
This article needs attention from an expert in Chemicals. The specific problem is: chemical names need verification and correction as necessary. (September 2016) |
This is a list of androgens/ anabolic steroids (AAS) or testosterone derivatives. Androgen esters are mostly not included in this list. The major classes of testosterone derivatives include the following (as well as combinations thereof):
- Testosterone derivatives: direct derivatives of testosterone not falling into the groups below
- 4,5α-Reduced/dihydrogenated testosterone derivatives: dihydrotestosterone (DHT) derivatives
- 19-Demethylated testosterone derivatives: 19-nortestosterone (nandrolone) derivatives
- 17α-Alkylated testosterone derivatives: methyltestosterone and ethyltestosterone derivatives
- 17α-Ethynylated/vinylated testosterone derivatives: ethynyltestosterone (ethisterone) and vinyltestosterone derivatives
The last group consists of progestins with mostly only very weak androgenic/anabolic activity.
This article pertains to steroidal androgens; nonsteroidal androgens like the selective androgen receptor modulators (SARMs) andarine and enobosarm (ostarine) are not included here.
Testosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
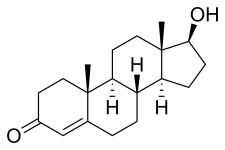
| Testosterone | Androst-4-en-17β-ol-3-one |
|
✓ |
| 4-Hydroxytestosterone | 4-Hydroxytestosterone |
|
– |
| 11-Ketotestosterone | 11-Ketotestosterone |
|
– |
| Boldenone | Δ1-Testosterone |
|
✓ |
| Clostebol | 4-Chlorotestosterone |
|
✓ |
Prohormone-like
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| 4-Androstenediol | 4-Androstenediol |
|
– |
| 4-Dehydroepiandrosterone (4-DHEA) | 4-Dehydroepiandrosterone |
|
– |
| 5-Androstenedione | 5-Androstenedione |
|
– |
| 5-Dehydroandrosterone (5-DHA) | 5-Dehydroandrosterone |
|
– |
| 11β-Hydroxyandrostenedione (11β-OHA4) | 11β-Hydroxy-4-androstenedione |
|
– |
| Adrenosterone (11-ketoandrostenedione, 11-KA4) | 11-Keto-4-androstenedione |
|
– |
| Androstenediol (5-androstenediol, A5) | 5-Androstenediol |
|
– |
| Androstenedione (4-androstenedione, A4) | 4-Androstenedione |
|
– |
| Atamestane | 1-Methyl-δ1-4-androstenedione |
|
– |
| Boldione (1,4-androstadienedione) | δ1-4-Androstenedione |
|
– |
| Dehydroepiandrosterone (DHEA, 5-DHEA; prasterone, androstenolone) | 5-Dehydroepiandrosterone |
|
✓ |
| Exemestane | 6-Methylidene-δ1-4-androstenedione |
|
✓ |
| Formestane | 4-Hydroxy-4-androstenedione |
|
✓ |
| Plomestane | 10-Propargyl-4-androstenedione |
|
– |
Prodrugs
Ethers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Cloxotestosterone | Testosterone 17-chloral hemiacetal ether |
|
✓ |
| Quinbolone | Δ1-Testosterone 17β-cyclopentenyl enol ether |
|
✓ |
| Silandrone | Testosterone 17β-trimethylsilyl ether |
|
– |
Dihydrotestosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Dihydrotestosterone (DHT); androstanolone, stanolone) | 4,5α-Dihydrotestosterone |
|
✓ |
| 1-Testosterone (dihydro-1-testosterone, dihydroboldenone) | 4,5α-Dihydro-δ1-testosterone |
|
– |
| 11-Ketodihydrotestosterone (11-KDHT) | 11-Keto-4,5α-dihydrotestosterone |
|
– |
| Drostanolone | 2α-Methyl-4,5α-dihydrotestosterone |
|
✓ |
| Epitiostanol (epithioandrostanol) | 2α,3α-Epithio-3-deketo-4,5α-dihydrotestosterone |
|
✓ |
| Mesterolone | 1α-Methyl-4,5α-dihydrotestosterone |
|
✓ |
| Metenolone (methenolone, methylandrostenolone) | 1-Methyl-4,5α-dihydro-δ1-testosterone |
|
✓ |
| Nisterime | 2α-Chloro-4,5α-dihydrotestosterone 3-O-(p-nitrophenyl)oxime |
|
– |
| Stenbolone | 2-Methyl-4,5α-dihydro-δ1-testosterone |
|
✓ |
Prohormone-like
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| 1-Androsterone (1-Andro, 1-DHEA) | 1-Dehydroepiandrosterone |
