| |
| Names | |
|---|---|
|
IUPAC name
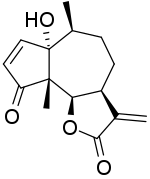
1-Hydroxy-6β,12-epoxyambrosa-2,11(13)-diene-4,12-dione
| |
|
Systematic IUPAC name
(3aS,6S,6aS,9aS,9bR)-6a-Hydroxy-6,9a-dimethyl-3-methylidene-3,3a,4,5,6,6a,9a,9b-octahydroazuleno[4,5-b]furan-2,9-dione | |
| Other names
Parthenicin
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C15H18O4 | |
| Molar mass | 262.305 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Parthenin is a chemical compound classified as a sesquiterpene lactone. It has been isolated from Parthenium hysterophorus.
It is genotoxic, allergenic, and an irritant. [1] Parthenin is believed to be responsible for the dermatitis caused by Parthenium hysterophorus. [2]
References
- ^ Ramos, Alberto; Rivero, Reinaldo; Visozo, Angel; Piloto, Janet; Garcı́a, Arilia (2002). "Parthenin, a sesquiterpene lactone of Parthenium hysterophorus L. Is a high toxicity clastogen". Mutation Research/Genetic Toxicology and Environmental Mutagenesis. 514 (1–2): 19–27. doi: 10.1016/S1383-5718(01)00321-7. PMID 11815241.
- ^ Picman, J.; Picman, A. K. (1985). "Treatment of dermatitis from parthenin". Contact Dermatitis. 13 (1): 9–13. doi: 10.1111/j.1600-0536.1985.tb02484.x. PMID 4042647. S2CID 40846706.

| |
| Names | |
|---|---|
|
IUPAC name
1-Hydroxy-6β,12-epoxyambrosa-2,11(13)-diene-4,12-dione
| |
|
Systematic IUPAC name
(3aS,6S,6aS,9aS,9bR)-6a-Hydroxy-6,9a-dimethyl-3-methylidene-3,3a,4,5,6,6a,9a,9b-octahydroazuleno[4,5-b]furan-2,9-dione | |
| Other names
Parthenicin
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C15H18O4 | |
| Molar mass | 262.305 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Parthenin is a chemical compound classified as a sesquiterpene lactone. It has been isolated from Parthenium hysterophorus.
It is genotoxic, allergenic, and an irritant. [1] Parthenin is believed to be responsible for the dermatitis caused by Parthenium hysterophorus. [2]
References
- ^ Ramos, Alberto; Rivero, Reinaldo; Visozo, Angel; Piloto, Janet; Garcı́a, Arilia (2002). "Parthenin, a sesquiterpene lactone of Parthenium hysterophorus L. Is a high toxicity clastogen". Mutation Research/Genetic Toxicology and Environmental Mutagenesis. 514 (1–2): 19–27. doi: 10.1016/S1383-5718(01)00321-7. PMID 11815241.
- ^ Picman, J.; Picman, A. K. (1985). "Treatment of dermatitis from parthenin". Contact Dermatitis. 13 (1): 9–13. doi: 10.1111/j.1600-0536.1985.tb02484.x. PMID 4042647. S2CID 40846706.