| PPIE | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | PPIE, CYP-33, CYP33, peptidylprolyl isomerase E | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 602435; MGI: 1917118; HomoloGene: 38142; GeneCards: PPIE; OMA: PPIE - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
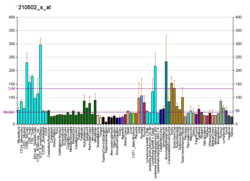
Peptidylprolyl isomerase E (cyclophilin E), also known as PPIE, is an enzyme which in humans is encoded by the PPIE gene on chromosome 1. As a member of the peptidyl-prolyl cis-trans isomerase (PPIase) family, this protein catalyzes the cis-trans isomerization of proline imidic peptide bonds, which allows it to facilitate folding or repair of proteins. [5] In addition, PPIE participates in many biological processes, including mitochondrial metabolism, apoptosis, and inflammation, as well as related diseases and conditions, such as ischemic reperfusion injury, AIDS, influenza, and cancer. [6] [7] [8]
Structure
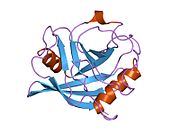
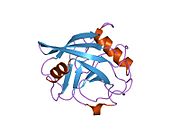
Like other cyclophilins, PPIE forms a β-barrel structure with a hydrophobic core. This β-barrel is composed of eight anti-parallel β-strands and capped by two α-helices at the top and bottom. In addition, the β-turns and loops in the strands contribute to the flexibility of the barrel. In particular, PPIE contains two RNA-binding domains at the N-terminal and a 165-bases long PPIase domain at the C-terminal. The PPIase domain is homologous to PPIA and can be bound and inhibited by CsA. [8]
Function
The protein encoded by this gene is a member of the peptidyl-prolyl cis-trans isomerase ( PPIase) family. PPIases catalyze the cis-trans isomerization of proline imidic peptide bonds in oligopeptides and accelerate the folding of proteins. [5] Generally, PPIases are found in all eubacteria and eukaryotes, as well as in a few archaebacteria, and thus are highly conserved. [6] [9] The PPIase family is further divided into three structurally distinct subfamilies: cyclophilin (CyP), FK506-binding protein ( FKBP), and parvulin (Pvn). [6] [8] As a cyclophilin, PPI binds cyclosporin A (CsA) and can be found within in the cell or secreted by the cell. [7] In eukaryotes, cyclophilins localize ubiquitously to many cell and tissue types. [7] [8] In addition to PPIase and protein chaperone activities, cyclophilins function in mitochondrial metabolism, apoptosis, immunological response, inflammation, and cell growth and proliferation. [6] [7] [8] PPIE in particular also exhibits RNA-binding activity. [5]
Clinical significance
Due to the close homology in the PPIase domain between PPIE and PPIA, PPIE may also be involved in the replication process of HIV. [8] Moreover, PPIE helps to prevent infections by influenza A virus. [10] As a cyclophilin, PPIE also binds the immunosuppressive drug CsA to form a CsA-cyclophilin complex, which then targets calcineurin to inhibit the signaling pathway for T-cell activation. [7]
In cardiac myogenic cells, cyclophilins have been observed to be activated by heat shock and hypoxia-reoxygenation as well as complex with heat shock proteins. Thus, cyclophilins may function in cardioprotection during ischemia-reperfusion injury.
Currently, cyclophilin expression is highly correlated with cancer pathogenesis, but the specific mechanisms remain to be elucidated. [7]
Interactions
PPIE (gene) has been shown to interact with CsA and MLL. [7] [11]
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000084072 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000028651 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ a b c "Entrez Gene: PPID peptidylprolyl isomerase E (cyclophilin E)".
- ^ a b c d Kazui T, Inoue N, Yamada O, Komatsu S (Jan 1992). "Selective cerebral perfusion during operation for aneurysms of the aortic arch: a reassessment". The Annals of Thoracic Surgery. 53 (1): 109–14. doi: 10.1016/0003-4975(92)90767-x. PMID 1530810.