|
– |
| 1-Androstenediol (dihydro-1-androstenediol) | 1-Androstenediol (4,5α-dihydro-δ1-4-androstenediol) |
|
– |
| 1-Androstenedione (dihydro-1-androstenedione) | 1-Androstenedione (4,5α-dihydro-δ1-4-androstenedione) |
|
– |
| 5α-Androst-2-en-17-one | 3-Deketo-2-androstenedione (3-deketo-4,5α-dihydro-δ2-4-androstenedione) |
|
– |
| Androsterone | Androsterone |
|
– |
| Epiandrosterone | Epiandrosterone |
|
– |
Prodrugs
Ethers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Mepitiostane | 2α,3α-Epithio-3-deketo-4,5α-dihydrotestosterone 17β-(1-methoxycyclopentane) ether |
|
✓ |
| Mesabolone | 4,5α-Dihydro-δ1-testosterone 17β-(1-methoxycyclohexane) ether |
|
– |
| Prostanozol | 2'H-5α-Androst-2-eno[3,2-c]pyrazol-17β-ol 17β-tetrahydropyran ether |
|
– |
Azine dimers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Bolazine (di(drostanolone) azine) | 3,3-[(1E,2E)-1,2-Hydrazinediylidene]di(2α-methyl-5α-androstan-17β-ol) ? |
|
✓ |
19-Nortestosterone (nandrolone) derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Nandrolone (nortestosterone) | 19-Nortestosterone |
|
✓ |
| 11β-Methyl-19-nortestosterone (11β-MNT) | 11β-Methyl-19-nortestosterone |
|
– |
| Dienolone | 19-Nor-δ9-testosterone |
|
– |
| Dimethandrolone | 7α,11β-Dimethyl-19-nortestosterone |
|
– |
| Norclostebol | 4-Chloro-19-nortestosterone |
|
✓ |
| Oxabolone | 4-Hydroxy-19-nortestosterone |
|
✓ |
| Trenbolone (trienolone) | 19-Nor-δ9,11-testosterone |
|
✓ |
| Trestolone (MENT) | 7α-Methyl-19-nortestosterone |
|
– |
Prohormone-like
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| 7α-Methyl-19-nor-4-androstenedione (MENT dione, trestione) | 7α-Methyl-19-nor-4-androstenedione |
|
– |
| 19-Nor-5-androstenediol | 19-Nor-5-androstenediol |
|
– |
| 19-Nor-5-androstenedione | 19-Nor-5-androstenedione |
|
– |
| 19-Nordehydroepiandrosterone | 19-Nor-5-dehydroepiandrosterone |
|
– |
| Bolandiol (nor-4-androstenediol) | 19-Nor-4-androstenediol |
|
✓ |
| Bolandione (nor-4-androstenedione) | 19-Nor-4-androstenedione |
|
– |
| Dienedione (nor-4,9-androstadienedione) | 19-Nor-δ9-4-androstenedione |
|
– |
| Methoxydienone (methoxygonadiene) | 18-Methyl-19-nor-δ2,5(10)-epiandrosterone 3-methyl ether |
|
– |
| Trendione (nor-4,9,11-androstatrienedione) | 19-Nor-δ9,11-4-androstenedione |
|
– |
Prodrugs
Esters
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Bolmantalate (nandrolone adamantoate) | 19-Nortestosterone 17β-adamantoate |
|
✓ |
17α-Alkylated testosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Bolasterone | 7α,17α-Dimethyltestosterone |
|
✓ |
| Calusterone | 7β,17α-Dimethyltestosterone |
|
✓ |
| Chlorodehydromethyltestosterone (CDMT) | 4-Chloro-17α-methyl-δ1-testosterone |
|
✓ |
| Enestebol | 4-Hydroxy-17α-methyl-δ1-testosterone |
|
– |
| Ethyltestosterone | 17α-Ethyltestosterone |
|
– |
| Fluoxymesterone | 9α-Fluoro-11β-hydroxy-17α-methyltestosterone |
|
✓ |
| Formebolone | 2-Formyl-11α-hydroxy-17α-methyl-δ1-testosterone |
|
✓ |
| Hydroxystenozole | 17α-Methyl-2'H-androsta-2,4-dieno[3,2-c]pyrazol-17β-ol |
|
– |
| Metandienone (methandienone, methandrostenolone) | 17α-Methyl-δ1-testosterone |
|
✓ |
| Methylclostebol (chloromethyltestosterone) | 4-Chloro-17α-methyltestosterone |
|
– |
| Methyltestosterone | 17α-Methyltestosterone |
|
✓ |
| Oxymesterone | 4-Hydroxy-17α-methyltestosterone |
|
✓ |
| Tiomesterone (thiomesterone) | 1α,7α-Diacetylthio-17α-methyltestosterone |
|
✓ |
Prohormone-like
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Chlorodehydromethylandrostenediol (CDMA) | 4-Chloro-17α-methyl-δ1-4-androstenediol |
|
– |
