| |
| Names | |
|---|---|
|
Preferred IUPAC name
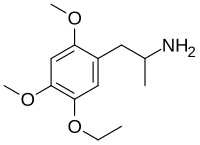
1-(5-Ethoxy-2,4-dimethoxyphenyl)propan-2-amine | |
| Other names
2,4-Dimethoxy-5-ethoxyamphetamine
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C13H21NO3 | |
| Molar mass | 239.315 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
MME (2,4-dimethoxy-5-ethoxyamphetamine) is a lesser-known psychedelic drug. It is a dimethoxy-ethoxy analog of TMA-2. MME was first synthesized by Alexander Shulgin from ethylvanillin. In his book PiHKAL, the minimum dosage is listed as 40 mg and above, and the duration listed as 6–10 hours. [1] Shulgin gives MME a ++ on the Shulgin Rating Scale.
Pharmacology
Shulgin describes in his book an experiment with MME, in which he admistered varying amounts of the drug to mice via injections. [1] Shulgin reports that 7 of the 9 mice injected with MME died as a result. [1] After describing his experiment, Shulgin speculates that MME may have an LD50 value of around 60–80 mg/Kg in mice when injected. [1] Shulgin describes that one of the mice began convulsing after being administered MME: "[...] the mouse went into a twitching series of convulsions (known as clonic in the trade) and in five minutes he was dead." The convulsions Shulgin noted may have been a sympom of serotonin syndrome in the mouse due to MME's affinity towards serotonin transporter in Mus musculus. [2]
Chemistry
MME has several isomers. MEM is one of them along with EMM. According to Alexander Shulgin EEM is not biologically active. [1] MEM is biologically active in humans and also possesses an affinity for the 5-HT2A receptor. [1] [3]
Synthesis
Shulgin describes the synthesis of MME in his book PiHKAL. Shulgin starts with 4-ethoxy-3-methoxybenzaldehyde. Shulgin labels the 4-ethoxy-3-methoxybenzaldehyde as ethylvanillin, although ethylvanillin is in fact 3-ethoxy-4-hydroxybenzaldehyde. Ethylvanillin can be methylate to 4-ethoxy-3-methoxybenzladehyde. The 4-ethoxy-3-methoxybenzaldehyde is then subjected to a Bayer-Villiger oxidation with peracetic acid and acetic acid to yield 4-ethoxy-3-methoxyphenol. The 4-ethoxy-3-methoxyphenol is methylated to yield 2,4-dimethoxy-1-ethoxybenzene. The 2,4-dimethoxy-1-ethoxybenzene is subjected to Reimer-Tiemann formylated to 2,4-dimethoxy-5-ethoxybenzaldehyde. The 2,4-dimethoxy-5-ethoxybenzaldehyde by subjecting it to a Knoevenagel condensation with acetic acid, ammonium acetate and nitroethane, and reducing the resulting 1-(2,4-dimethoxy-5-ethoxyphenyl)-2-nitropropene to MME with lithium aluminium hydride under an inert atmosphere. [1]
See also
References
- ^ a b c d e f g MEM entry in PiHKAL
- ^ Haberzettl R, Bert B, Fink H, Fox MA (November 2013). "Animal models of the serotonin syndrome: a systematic review". Behavioural Brain Research. 256: 328–345. doi: 10.1016/j.bbr.2013.08.045. PMID 24004848.
- ^ Ray TS (February 2010). "Psychedelics and the human receptorome". PLOS ONE. 5 (2): e9019. Bibcode: 2010PLoSO...5.9019R. doi: 10.1371/journal.pone.0009019. PMC 2814854. PMID 20126400.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
1-(5-Ethoxy-2,4-dimethoxyphenyl)propan-2-amine | |
| Other names
2,4-Dimethoxy-5-ethoxyamphetamine
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C13H21NO3 | |
| Molar mass | 239.315 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
MME (2,4-dimethoxy-5-ethoxyamphetamine) is a lesser-known psychedelic drug. It is a dimethoxy-ethoxy analog of TMA-2. MME was first synthesized by Alexander Shulgin from ethylvanillin. In his book PiHKAL, the minimum dosage is listed as 40 mg and above, and the duration listed as 6–10 hours. [1] Shulgin gives MME a ++ on the Shulgin Rating Scale.
Pharmacology
Shulgin describes in his book an experiment with MME, in which he admistered varying amounts of the drug to mice via injections. [1] Shulgin reports that 7 of the 9 mice injected with MME died as a result. [1] After describing his experiment, Shulgin speculates that MME may have an LD50 value of around 60–80 mg/Kg in mice when injected. [1] Shulgin describes that one of the mice began convulsing after being administered MME: "[...] the mouse went into a twitching series of convulsions (known as clonic in the trade) and in five minutes he was dead." The convulsions Shulgin noted may have been a sympom of serotonin syndrome in the mouse due to MME's affinity towards serotonin transporter in Mus musculus. [2]
Chemistry
MME has several isomers. MEM is one of them along with EMM. According to Alexander Shulgin EEM is not biologically active. [1] MEM is biologically active in humans and also possesses an affinity for the 5-HT2A receptor. [1] [3]
Synthesis
Shulgin describes the synthesis of MME in his book PiHKAL. Shulgin starts with 4-ethoxy-3-methoxybenzaldehyde. Shulgin labels the 4-ethoxy-3-methoxybenzaldehyde as ethylvanillin, although ethylvanillin is in fact 3-ethoxy-4-hydroxybenzaldehyde. Ethylvanillin can be methylate to 4-ethoxy-3-methoxybenzladehyde. The 4-ethoxy-3-methoxybenzaldehyde is then subjected to a Bayer-Villiger oxidation with peracetic acid and acetic acid to yield 4-ethoxy-3-methoxyphenol. The 4-ethoxy-3-methoxyphenol is methylated to yield 2,4-dimethoxy-1-ethoxybenzene. The 2,4-dimethoxy-1-ethoxybenzene is subjected to Reimer-Tiemann formylated to 2,4-dimethoxy-5-ethoxybenzaldehyde. The 2,4-dimethoxy-5-ethoxybenzaldehyde by subjecting it to a Knoevenagel condensation with acetic acid, ammonium acetate and nitroethane, and reducing the resulting 1-(2,4-dimethoxy-5-ethoxyphenyl)-2-nitropropene to MME with lithium aluminium hydride under an inert atmosphere. [1]
See also
References
- ^ a b c d e f g MEM entry in PiHKAL
- ^ Haberzettl R, Bert B, Fink H, Fox MA (November 2013). "Animal models of the serotonin syndrome: a systematic review". Behavioural Brain Research. 256: 328–345. doi: 10.1016/j.bbr.2013.08.045. PMID 24004848.
- ^ Ray TS (February 2010). "Psychedelics and the human receptorome". PLOS ONE. 5 (2): e9019. Bibcode: 2010PLoSO...5.9019R. doi: 10.1371/journal.pone.0009019. PMC 2814854. PMID 20126400.