| |
| |
| Names | |
|---|---|
|
Preferred IUPAC name
Benzene-1,2-dicarboxylic acid | |
| Other names
1,2-Benzenedioic acid
Phthalic acid Benzene-1,2-dioic acid ortho-Phthalic acid Dicarboxylbenzene Carboxylbenzoic acid 1,2-Phenylenedicarboxylic acid Phenylene-1,2-dicarboxylic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.001.703 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C8H6O4 | |
| Molar mass | 166.132 g/mol |
| Appearance | white solid |
| Density | 1.593 g/cm3, solid |
| Melting point | 207 °C (405 °F; 480 K) [3] |
| 0.6 g / 100 mL [1] | |
| Acidity (pKa) | 2.89, 5.51 [2] |
| -83.61·10−6 cm3/mol | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related
carboxylic acids
|
Isophthalic acid Terephthalic acid |
Related compounds
|
Phthalic anhydride Phthalimide Phthalhydrazide Phthaloyl chloride Benzene-1,2- dicarboxaldehyde |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
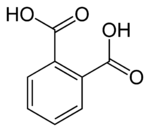
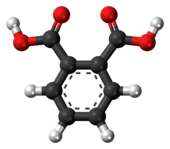
In organic chemistry, phthalic acid is an aromatic dicarboxylic acid, with formula C6H4(CO2H)2 and structure HO(O)C− C6H4− C(O)OH. Although phthalic acid is of modest commercial importance, the closely related derivative phthalic anhydride is a commodity chemical produced on a large scale. [4] Phthalic acid is one of three isomers of benzenedicarboxylic acid, the others being isophthalic acid and terephthalic acid.
Phthalic acid is produced by the catalytic oxidation of naphthalene or ortho-xylene directly to phthalic anhydride and a subsequent hydrolysis of the anhydride. [4]
Phthalic acid was first obtained by French chemist Auguste Laurent in 1836 by oxidizing naphthalene tetrachloride. [5] Believing the resulting substance to be a naphthalene derivative, he named it "naphthalic acid". [5] [6] [7] After the Swiss chemist Jean Charles Galissard de Marignac determined its correct formula, [8] Laurent gave it its present name. [5] [9] [10] Manufacturing methods in the nineteenth century included oxidation of naphthalene tetrachloride with nitric acid, or, better, oxidation of the hydrocarbon with fuming sulfuric acid, using mercury or mercury(II) sulfate as a catalyst. [5]
Naphthalene, on oxidation with potassium permanganate or potassium dichromate, gives Phthalic anhydride, [11] which, through hydrolysis with hot water, gives Phthalic acid. [12]

It is a dibasic acid, with pKas of 2.89 and 5.51. The monopotassium salt, potassium hydrogen phthalate is a standard acid in analytical chemistry. Typically phthalate esters are prepared from the widely available phthalic anhydride. Reduction of phthalic acid with sodium amalgam in the presence of water gives the 1,3-cyclohexadiene derivative. [13]
The toxicity of phthalic acid is moderate with LD50 (mouse) of 550 mg/kg.
The bacteria Pseudomonas sp. P1 degrades phthalic acid. [14]
- Isophthalic acid
- Phthalate
- Phthalic anhydride
- Potassium hydrogen phthalate, a primary standard for acid–base titrations
- Terephthalic acid
- ^ "PHTHALIC ACID". hazard.com.
- ^ Brown, H.C., et al., in Baude, E.A. and Nachod, F.C., Determination of Organic Structures by Physical Methods, Academic Press, New York, 1955.
- ^ Several melting points are reported, for example: (i) 480. K (NIST website), (ii) 210−211 °C with decomposition (Sigma-Aldrich on-line), (iii) 191 °C in a sealed tube (Ullmann's Encyclopedia of Industrial Chemistry), (iv) 230 °C with conversion to phthalic anhydride and water (J.T.Baker MSDS).
- ^ a b Lorz, Peter M.; Towae, Friedrich K.; Enke, Walter; et al. (2007). "Phthalic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a20_181.pub2. ISBN 978-3527306732.
- ^ a b c d Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica. Vol. 21 (11th ed.). Cambridge University Press. pp. 545–546.
- ^ Auguste Laurent (1836) "Sur l'acide naphtalique et ses combinaisons" (On naphthalic acid and its compounds), Annales de Chimie et de Physique, 61 : 113-125. (Note: The empirical formulae of the compounds that were analyzed in this article are incorrect, in part because, during this period, chemists used incorrect atomic masses for carbon (6 instead of 12) and other elements.)