| Chloromethylandrostenediol (CMA) | 4-Chloro-17α-methyl-4-androstenediol |
|
– |
| Methandriol (methylandrostenediol) | 17α-Methyl-5-androstenediol |
|
✓ |
Prodrugs
Ethers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Methyltestosterone 3-hexyl ether | 17α-Methyl-4-hydro-δ3,5-testosterone 3-hexyl ether ? |
|
✓ |
| Penmesterol (penmestrol) | 17α-Methyl-4-hydro-δ3,5-testosterone 3-cyclopentyl ether ? |
|
✓ |
17α-Alkylated dihydrotestosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Androisoxazole | 17α-Methyl-5α-androstano[3,2-c]isoxazol-17β-ol |
|
✓ |
| Desoxymethyltestosterone | 3-Deketo-17α-methyl-4,5α-dihydro-δ2-testosterone |
|
– |
| Furazabol | 17α-Methyl-5α-androstano[2,3-c][1,2,5]oxadiazol-17β-ol |
|
✓ |
| Mestanolone (methyl- DHT) | 17α-Methyl-4,5α-dihydrotestosterone |
|
✓ |
| Methasterone (methyldrostanolone) | 2α,17α-Dimethyl-4,5α-dihydrotestosterone |
|
– |
| Methyl-1-testosterone (methyldihydro-1-testosterone) | 17α-Methyl-4,5α-dihydro-δ1-testosterone |
|
– |
| Methyldiazinol | 3-Azi-17α-methyl-4,5α-dihydrotestosterone |
|
– |
| Methylepitiostanol | 2α,3α-Epithio-3-deketo-17α-methyl-4,5α-dihydrotestosterone |
|
– |
| Methylstenbolone | 2,17α-Dimethyl-4,5α-dihydro-δ1-testosterone |
|
– |
| Oxandrolone | 2-Oxa-17α-methyl-4,5α-dihydrotestosterone |
|
✓ |
| Oxymetholone | 2-Hydroxymethylene-4,5α-dihydro-17α-methyltestosterone |
|
✓ |
| Stanozolol | 17α-Methyl-2'H-5α-androst-2-eno[3,2-c]pyrazol-17β-ol |
|
✓ |
Prodrugs
Azine dimers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Mebolazine (dimethazine, di(methasterone) azine) | 3,3-[(1E,2E)-1,2-Hydrazinediylidene]di(2α,17α-dimethyl-5α-androstan-17β-ol) ? |
|
✓ |
17α-Alkylated 19-nortestosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Dimethyltrienolone (7α,17α-dimethyltrenbolone) | 7α,17α-Dimethyl-19-nor-δ9,11-testosterone |
|
– |
| Dimethyldienolone (7α,17α-dimethyldienolone) | 7α,17α-Dimethyl-19-nor-δ9-testosterone |
|
– |
| Ethyldienolone | 17α-Ethyl-19-nor-δ9-testosterone |
|
– |
| Ethylestrenol (ethylnandrol) | 17α-Ethyl-3-deketo-19-nortestosterone |
|
✓ |
| Methyldienolone | 17α-Methyl-19-nor-δ9-testosterone |
|
– |
| Methylhydroxynandrolone (MOHN, MHN) | 4-Hydroxy-17α-methyl-19-nortestosterone |
|
– |
| Metribolone (methyltrienolone, R-1881) | 17α-Methyl-19-nor-δ9,11-testosterone |
|
– |
| Mibolerone | 7α,17α-Dimethyl-19-nortestosterone |
|
✓ |
| Norboletone | 17α-Ethyl-18-methyl-19-nortestosterone |
|
– |
| Norethandrolone (ethylnandrolone, ethylestrenolone) | 17α-Ethyl-19-nortestosterone |
|
✓ |
| Normethandrone (methylestrenolone, normethisterone) | 17α-Methyl-19-nortestosterone |
|
✓ |
| RU-2309 (18-methymetribolone, 17α-methyl-THG) | 17α,18-Dimethyl-19-nor-δ9,11-testosterone |
|
– |
| Tetrahydrogestrinone (THG) | 17α-Ethyl-18-methyl-19-nor-δ9,11-testosterone |
|
– |
Prohormone-like
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Bolenol (ethylnorandrostenol) | 3-Deketo-17α-ethyl-19-nor-5-androstenediol |
|
– |
Prodrugs
Esters
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Propetandrol | 17α-Ethyl-19-nortestosterone 3-propionate |
|
✓ |
17α-Vinylated testosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Vinyltestosterone | 17α-Ethenyltestosterone |
|
– |
17α-Vinylated 19-nortestosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Norvinisterone (vinylnortestosterone) | 17α-Ethenyl-19-nortestosterone |
|
✓ |
Commentary
The 17α- ethenylated (vinylated) testosterone derivative norvinisterone (vinylnortestosterone) is much more potent as an AAS than the 17α- ethynylated testosterone derivatives and is intermediate in potency between the 17α-ethynylated progestins and conventional AAS, with approximately one-third and one-fifth of the respective androgenic and anabolic activity of nandrolone in animal bioassays. [1]