- ^ a b c d e f g Yao Q, Li M, Yang H, Chai H, Fisher W, Chen C (Mar 2005). "Roles of cyclophilins in cancers and other organ systems". World Journal of Surgery. 29 (3): 276–80. doi: 10.1007/s00268-004-7812-7. PMID 15706440. S2CID 11678319.
- ^ a b c d e f Wang T, Yun CH, Gu SY, Chang WR, Liang DC (Aug 2005). "1.88 A crystal structure of the C domain of hCyP33: a novel domain of peptidyl-prolyl cis-trans isomerase". Biochemical and Biophysical Research Communications. 333 (3): 845–9. doi: 10.1016/j.bbrc.2005.06.006. PMID 15963461.
- ^ Hoffmann H, Schiene-Fischer C (Jul 2014). "Functional aspects of extracellular cyclophilins". Biological Chemistry. 395 (7–8): 721–35. doi: 10.1515/hsz-2014-0125. PMID 24713575. S2CID 32395688.
- ^ Wang Z, Liu X, Zhao Z, Xu C, Zhang K, Chen C, Sun L, Gao GF, Ye X, Liu W (2011). "Cyclophilin E functions as a negative regulator to influenza virus replication by impairing the formation of the viral ribonucleoprotein complex". PLOS ONE. 6 (8): e22625. Bibcode: 2011PLoSO...622625W. doi: 10.1371/journal.pone.0022625. PMC 3160840. PMID 21887220.
- ^ Fair K, Anderson M, Bulanova E, Mi H, Tropschug M, Diaz MO (May 2001). "Protein interactions of the MLL PHD fingers modulate MLL target gene regulation in human cells". Molecular and Cellular Biology. 21 (10): 3589–97. doi: 10.1128/MCB.21.10.3589-3597.2001. PMC 100280. PMID 11313484.
Further reading
- Mi H, Kops O, Zimmermann E, Jäschke A, Tropschug M (Dec 1996). "A nuclear RNA-binding cyclophilin in human T cells". FEBS Letters. 398 (2–3): 201–5. doi: 10.1016/S0014-5793(96)01248-3. PMID 8977107. S2CID 940022.
- Fair K, Anderson M, Bulanova E, Mi H, Tropschug M, Diaz MO (May 2001). "Protein interactions of the MLL PHD fingers modulate MLL target gene regulation in human cells". Molecular and Cellular Biology. 21 (10): 3589–97. doi: 10.1128/MCB.21.10.3589-3597.2001. PMC 100280. PMID 11313484.
- Jurica MS, Licklider LJ, Gygi SR, Grigorieff N, Moore MJ (Apr 2002). "Purification and characterization of native spliceosomes suitable for three-dimensional structural analysis". RNA. 8 (4): 426–39. doi: 10.1017/S1355838202021088. PMC 1370266. PMID 11991638.
- Rual JF, Venkatesan K, Hao T, Hirozane-Kishikawa T, Dricot A, Li N, Berriz GF, Gibbons FD, Dreze M, Ayivi-Guedehoussou N, Klitgord N, Simon C, Boxem M, Milstein S, Rosenberg J, Goldberg DS, Zhang LV, Wong SL, Franklin G, Li S, Albala JS, Lim J, Fraughton C, Llamosas E, Cevik S, Bex C, Lamesch P, Sikorski RS, Vandenhaute J, Zoghbi HY, Smolyar A, Bosak S, Sequerra R, Doucette-Stamm L, Cusick ME, Hill DE, Roth FP, Vidal M (Oct 2005). "Towards a proteome-scale map of the human protein-protein interaction network". Nature. 437 (7062): 1173–8. Bibcode: 2005Natur.437.1173R. doi: 10.1038/nature04209. PMID 16189514. S2CID 4427026.
