. (1904). Electricity and Matter. Mrs. Hepsa Ely Silliman Memorial Lectures. New Haven: Yale University Press. ISBN 978-0-686-83533-2.
- ^ Angelo, Joseph A. (2004). Nuclear Technology. Greenwood Publishing. p. 110. ISBN 978-1-57356-336-9.
- ^ Heilbron, John L. (2013). "The path to the quantum atom". Nature. 498 (7452): 27–30. doi: 10.1038/498027a. PMID 23739408. S2CID 4355108.
- ^ Hentschel, Klaus (2009). "Zeeman Effect". In Greenberger, Daniel; Hentschel, Klaus; Weinert, Friedel (eds.). Compendium of Quantum Physics. Berlin, Heidelberg: Springer. pp. 862–864. doi: 10.1007/978-3-540-70626-7_241. ISBN 978-3-540-70622-9.
- ^ Eckert, Michael (April 2014). "How Sommerfeld extended Bohr's model of the atom (1913–1916)". The European Physical Journal H. 39 (2): 141–156. Bibcode: 2014EPJH...39..141E. doi: 10.1140/epjh/e2013-40052-4. S2CID 256006474.
- ^ Levin, Y.; Arenzon, J. J. (2003). "Why charges go to the Surface: A generalized Thomson Problem". Europhys. Lett. 63 (3): 415–418. arXiv: cond-mat/0302524. Bibcode: 2003EL.....63..415L. doi: 10.1209/epl/i2003-00546-1. S2CID 250764497.
-
^ Roth, J. (2007-10-24).
"Description of a highly symmetric polytope observed in Thomson's problem of charges on a hypersphere". Physical Review E. 76 (4): 047702.
Bibcode:
2007PhRvE..76d7702R.
doi:
10.1103/PhysRevE.76.047702.
ISSN
1539-3755.
PMID
17995142.
Although Thomson's model has been outdated for a long time by quantum mechanics, his problem of placing charges on a sphere is still noteworthy.
- ^ "What is Matter?". The Chemist and Druggist. 69 (8): 329–330. 25 August 1906.
Bibliography
- J. J. Thomson (1907). The Corpuscular Theory of Matter. Charles Scribner's Sons.