Vinyltestosterone has been described as a weak AAS, though stronger than its 17α-ethynylated analogue ethisterone. [2]
17α-Ethynylated testosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Ethisterone (ethinyltestosterone) | 17α-Ethynyltestosterone |
|
✓ |
| Danazol (2,3-isoxazolethisterone) | 2,3-Isoxazol-17α-ethynyltestosterone |
|
✓ |
17α-Ethynylated 19-nortestosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Norethisterone (norethindrone) | 17α-Ethynyl-19-nortestosterone |
|
✓ |
| Etynodiol (ethynodiol, 3β-hydroxynorethisterone) | 17α-Ethynyl-3-deketo-3β-hydroxy-19-nortestosterone |
|
– |
| Gestrinone (ethylnorgestrienone, R-2323) | 17α-Ethynyl-18-methyl-19-nor-δ9,11-testosterone |
|
✓ |
| Levonorgestrel ((−)-norgestrel) | (−)-17α-Ethynyl-18-methyl-19-nortestosterone |
|
✓ |
| Lynestrenol (3-deketonorethisterone) | 17α-Ethynyl-3-deketo-19-nortestosterone |
|
✓ |
| Norgestrel (18-methylnorethisterone) | 17α-Ethynyl-18-methyl-19-nortestosterone |
|
✓ |
| Norgestrienone (ethynyltrenbolone) | 17α-Ethynyl-19-nor-δ9,11-testosterone |
|
✓ |
| Tibolone (7α-methylnoretynodrel) | 7α-Methyl-17α-ethynyl-19-nor-δ5(10)-testosterone |
|
✓ |
Prodrugs
Ethers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Quingestanol | 4-Hydro-19-nor-δ3,5-testosterone 3-cyclopentyl ether ? |
|
– |
Esters
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Etynodiol diacetate (ethynodiol diacetate) | 17α-Ethynyl-3-deketo-3β-hydroxy-19-nortestosterone 3β,17β-diacetate |
|
✓ |
| Norethisterone acetate (norethindrone acetate) | 17α-Ethynyl-19-nortestosterone 17β-acetate |
|
✓ |
| Norethisterone enanthate (norethindrone enanthate) | 17α-Ethynyl-19-nortestosterone 17β-enanthate |
|
✓ |
Ethers and esters
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Quingestanol acetate | 4-Hydro-17α-ethynyl-19-nor-δ3,5-testosterone 3-cyclopentyl ether 17β-acetate ? |
|
✓ |
Commentary
17α-Ethynylated testosterone derivatives are potent progestins with only very weak androgenic/anabolic activity and are used as oral contraceptives or for the treatment of gynecological conditions in women. They are invariably classified as progestins rather than as AAS. However, these progestins are testosterone derivatives and do have significant androgenic/anabolic activity, sometimes producing acne and other mild androgenic effects in women. Conversely, in men, these drugs may actually have functional antiandrogen effects due to their potent progestogenic and hence antigonadotropic activity and capacity to suppress gonadal testosterone production. [3]
See also
- List of steroids
- List of designer drugs § Androgens
- List of androgens/anabolic steroids available in the United States
Notes
? = Chemical names that are unverified.
References
- ^ Saunders, Francis J.; Drill, Victor A. (1956). "The Myotrophic and Androgenic Effects of 17-Ethyl-19-Nortestosterone and Related Compounds". Endocrinology. 58 (5): 567–572. doi: 10.1210/endo-58-5-567. ISSN 0013-7227. PMID 13317831.
- ^ LEWIS RA, DeMAJO S, ROSEMBERG E (1949). "The effects of 17-vinyl testosterone upon the rat adrenal". Endocrinology. 45 (6): 564–70. doi: 10.1210/endo-45-6-564. PMID 15402199.
-
^ Paulsen, C. Alvin; Leach, Robert B.; Lanman, John; Goldston, Norman; Maddock, W. O.; Heller, Carl G. (1962). "Inherent Estrogenicity of Norethindrone and Norethynodrel: Comparison with Other Synthetic Progestins and Progesterone1". The Journal of Clinical Endocrinology & Metabolism. 22 (10): 1033–1039.
doi:
10.1210/jcem-22-10-1033.
ISSN
0021-972X.
PMID
13942007.