- Davis TL, Walker JR, Campagna-Slater V, et al. (2010). "Structural and biochemical characterization of the human cyclophilin family of peptidyl-prolyl isomerases". PLOS Biol. 8 (7): e1000439. doi: 10.1371/journal.pbio.1000439. PMC 2911226. PMID 20676357.
| PPIE | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | PPIE, CYP-33, CYP33, peptidylprolyl isomerase E | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 602435; MGI: 1917118; HomoloGene: 38142; GeneCards: PPIE; OMA: PPIE - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Peptidylprolyl isomerase E (cyclophilin E), also known as PPIE, is an enzyme which in humans is encoded by the PPIE gene on chromosome 1. As a member of the peptidyl-prolyl cis-trans isomerase (PPIase) family, this protein catalyzes the cis-trans isomerization of proline imidic peptide bonds, which allows it to facilitate folding or repair of proteins. [5] In addition, PPIE participates in many biological processes, including mitochondrial metabolism, apoptosis, and inflammation, as well as related diseases and conditions, such as ischemic reperfusion injury, AIDS, influenza, and cancer. [6] [7] [8]
Structure
Like other cyclophilins, PPIE forms a β-barrel structure with a hydrophobic core. This β-barrel is composed of eight anti-parallel β-strands and capped by two α-helices at the top and bottom. In addition, the β-turns and loops in the strands contribute to the flexibility of the barrel. In particular, PPIE contains two RNA-binding domains at the N-terminal and a 165-bases long PPIase domain at the C-terminal. The PPIase domain is homologous to PPIA and can be bound and inhibited by CsA. [8]
Function
The protein encoded by this gene is a member of the peptidyl-prolyl cis-trans isomerase ( PPIase) family. PPIases catalyze the cis-trans isomerization of proline imidic peptide bonds in oligopeptides and accelerate the folding of proteins. [5] Generally, PPIases are found in all eubacteria and eukaryotes, as well as in a few archaebacteria, and thus are highly conserved. [6] [9] The PPIase family is further divided into three structurally distinct subfamilies: cyclophilin (CyP), FK506-binding protein ( FKBP), and parvulin (Pvn). [6] [8] As a cyclophilin, PPI binds cyclosporin A (CsA) and can be found within in the cell or secreted by the cell. [7] In eukaryotes, cyclophilins localize ubiquitously to many cell and tissue types. [7] [8] In addition to PPIase and protein chaperone activities, cyclophilins function in mitochondrial metabolism, apoptosis, immunological response, inflammation, and cell growth and proliferation. [6] [7] [8] PPIE in particular also exhibits RNA-binding activity. [5]
Clinical significance
Due to the close homology in the PPIase domain between PPIE and PPIA, PPIE may also be involved in the replication process of HIV. [8] Moreover, PPIE helps to prevent infections by influenza A virus. [10] As a cyclophilin, PPIE also binds the immunosuppressive drug CsA to form a CsA-cyclophilin complex, which then targets calcineurin to inhibit the signaling pathway for T-cell activation. [7]
In cardiac myogenic cells, cyclophilins have been observed to be activated by heat shock and hypoxia-reoxygenation as well as complex with heat shock proteins. Thus, cyclophilins may function in cardioprotection during ischemia-reperfusion injury.
Currently, cyclophilin expression is highly correlated with cancer pathogenesis, but the specific mechanisms remain to be elucidated. [7]
Interactions
PPIE (gene) has been shown to interact with CsA and MLL. [7] [11]
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000084072 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000028651 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ a b c "Entrez Gene: PPID peptidylprolyl isomerase E (cyclophilin E)".
- ^ a b c d Kazui T, Inoue N, Yamada O, Komatsu S (Jan 1992). "Selective cerebral perfusion during operation for aneurysms of the aortic arch: a reassessment". The Annals of Thoracic Surgery. 53 (1): 109–14. doi: 10.1016/0003-4975(92)90767-x. PMID 1530810.
