Perfluoropolyethers (PFPEs) are a class of organofluorine compound. Some types are synthetic liquid lubricants that have been used in the aerospace industry for over 30 years. [1] The main properties of PFPE are being temperature resistant between −58 °C (215 K) and 257 °C (530 K) (depending on specific composites), having very low outgassing compared to other fluids ( vapour pressure of 6×10−8 Torr) and having a dielectric strength of around 15.7 MV/m. [2]
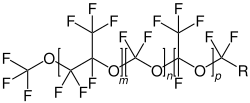
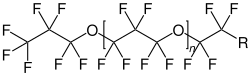
Perfluoropolyethers consists of a polymer chain in which monomers consisting of perfluoro- alkyl groups are joined by ether linkages. The bonds between carbon and oxygen or fluorine are strong. [3] Perfluoropolyethers are a type of PFAS. [4]
The thermal and chemical stability of PFPEs along with a vapor–liquid equilibrium of 230 °C when mixed with the right composites make it a suitable candidate for vapor phase soldering technologies. [5]
History
Perfluoropolyethers were developed in the early 1960s for the USAF, who needed a lubricant that would not react with liquid or gaseous oxygen (O2). [6]
Manufacture
Each type of perfluoropolyether is synthesized differently: PFPE-K is obtained from HFPO by anionic polymerization. For PFPE-Y and PFBE-Z, photooxidation of hexafluoropropylene and tetrafluoroethylene, respectively, is applied. PFPE-D is synthesized by anionic ring-opening polymerization of 2,2,3,3-tetrafluorooxetane. PFPE-A is obtained by an initial esterification of polyethylene glycol with a perfluoroacyl fluoride. Then, it is converted into a fully fluorined polymer using direct fluorination. [3] [7]
- Structural formulas of different types
-
PFPE-K
-
PFPE-Y
-
PFPE-Z
-
PFPE-D
-
PFPE-A
Properties
PFPEs are chemically inert to many acids and oxidants (like fuming sulfuric acid (oleum, SO3), chlorine gas, oxygen) and solvents, etc.
PFPEs are non-toxic under normal conditions, nonflammable, and exhibit unusually high load carry capabilities. [6] [8]: 275 PFPEs can withstand gamma ray degradation. [6]
Electrical resistivity is 1014 ohm/cm (20 °C (68 °F)) [6]
Applications
Generally, PFPEs may be used as lubricant in all sorts of bearing, plug valves, gaskets, chains, and joint bearing applications, where oxygen inertness of a material is a requirement. [6] [8] Examples include aircraft fuel systems, mechanical components of devices used in airspace, deep space or high vacuum and at cryogenic temperatures. [6]
PFPEs may be used in mold release agent for plastic injection molding. [6]
As top coating lubricant on computer disc drives and Scanning Electron Microscope [6] [8]: 275
As anti-galling compounds. [6]
As fluid medium in ferrofluidic seals. [6]
Semiconductor industry
In the semiconductor industry, PFPEs may be used as a vacuum grease or in plasma etching equipment, or for robots used in semiconductor wafer handling, clean rooms, and other commercial environments. [6]
See also
- Krytox, a PFPE-based lubricant
References
- ^ Bradley Shogrin (August 1995). "Spontaneous Dewetting of a Perfluoropolyether" (PDF). National Aeronautics and Space Administration. Retrieved November 29, 2017.
- ^ "Fomblin® PFPE Lubricants".
- ^ a b Fluorochemie. "Brief Introduction of PFPE Synthesis Methods". Fuzhou Topda New Material Co., Ltd. Retrieved June 27, 2022.
- ^ Kwiatkowski, Carol F.; Andrews, David Q.; Birnbaum, Linda S.; Bruton, Thomas A.; DeWitt, Jamie C.; Knappe, Detlef R. U.; Maffini, Maricel V.; Miller, Mark F.; Pelch, Katherine E.; Reade, Anna; Soehl, Anna; Trier, Xenia; Venier, Marta; Wagner, Charlotte C.; Wang, Zhanyun; Blum, Arlene (11 August 2020). "Scientific Basis for Managing PFAS as a Chemical Class". Environmental Science & Technology Letters. 7 (8): 532–543. doi: 10.1021/acs.estlett.0c00255. hdl: 20.500.11850/438999.
-
^
"Archived copy" (PDF). Archived from
the original (PDF) on 2021-02-27. Retrieved 2020-08-23.
{{ cite web}}: CS1 maint: archived copy as title ( link) - ^ a b c d e f g h i j k "HUSKEY Specialty lubricants" (PDF). HUSK-ITT Corporation.
- ^ Synthesis Report on Understanding Perfluoropolyethers (PFPEs) and Their Life Cycle (PDF). OECD. 2024.
- ^ a b c Ash, Michael (2004). Handbook of green chemicals. Irene Ash (2 ed.). Endicott, NY: Synapse Information Resources. ISBN 978-1-934764-29-9. OCLC 747426845.