Androgenic effects were absent for each of the compounds in the doses administered as judged by: (a) marked decrease in libido and sexual potentia in each of 21 normal male subjects receiving norethynodrel, norethindrone and norethandrolone; (b) failure to increase libido and sexual potentia in each of four hypogonadotrophic eunuchoidal men receiving norethandrolone (each had previously responded to testosterone administration); (c) no virilization of 14 of 15 postmenopausal women receiving the three progestins (one who was taking norethandrolone at the dose level of 30 mg daily noted lowering in the pitch of her voice during the second month of therapy).
Further reading
- Kicman, A T (2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–521. doi: 10.1038/bjp.2008.165. ISSN 0007-1188. PMC 2439524. PMID 18500378.
- Fragkaki AG, Angelis YS, Koupparis M, Tsantili-Kakoulidou A, Kokotos G, Georgakopoulos C (2009). "Structural characteristics of anabolic androgenic steroids contributing to binding to the androgen receptor and to their anabolic and androgenic activities. Applied modifications in the steroidal structure". Steroids. 74 (2): 172–97. doi: 10.1016/j.steroids.2008.10.016. PMID 19028512. S2CID 41356223.
- McRobb L, Handelsman DJ, Kazlauskas R, Wilkinson S, McLeod MD, Heather AK (2008). "Structure-activity relationships of synthetic progestins in a yeast-based in vitro androgen bioassay". J. Steroid Biochem. Mol. Biol. 110 (1–2): 39–47. doi: 10.1016/j.jsbmb.2007.10.008. PMID 18395441. S2CID 5612000.


This article needs attention from an expert in Chemicals. The specific problem is: chemical names need verification and correction as necessary. (September 2016) |
This is a list of androgens/ anabolic steroids (AAS) or testosterone derivatives. Androgen esters are mostly not included in this list. The major classes of testosterone derivatives include the following (as well as combinations thereof):
- Testosterone derivatives: direct derivatives of testosterone not falling into the groups below
- 4,5α-Reduced/dihydrogenated testosterone derivatives: dihydrotestosterone (DHT) derivatives
- 19-Demethylated testosterone derivatives: 19-nortestosterone (nandrolone) derivatives
- 17α-Alkylated testosterone derivatives: methyltestosterone and ethyltestosterone derivatives
- 17α-Ethynylated/vinylated testosterone derivatives: ethynyltestosterone (ethisterone) and vinyltestosterone derivatives
The last group consists of progestins with mostly only very weak androgenic/anabolic activity.
This article pertains to steroidal androgens; nonsteroidal androgens like the selective androgen receptor modulators (SARMs) andarine and enobosarm (ostarine) are not included here.
Testosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Testosterone | Androst-4-en-17β-ol-3-one |
|
✓ |
| 4-Hydroxytestosterone | 4-Hydroxytestosterone |
|
– |
| 11-Ketotestosterone | 11-Ketotestosterone |
|
– |
| Boldenone | Δ1-Testosterone |
|
✓ |
| Clostebol | 4-Chlorotestosterone |
|
✓ |
Prohormone-like
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| 4-Androstenediol | 4-Androstenediol |
|
– |
| 4-Dehydroepiandrosterone (4-DHEA) | 4-Dehydroepiandrosterone |
|
– |
| 5-Androstenedione | 5-Androstenedione |
|
– |
| 5-Dehydroandrosterone (5-DHA) | 5-Dehydroandrosterone |
|
– |
| 11β-Hydroxyandrostenedione (11β-OHA4) | 11β-Hydroxy-4-androstenedione |
|
– |
| Adrenosterone (11-ketoandrostenedione, 11-KA4) | 11-Keto-4-androstenedione |
|
– |
| Androstenediol (5-androstenediol, A5) | 5-Androstenediol |
|
– |
| Androstenedione (4-androstenedione, A4) | 4-Androstenedione |
|
– |
| Atamestane | 1-Methyl-δ1-4-androstenedione |
|
– |
| Boldione (1,4-androstadienedione) | δ1-4-Androstenedione |
|
– |
| Dehydroepiandrosterone (DHEA, 5-DHEA; prasterone, androstenolone) | 5-Dehydroepiandrosterone |
|
✓ |