- ^ Reprinted in German as: Auguste Laurent (1836) "Ueber Naphthalinsäure und ihre Verbindungen" (On naphthalenic acid and its compounds), Annalen der Pharmacie, 19 (1) : 38-50; for the preparation of phthalic acid, see page 41.
- ^ C. de Marignac (1841) "Ueber die Naphtalinsäure und ein bei ihrer Darstellung entstehendes flüchtiges Produkt" ("On naphthalinic acid and a volatile product that arises during its preparation"), Annalen der Chemie und Pharmacie, 38 (1) : 13-20. (Note: Again, Marignac's empirical formulae are wrong because chemists at this time used incorrect atomic masses.)
- ^ Auguste Laurent (1841) "Sur de nouvelles combinaisons nitrogénées de la naphtaline et sur les acides phtalique et nitrophtalique" (On new nitrogenous compounds of naphthalene, and on phthalic acid and nitrophthalic acid), Revue Scientifique et Industrielle, 6 : 76-99; on page 92, Laurent coins the name "acide phtalique" (phthalic acid) and admits that his earlier empirical formula for phthalic acid was wrong.
- ^ Reprinted in German as: Auguste Laurent (1842) "Ueber neue stickstoffhaltige Verbindungen des Naphtalins, über Phtalinsäure und Nitrophtalinsäure" (On new nitrogenous compounds of naphthalene, on phthalic acid and nitrophthalic acid), Annalen der Chemie und Pharmacie, 41 (1) : 98-114; on page 108, Laurent coins the name "Phtalinsäure" (phthalic acid).
- ^ "Oxidation of Naphthalene to Phthalic Anhydride". public.websites.umich.edu. Retrieved 2024-04-06.
- ^ Noller, Carl R. (1965). Chemistry of Organic Compounds (3rd ed.). Philadelphia: W. B. Saunders. p. 602.
- ^ Richard N. McDonald and Charles E. Reineke (1988). "trans-1,2-Dihydrophthalic Acid". Organic Syntheses; Collected Volumes, vol. 6, p. 461.
- ^ Ishtiaq Ali, Muhammad (2011). Microbial degradation of polyvinyl chloride plastics (PDF) (Ph.D.). Quaid-i-Azam University. p. 47.
- Merck Index, 9th ed, #7178

| |

| |
| Names | |
|---|---|
|
Preferred IUPAC name
Benzene-1,2-dicarboxylic acid | |
| Other names
1,2-Benzenedioic acid
Phthalic acid Benzene-1,2-dioic acid ortho-Phthalic acid Dicarboxylbenzene Carboxylbenzoic acid 1,2-Phenylenedicarboxylic acid Phenylene-1,2-dicarboxylic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.001.703 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C8H6O4 | |
| Molar mass | 166.132 g/mol |
| Appearance | white solid |
| Density | 1.593 g/cm3, solid |
| Melting point | 207 °C (405 °F; 480 K) [3] |
| 0.6 g / 100 mL [1] | |
| Acidity (pKa) | 2.89, 5.51 [2] |
| -83.61·10−6 cm3/mol | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related
carboxylic acids
|
Isophthalic acid Terephthalic acid |
Related compounds
|
Phthalic anhydride Phthalimide Phthalhydrazide Phthaloyl chloride Benzene-1,2- dicarboxaldehyde |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
In organic chemistry, phthalic acid is an aromatic dicarboxylic acid, with formula C6H4(CO2H)2 and structure HO(O)C− C6H4− C(O)OH. Although phthalic acid is of modest commercial importance, the closely related derivative phthalic anhydride is a commodity chemical produced on a large scale. [4] Phthalic acid is one of three isomers of benzenedicarboxylic acid, the others being isophthalic acid and terephthalic acid.