Perfluoropolyethers (PFPEs) are a class of organofluorine compound. Some types are synthetic liquid lubricants that have been used in the aerospace industry for over 30 years. [1] The main properties of PFPE are being temperature resistant between −58 °C (215 K) and 257 °C (530 K) (depending on specific composites), having very low outgassing compared to other fluids ( vapour pressure of 6×10−8 Torr) and having a dielectric strength of around 15.7 MV/m. [2]
Perfluoropolyethers consists of a polymer chain in which monomers consisting of perfluoro- alkyl groups are joined by ether linkages. The bonds between carbon and oxygen or fluorine are strong. [3] Perfluoropolyethers are a type of PFAS. [4]
The thermal and chemical stability of PFPEs along with a vapor–liquid equilibrium of 230 °C when mixed with the right composites make it a suitable candidate for vapor phase soldering technologies. [5]
History
Perfluoropolyethers were developed in the early 1960s for the USAF, who needed a lubricant that would not react with liquid or gaseous oxygen (O2). [6]
Manufacture
Each type of perfluoropolyether is synthesized differently: PFPE-K is obtained from HFPO by anionic polymerization. For PFPE-Y and PFBE-Z, photooxidation of hexafluoropropylene and tetrafluoroethylene, respectively, is applied. PFPE-D is synthesized by anionic ring-opening polymerization of 2,2,3,3-tetrafluorooxetane. PFPE-A is obtained by an initial esterification of polyethylene glycol with a perfluoroacyl fluoride. Then, it is converted into a fully fluorined polymer using direct fluorination. [3] [7]
- Structural formulas of different types
-
PFPE-K
-
PFPE-Y
-
PFPE-Z
-
PFPE-D
-
PFPE-A
Properties
PFPEs are chemically inert to many acids and oxidants (like fuming sulfuric acid (oleum, SO3), chlorine gas, oxygen) and solvents, etc.
PFPEs are non-toxic under normal conditions, nonflammable, and exhibit unusually high load carry capabilities. [6] [8]: 275 PFPEs can withstand gamma ray degradation. [6]
Electrical resistivity is 1014 ohm/cm (20 °C (68 °F)) [6]
Applications
Generally, PFPEs may be used as lubricant in all sorts of bearing, plug valves, gaskets, chains, and joint bearing applications, where oxygen inertness of a material is a requirement. [6] [8] Examples include aircraft fuel systems, mechanical components of devices used in airspace, deep space or high vacuum and at cryogenic temperatures. [6]
PFPEs may be used in mold release agent for plastic injection molding. [6]
As top coating lubricant on computer disc drives and Scanning Electron Microscope [6] [8]: 275
As anti-galling compounds. [6]
As fluid medium in ferrofluidic seals. [6]
Semiconductor industry
In the semiconductor industry, PFPEs may be used as a vacuum grease or in plasma etching equipment, or for robots used in semiconductor wafer handling, clean rooms, and other commercial environments. [6]
See also
- Krytox, a PFPE-based lubricant
References
- ^ Bradley Shogrin (August 1995). "Spontaneous Dewetting of a Perfluoropolyether" (PDF). National Aeronautics and Space Administration. Retrieved November 29, 2017.
- ^ "Fomblin® PFPE Lubricants".
- ^ a b Fluorochemie. "Brief Introduction of PFPE Synthesis Methods". Fuzhou Topda New Material Co., Ltd. Retrieved June 27, 2022.
- ^ Kwiatkowski, Carol F.; Andrews, David Q.; Birnbaum, Linda S.; Bruton, Thomas A.; DeWitt, Jamie C.; Knappe, Detlef R. U.; Maffini, Maricel V.; Miller, Mark F.; Pelch, Katherine E.; Reade, Anna; Soehl, Anna; Trier, Xenia; Venier, Marta; Wagner, Charlotte C.; Wang, Zhanyun; Blum, Arlene (11 August 2020). "Scientific Basis for Managing PFAS as a Chemical Class". Environmental Science & Technology Letters. 7 (8): 532–543. doi: 10.1021/acs.estlett.0c00255. hdl: 20.500.11850/438999.
-
^
"Archived copy" (PDF). Archived from
the original (PDF) on 2021-02-27. Retrieved 2020-08-23.
{{ cite web}}: CS1 maint: archived copy as title ( link) - ^ a b c d e f g h i j k "HUSKEY Specialty lubricants" (PDF). HUSK-ITT Corporation.
- ^ Synthesis Report on Understanding Perfluoropolyethers (PFPEs) and Their Life Cycle (PDF). OECD. 2024.
- ^ a b c Ash, Michael (2004). Handbook of green chemicals. Irene Ash (2 ed.). Endicott, NY: Synapse Information Resources. ISBN 978-1-934764-29-9. OCLC 747426845.