| Exemestane | 6-Methylidene-δ1-4-androstenedione |
|
✓ |
| Formestane | 4-Hydroxy-4-androstenedione |
|
✓ |
| Plomestane | 10-Propargyl-4-androstenedione |
|
– |
Prodrugs
Ethers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Cloxotestosterone | Testosterone 17-chloral hemiacetal ether |
|
✓ |
| Quinbolone | Δ1-Testosterone 17β-cyclopentenyl enol ether |
|
✓ |
| Silandrone | Testosterone 17β-trimethylsilyl ether |
|
– |
Dihydrotestosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Dihydrotestosterone (DHT); androstanolone, stanolone) | 4,5α-Dihydrotestosterone |
|
✓ |
| 1-Testosterone (dihydro-1-testosterone, dihydroboldenone) | 4,5α-Dihydro-δ1-testosterone |
|
– |
| 11-Ketodihydrotestosterone (11-KDHT) | 11-Keto-4,5α-dihydrotestosterone |
|
– |
| Drostanolone | 2α-Methyl-4,5α-dihydrotestosterone |
|
✓ |
| Epitiostanol (epithioandrostanol) | 2α,3α-Epithio-3-deketo-4,5α-dihydrotestosterone |
|
✓ |
| Mesterolone | 1α-Methyl-4,5α-dihydrotestosterone |
|
✓ |
| Metenolone (methenolone, methylandrostenolone) | 1-Methyl-4,5α-dihydro-δ1-testosterone |
|
✓ |
| Nisterime | 2α-Chloro-4,5α-dihydrotestosterone 3-O-(p-nitrophenyl)oxime |
|
– |
| Stenbolone | 2-Methyl-4,5α-dihydro-δ1-testosterone |
|
✓ |
Prohormone-like
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| 1-Androsterone (1-Andro, 1-DHEA) | 1-Dehydroepiandrosterone |
|
– |
| 1-Androstenediol (dihydro-1-androstenediol) | 1-Androstenediol (4,5α-dihydro-δ1-4-androstenediol) |
|
– |
| 1-Androstenedione (dihydro-1-androstenedione) | 1-Androstenedione (4,5α-dihydro-δ1-4-androstenedione) |
|
– |
| 5α-Androst-2-en-17-one | 3-Deketo-2-androstenedione (3-deketo-4,5α-dihydro-δ2-4-androstenedione) |
|
– |
| Androsterone | Androsterone |
|
– |
| Epiandrosterone | Epiandrosterone |
|
– |
Prodrugs
Ethers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Mepitiostane | 2α,3α-Epithio-3-deketo-4,5α-dihydrotestosterone 17β-(1-methoxycyclopentane) ether |
|
✓ |
| Mesabolone | 4,5α-Dihydro-δ1-testosterone 17β-(1-methoxycyclohexane) ether |
|
– |
| Prostanozol | 2'H-5α-Androst-2-eno[3,2-c]pyrazol-17β-ol 17β-tetrahydropyran ether |
|
– |
Azine dimers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Bolazine (di(drostanolone) azine) | 3,3-[(1E,2E)-1,2-Hydrazinediylidene]di(2α-methyl-5α-androstan-17β-ol) ? |
|
✓ |
19-Nortestosterone (nandrolone) derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Nandrolone (nortestosterone) | 19-Nortestosterone |
|
✓ |
| 11β-Methyl-19-nortestosterone (11β-MNT) | 11β-Methyl-19-nortestosterone |
|
– |
| Dienolone | 19-Nor-δ9-testosterone |
|
– |
| Dimethandrolone | 7α,11β-Dimethyl-19-nortestosterone |
|
– |
| Norclostebol | 4-Chloro-19-nortestosterone |
|
✓ |
| Oxabolone | 4-Hydroxy-19-nortestosterone |
|
✓ |
| Trenbolone (trienolone) | 19-Nor-δ9,11-testosterone |
|
✓ |
| Trestolone (MENT) | 7α-Methyl-19-nortestosterone |
|
– |
Prohormone-like
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| 7α-Methyl-19-nor-4-androstenedione (MENT dione, trestione) | 7α-Methyl-19-nor-4-androstenedione |
|
– |
| 19-Nor-5-androstenediol | 19-Nor-5-androstenediol |
|
– |
| 19-Nor-5-androstenedione | 19-Nor-5-androstenedione |
|
– |
| 19-Nordehydroepiandrosterone | 19-Nor-5-dehydroepiandrosterone |
|
– |
| Bolandiol (nor-4-androstenediol) | 19-Nor-4-androstenediol |
|
✓ |
| Bolandione (nor-4-androstenedione) | 19-Nor-4-androstenedione |
|
– |
| Dienedione (nor-4,9-androstadienedione) | 19-Nor-δ9-4-androstenedione |
|
– |
| Methoxydienone (methoxygonadiene) | 18-Methyl-19-nor-δ2,5(10)-epiandrosterone 3-methyl ether |
|
– |
| Trendione (nor-4,9,11-androstatrienedione) | 19-Nor-δ9,11-4-androstenedione |
|
– |
Prodrugs
Esters
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Bolmantalate (nandrolone adamantoate) | 19-Nortestosterone 17β-adamantoate |
|
✓ |
