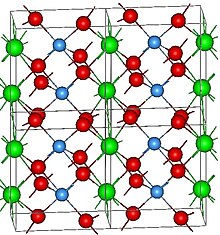
 The structure of Neodymium tanatalate. Atoms are: O(red), Ta(blue) and Nd(green)
| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
PubChem
CID
|
|
| |
| |
| Properties | |
| NdTaO4 | |
| Molar mass | 389.19 |
| Appearance | gray-purple solid [1] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Neodymium tantalate is an inorganic compound with the chemical formula NdTaO4. It is prepared by reacting neodymium oxide and tantalum pentoxide at 1200 °C. [1] It reacts with a mixture of tantalum pentoxide and chlorine gas at high temperature to obtain Nd2Ta2O7Cl2. [2] It is ammonolyzed at high temperature to obtain oxynitrides of Nd-Ta. [3]
This section may be too technical for most readers to understand. (May 2022) |
Neodymium tantalate forms violet crystals of the monoclinic system, with space group I 2/a, cell parameters a = 0.55153 nm, b = 1.12388 nm, c = 0.51184 nm, β = 95.731°, Z = 4. [4]
There is a metastable high-pressure phase of the monoclinic system, space group P 21/c, cell parameters a = 0.75920 nm, b = 0.54673 nm, c = 0.77022 nm, β = 100.032°, Z = 4. [5]
Neodymium tantalate is insoluble in water.[ citation needed]
- ^ a b Krylov, E. I.; Strelina, M. M. Praseodymium and neodymium orthotantalates. Zhurnal Neorganicheskoi Khimii, 1961. 6: 235-235. ISSN 0044-457X.
- ^ U. Schaffrath, R. Gruehn (1988-12-01). "Nd7,33Ta8O28CI6 — ein Zwischenprodukt des thermischen Abbaus von Nd2Ta2O7Cl2: Präparation, Struktur und magnetische Eigenschaften / Nd7,33Ta8O28Cl6 — an Intermediate Product of the Thermal Decomposition of Nd2Ta2O7Cl2: Preparation, Crystal Structure and Magnetic Properties". Zeitschrift für Naturforschung B. 43 (12): 1567–1574. doi: 10.1515/znb-1988-1208. ISSN 1865-7117. S2CID 98811409.
-
^ Pascal Maillard, Franck Tessier, Emmanuelle Orhan, François Cheviré, Roger Marchand (January 2005).
"Thermal Ammonolysis Study of the Rare-Earth Tantalates RTaO 4". Chemistry of Materials. 17 (1): 152–156.
doi:
10.1021/cm040131p.
ISSN
0897-4756.
S2CID
95148003. Retrieved 2021-06-06.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Kaijie Ning, Qingli Zhang, Deming Zhang, Jintai Fan, Dunlu Sun, Xiaofei Wang, Yin Hang (2014).
"Crystal growth, characterization of NdTaO4: A new promising stoichiometric neodymium laser material". Journal of Crystal Growth. 388 (Journal of Crystal Growth ed.): 83–86.
Bibcode:
2014JCrGr.388...83N.
doi:
10.1016/j.jcrysgro.2013.10.030.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Titov Yu. A., Sych A. M., Sokolov A. N., Kapshuk A. A., Markiv V. Ya., Belyavina N. M. (2000).
"Crystal structure of the high-pressure modification of NdTaO4". 311 (2) (Journal of alloys and compounds ed.): 252–255. Archived from
the original on 2014-08-12.
{{ cite journal}}: Cite journal requires|journal=( help)CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link)
- Patrick N. Walsh, Harold W. Goldstein, David White (May 1960).
"Vaporization of Rare-Earth Oxides". Journal of the American Ceramic Society. 43 (5): 229–233.
doi:
10.1111/j.1151-2916.1960.tb14589.x.
ISSN
0002-7820. Retrieved 2021-06-06.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link)
 The structure of Neodymium tanatalate. Atoms are: O(red), Ta(blue) and Nd(green)
| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
PubChem
CID
|
|
| |
| |
| Properties | |
| NdTaO4 | |
| Molar mass | 389.19 |
| Appearance | gray-purple solid [1] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Neodymium tantalate is an inorganic compound with the chemical formula NdTaO4. It is prepared by reacting neodymium oxide and tantalum pentoxide at 1200 °C. [1] It reacts with a mixture of tantalum pentoxide and chlorine gas at high temperature to obtain Nd2Ta2O7Cl2. [2] It is ammonolyzed at high temperature to obtain oxynitrides of Nd-Ta. [3]
This section may be too technical for most readers to understand. (May 2022) |
Neodymium tantalate forms violet crystals of the monoclinic system, with space group I 2/a, cell parameters a = 0.55153 nm, b = 1.12388 nm, c = 0.51184 nm, β = 95.731°, Z = 4. [4]
There is a metastable high-pressure phase of the monoclinic system, space group P 21/c, cell parameters a = 0.75920 nm, b = 0.54673 nm, c = 0.77022 nm, β = 100.032°, Z = 4. [5]
Neodymium tantalate is insoluble in water.[ citation needed]
- ^ a b Krylov, E. I.; Strelina, M. M. Praseodymium and neodymium orthotantalates. Zhurnal Neorganicheskoi Khimii, 1961. 6: 235-235. ISSN 0044-457X.
- ^ U. Schaffrath, R. Gruehn (1988-12-01). "Nd7,33Ta8O28CI6 — ein Zwischenprodukt des thermischen Abbaus von Nd2Ta2O7Cl2: Präparation, Struktur und magnetische Eigenschaften / Nd7,33Ta8O28Cl6 — an Intermediate Product of the Thermal Decomposition of Nd2Ta2O7Cl2: Preparation, Crystal Structure and Magnetic Properties". Zeitschrift für Naturforschung B. 43 (12): 1567–1574. doi: 10.1515/znb-1988-1208. ISSN 1865-7117. S2CID 98811409.
-
^ Pascal Maillard, Franck Tessier, Emmanuelle Orhan, François Cheviré, Roger Marchand (January 2005).
"Thermal Ammonolysis Study of the Rare-Earth Tantalates RTaO 4". Chemistry of Materials. 17 (1): 152–156.
doi:
10.1021/cm040131p.
ISSN
0897-4756.
S2CID
95148003. Retrieved 2021-06-06.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Kaijie Ning, Qingli Zhang, Deming Zhang, Jintai Fan, Dunlu Sun, Xiaofei Wang, Yin Hang (2014).
"Crystal growth, characterization of NdTaO4: A new promising stoichiometric neodymium laser material". Journal of Crystal Growth. 388 (Journal of Crystal Growth ed.): 83–86.
Bibcode:
2014JCrGr.388...83N.
doi:
10.1016/j.jcrysgro.2013.10.030.
{{ cite journal}}: CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link) -
^ Titov Yu. A., Sych A. M., Sokolov A. N., Kapshuk A. A., Markiv V. Ya., Belyavina N. M. (2000).
"Crystal structure of the high-pressure modification of NdTaO4". 311 (2) (Journal of alloys and compounds ed.): 252–255. Archived from
the original on 2014-08-12.
{{ cite journal}}: Cite journal requires|journal=( help)CS1 maint: date and year ( link) CS1 maint: multiple names: authors list ( link)
- Patrick N. Walsh, Harold W. Goldstein, David White (May 1960).
"Vaporization of Rare-Earth Oxides". Journal of the American Ceramic Society. 43 (5): 229–233.
doi:
10.1111/j.1151-2916.1960.tb14589.x.
ISSN
0002-7820. Retrieved 2021-06-06.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link)