Phthalic acid is produced by the catalytic oxidation of naphthalene or ortho-xylene directly to phthalic anhydride and a subsequent hydrolysis of the anhydride. [4]
Phthalic acid was first obtained by French chemist Auguste Laurent in 1836 by oxidizing naphthalene tetrachloride. [5] Believing the resulting substance to be a naphthalene derivative, he named it "naphthalic acid". [5] [6] [7] After the Swiss chemist Jean Charles Galissard de Marignac determined its correct formula, [8] Laurent gave it its present name. [5] [9] [10] Manufacturing methods in the nineteenth century included oxidation of naphthalene tetrachloride with nitric acid, or, better, oxidation of the hydrocarbon with fuming sulfuric acid, using mercury or mercury(II) sulfate as a catalyst. [5]
Naphthalene, on oxidation with potassium permanganate or potassium dichromate, gives Phthalic anhydride, [11] which, through hydrolysis with hot water, gives Phthalic acid. [12]

It is a dibasic acid, with pKas of 2.89 and 5.51. The monopotassium salt, potassium hydrogen phthalate is a standard acid in analytical chemistry. Typically phthalate esters are prepared from the widely available phthalic anhydride. Reduction of phthalic acid with sodium amalgam in the presence of water gives the 1,3-cyclohexadiene derivative. [13]
The toxicity of phthalic acid is moderate with LD50 (mouse) of 550 mg/kg.
The bacteria Pseudomonas sp. P1 degrades phthalic acid. [14]
- Isophthalic acid
- Phthalate
- Phthalic anhydride
- Potassium hydrogen phthalate, a primary standard for acid–base titrations
- Terephthalic acid
- ^ "PHTHALIC ACID". hazard.com.
- ^ Brown, H.C., et al., in Baude, E.A. and Nachod, F.C., Determination of Organic Structures by Physical Methods, Academic Press, New York, 1955.
- ^ Several melting points are reported, for example: (i) 480. K (NIST website), (ii) 210−211 °C with decomposition (Sigma-Aldrich on-line), (iii) 191 °C in a sealed tube (Ullmann's Encyclopedia of Industrial Chemistry), (iv) 230 °C with conversion to phthalic anhydride and water (J.T.Baker MSDS).
- ^ a b Lorz, Peter M.; Towae, Friedrich K.; Enke, Walter; et al. (2007). "Phthalic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a20_181.pub2. ISBN 978-3527306732.
- ^ a b c d Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica. Vol. 21 (11th ed.). Cambridge University Press. pp. 545–546.
- ^ Auguste Laurent (1836) "Sur l'acide naphtalique et ses combinaisons" (On naphthalic acid and its compounds), Annales de Chimie et de Physique, 61 : 113-125. (Note: The empirical formulae of the compounds that were analyzed in this article are incorrect, in part because, during this period, chemists used incorrect atomic masses for carbon (6 instead of 12) and other elements.)
- ^ Reprinted in German as: Auguste Laurent (1836) "Ueber Naphthalinsäure und ihre Verbindungen" (On naphthalenic acid and its compounds), Annalen der Pharmacie, 19 (1) : 38-50; for the preparation of phthalic acid, see page 41.
- ^ C. de Marignac (1841) "Ueber die Naphtalinsäure und ein bei ihrer Darstellung entstehendes flüchtiges Produkt" ("On naphthalinic acid and a volatile product that arises during its preparation"), Annalen der Chemie und Pharmacie, 38 (1) : 13-20. (Note: Again, Marignac's empirical formulae are wrong because chemists at this time used incorrect atomic masses.)
- ^ Auguste Laurent (1841) "Sur de nouvelles combinaisons nitrogénées de la naphtaline et sur les acides phtalique et nitrophtalique" (On new nitrogenous compounds of naphthalene, and on phthalic acid and nitrophthalic acid), Revue Scientifique et Industrielle, 6 : 76-99; on page 92, Laurent coins the name "acide phtalique" (phthalic acid) and admits that his earlier empirical formula for phthalic acid was wrong.
- ^ Reprinted in German as: Auguste Laurent (1842) "Ueber neue stickstoffhaltige Verbindungen des Naphtalins, über Phtalinsäure und Nitrophtalinsäure" (On new nitrogenous compounds of naphthalene, on phthalic acid and nitrophthalic acid), Annalen der Chemie und Pharmacie, 41 (1) : 98-114; on page 108, Laurent coins the name "Phtalinsäure" (phthalic acid).
- ^ "Oxidation of Naphthalene to Phthalic Anhydride". public.websites.umich.edu. Retrieved 2024-04-06.
- ^ Noller, Carl R. (1965). Chemistry of Organic Compounds (3rd ed.). Philadelphia: W. B. Saunders. p. 602.
- ^ Richard N. McDonald and Charles E. Reineke (1988). "trans-1,2-Dihydrophthalic Acid". Organic Syntheses; Collected Volumes, vol. 6, p. 461.
- ^ Ishtiaq Ali, Muhammad (2011). Microbial degradation of polyvinyl chloride plastics (PDF) (Ph.D.). Quaid-i-Azam University. p. 47.
- Merck Index, 9th ed, #7178