17α-Alkylated testosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Bolasterone | 7α,17α-Dimethyltestosterone |
|
✓ |
| Calusterone | 7β,17α-Dimethyltestosterone |
|
✓ |
| Chlorodehydromethyltestosterone (CDMT) | 4-Chloro-17α-methyl-δ1-testosterone |
|
✓ |
| Enestebol | 4-Hydroxy-17α-methyl-δ1-testosterone |
|
– |
| Ethyltestosterone | 17α-Ethyltestosterone |
|
– |
| Fluoxymesterone | 9α-Fluoro-11β-hydroxy-17α-methyltestosterone |
|
✓ |
| Formebolone | 2-Formyl-11α-hydroxy-17α-methyl-δ1-testosterone |
|
✓ |
| Hydroxystenozole | 17α-Methyl-2'H-androsta-2,4-dieno[3,2-c]pyrazol-17β-ol |
|
– |
| Metandienone (methandienone, methandrostenolone) | 17α-Methyl-δ1-testosterone |
|
✓ |
| Methylclostebol (chloromethyltestosterone) | 4-Chloro-17α-methyltestosterone |
|
– |
| Methyltestosterone | 17α-Methyltestosterone |
|
✓ |
| Oxymesterone | 4-Hydroxy-17α-methyltestosterone |
|
✓ |
| Tiomesterone (thiomesterone) | 1α,7α-Diacetylthio-17α-methyltestosterone |
|
✓ |
Prohormone-like
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Chlorodehydromethylandrostenediol (CDMA) | 4-Chloro-17α-methyl-δ1-4-androstenediol |
|
– |
| Chloromethylandrostenediol (CMA) | 4-Chloro-17α-methyl-4-androstenediol |
|
– |
| Methandriol (methylandrostenediol) | 17α-Methyl-5-androstenediol |
|
✓ |
Prodrugs
Ethers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Methyltestosterone 3-hexyl ether | 17α-Methyl-4-hydro-δ3,5-testosterone 3-hexyl ether ? |
|
✓ |
| Penmesterol (penmestrol) | 17α-Methyl-4-hydro-δ3,5-testosterone 3-cyclopentyl ether ? |
|
✓ |
17α-Alkylated dihydrotestosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Androisoxazole | 17α-Methyl-5α-androstano[3,2-c]isoxazol-17β-ol |
|
✓ |
| Desoxymethyltestosterone | 3-Deketo-17α-methyl-4,5α-dihydro-δ2-testosterone |
|
– |
| Furazabol | 17α-Methyl-5α-androstano[2,3-c][1,2,5]oxadiazol-17β-ol |
|
✓ |
| Mestanolone (methyl- DHT) | 17α-Methyl-4,5α-dihydrotestosterone |
|
✓ |
| Methasterone (methyldrostanolone) | 2α,17α-Dimethyl-4,5α-dihydrotestosterone |
|
– |
| Methyl-1-testosterone (methyldihydro-1-testosterone) | 17α-Methyl-4,5α-dihydro-δ1-testosterone |
|
– |
| Methyldiazinol | 3-Azi-17α-methyl-4,5α-dihydrotestosterone |
|
– |
| Methylepitiostanol | 2α,3α-Epithio-3-deketo-17α-methyl-4,5α-dihydrotestosterone |
|
– |
| Methylstenbolone | 2,17α-Dimethyl-4,5α-dihydro-δ1-testosterone |
|
– |
| Oxandrolone | 2-Oxa-17α-methyl-4,5α-dihydrotestosterone |
|
✓ |
| Oxymetholone | 2-Hydroxymethylene-4,5α-dihydro-17α-methyltestosterone |
|
✓ |
| Stanozolol | 17α-Methyl-2'H-5α-androst-2-eno[3,2-c]pyrazol-17β-ol |
|
✓ |
Prodrugs
Azine dimers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Mebolazine (dimethazine, di(methasterone) azine) | 3,3-[(1E,2E)-1,2-Hydrazinediylidene]di(2α,17α-dimethyl-5α-androstan-17β-ol) ? |
|
✓ |
17α-Alkylated 19-nortestosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Dimethyltrienolone (7α,17α-dimethyltrenbolone) | 7α,17α-Dimethyl-19-nor-δ9,11-testosterone |
|
– |
| Dimethyldienolone (7α,17α-dimethyldienolone) | 7α,17α-Dimethyl-19-nor-δ9-testosterone |
|
– |
| Ethyldienolone | 17α-Ethyl-19-nor-δ9-testosterone |
|
– |
| Ethylestrenol (ethylnandrol) | 17α-Ethyl-3-deketo-19-nortestosterone |
|
✓ |
| Methyldienolone | 17α-Methyl-19-nor-δ9-testosterone |
|
– |
| Methylhydroxynandrolone (MOHN, MHN) | 4-Hydroxy-17α-methyl-19-nortestosterone |
|
– |
| Metribolone (methyltrienolone, R-1881) | 17α-Methyl-19-nor-δ9,11-testosterone |
|
– |
| Mibolerone | 7α,17α-Dimethyl-19-nortestosterone |
|
✓ |
| Norboletone | 17α-Ethyl-18-methyl-19-nortestosterone |
|
– |
| Norethandrolone (ethylnandrolone, ethylestrenolone) | 17α-Ethyl-19-nortestosterone |
|
✓ |
| Normethandrone (methylestrenolone, normethisterone) | 17α-Methyl-19-nortestosterone |
|
✓ |
| RU-2309 (18-methymetribolone, 17α-methyl-THG) | 17α,18-Dimethyl-19-nor-δ9,11-testosterone |
|
– |