- ^ a b c d e f g Yao Q, Li M, Yang H, Chai H, Fisher W, Chen C (Mar 2005). "Roles of cyclophilins in cancers and other organ systems". World Journal of Surgery. 29 (3): 276–80. doi: 10.1007/s00268-004-7812-7. PMID 15706440. S2CID 11678319.
- ^ a b c d e f Wang T, Yun CH, Gu SY, Chang WR, Liang DC (Aug 2005). "1.88 A crystal structure of the C domain of hCyP33: a novel domain of peptidyl-prolyl cis-trans isomerase". Biochemical and Biophysical Research Communications. 333 (3): 845–9. doi: 10.1016/j.bbrc.2005.06.006. PMID 15963461.
- ^ Hoffmann H, Schiene-Fischer C (Jul 2014). "Functional aspects of extracellular cyclophilins". Biological Chemistry. 395 (7–8): 721–35. doi: 10.1515/hsz-2014-0125. PMID 24713575. S2CID 32395688.
- ^ Wang Z, Liu X, Zhao Z, Xu C, Zhang K, Chen C, Sun L, Gao GF, Ye X, Liu W (2011). "Cyclophilin E functions as a negative regulator to influenza virus replication by impairing the formation of the viral ribonucleoprotein complex". PLOS ONE. 6 (8): e22625. Bibcode: 2011PLoSO...622625W. doi: 10.1371/journal.pone.0022625. PMC 3160840. PMID 21887220.
- ^ Fair K, Anderson M, Bulanova E, Mi H, Tropschug M, Diaz MO (May 2001). "Protein interactions of the MLL PHD fingers modulate MLL target gene regulation in human cells". Molecular and Cellular Biology. 21 (10): 3589–97. doi: 10.1128/MCB.21.10.3589-3597.2001. PMC 100280. PMID 11313484.
Further reading
- Mi H, Kops O, Zimmermann E, Jäschke A, Tropschug M (Dec 1996). "A nuclear RNA-binding cyclophilin in human T cells". FEBS Letters. 398 (2–3): 201–5. doi: 10.1016/S0014-5793(96)01248-3. PMID 8977107. S2CID 940022.
- Fair K, Anderson M, Bulanova E, Mi H, Tropschug M, Diaz MO (May 2001). "Protein interactions of the MLL PHD fingers modulate MLL target gene regulation in human cells". Molecular and Cellular Biology. 21 (10): 3589–97. doi: 10.1128/MCB.21.10.3589-3597.2001. PMC 100280. PMID 11313484.
- Jurica MS, Licklider LJ, Gygi SR, Grigorieff N, Moore MJ (Apr 2002). "Purification and characterization of native spliceosomes suitable for three-dimensional structural analysis". RNA. 8 (4): 426–39. doi: 10.1017/S1355838202021088. PMC 1370266. PMID 11991638.
- Rual JF, Venkatesan K, Hao T, Hirozane-Kishikawa T, Dricot A, Li N, Berriz GF, Gibbons FD, Dreze M, Ayivi-Guedehoussou N, Klitgord N, Simon C, Boxem M, Milstein S, Rosenberg J, Goldberg DS, Zhang LV, Wong SL, Franklin G, Li S, Albala JS, Lim J, Fraughton C, Llamosas E, Cevik S, Bex C, Lamesch P, Sikorski RS, Vandenhaute J, Zoghbi HY, Smolyar A, Bosak S, Sequerra R, Doucette-Stamm L, Cusick ME, Hill DE, Roth FP, Vidal M (Oct 2005). "Towards a proteome-scale map of the human protein-protein interaction network". Nature. 437 (7062): 1173–8. Bibcode: 2005Natur.437.1173R. doi: 10.1038/nature04209. PMID 16189514. S2CID 4427026.
- Davis TL, Walker JR, Campagna-Slater V, et al. (2010). "Structural and biochemical characterization of the human cyclophilin family of peptidyl-prolyl isomerases". PLOS Biol. 8 (7): e1000439. doi: 10.1371/journal.pbio.1000439. PMC 2911226. PMID 20676357.