| Tetrahydrogestrinone (THG) | 17α-Ethyl-18-methyl-19-nor-δ9,11-testosterone |
|
– |
Prohormone-like
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Bolenol (ethylnorandrostenol) | 3-Deketo-17α-ethyl-19-nor-5-androstenediol |
|
– |
Prodrugs
Esters
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Propetandrol | 17α-Ethyl-19-nortestosterone 3-propionate |
|
✓ |
17α-Vinylated testosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Vinyltestosterone | 17α-Ethenyltestosterone |
|
– |
17α-Vinylated 19-nortestosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Norvinisterone (vinylnortestosterone) | 17α-Ethenyl-19-nortestosterone |
|
✓ |
Commentary
The 17α- ethenylated (vinylated) testosterone derivative norvinisterone (vinylnortestosterone) is much more potent as an AAS than the 17α- ethynylated testosterone derivatives and is intermediate in potency between the 17α-ethynylated progestins and conventional AAS, with approximately one-third and one-fifth of the respective androgenic and anabolic activity of nandrolone in animal bioassays. [1]
Vinyltestosterone has been described as a weak AAS, though stronger than its 17α-ethynylated analogue ethisterone. [2]
17α-Ethynylated testosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Ethisterone (ethinyltestosterone) | 17α-Ethynyltestosterone |
|
✓ |
| Danazol (2,3-isoxazolethisterone) | 2,3-Isoxazol-17α-ethynyltestosterone |
|
✓ |
17α-Ethynylated 19-nortestosterone derivatives
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Norethisterone (norethindrone) | 17α-Ethynyl-19-nortestosterone |
|
✓ |
| Etynodiol (ethynodiol, 3β-hydroxynorethisterone) | 17α-Ethynyl-3-deketo-3β-hydroxy-19-nortestosterone |
|
– |
| Gestrinone (ethylnorgestrienone, R-2323) | 17α-Ethynyl-18-methyl-19-nor-δ9,11-testosterone |
|
✓ |
| Levonorgestrel ((−)-norgestrel) | (−)-17α-Ethynyl-18-methyl-19-nortestosterone |
|
✓ |
| Lynestrenol (3-deketonorethisterone) | 17α-Ethynyl-3-deketo-19-nortestosterone |
|
✓ |
| Norgestrel (18-methylnorethisterone) | 17α-Ethynyl-18-methyl-19-nortestosterone |
|
✓ |
| Norgestrienone (ethynyltrenbolone) | 17α-Ethynyl-19-nor-δ9,11-testosterone |
|
✓ |
| Tibolone (7α-methylnoretynodrel) | 7α-Methyl-17α-ethynyl-19-nor-δ5(10)-testosterone |
|
✓ |
Prodrugs
Ethers
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Quingestanol | 4-Hydro-19-nor-δ3,5-testosterone 3-cyclopentyl ether ? |
|
– |
Esters
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Etynodiol diacetate (ethynodiol diacetate) | 17α-Ethynyl-3-deketo-3β-hydroxy-19-nortestosterone 3β,17β-diacetate |
|
✓ |
| Norethisterone acetate (norethindrone acetate) | 17α-Ethynyl-19-nortestosterone 17β-acetate |
|
✓ |
| Norethisterone enanthate (norethindrone enanthate) | 17α-Ethynyl-19-nortestosterone 17β-enanthate |
|
✓ |
Ethers and esters
| Compound | Chemical name | Structure | Marketed |
|---|---|---|---|
| Quingestanol acetate | 4-Hydro-17α-ethynyl-19-nor-δ3,5-testosterone 3-cyclopentyl ether 17β-acetate ? |
|
✓ |
Commentary
17α-Ethynylated testosterone derivatives are potent progestins with only very weak androgenic/anabolic activity and are used as oral contraceptives or for the treatment of gynecological conditions in women. They are invariably classified as progestins rather than as AAS. However, these progestins are testosterone derivatives and do have significant androgenic/anabolic activity, sometimes producing acne and other mild androgenic effects in women. Conversely, in men, these drugs may actually have functional antiandrogen effects due to their potent progestogenic and hence antigonadotropic activity and capacity to suppress gonadal testosterone production. [3]
See also
- List of steroids
- List of designer drugs § Androgens
- List of androgens/anabolic steroids available in the United States
Notes
? = Chemical names that are unverified.
References
- ^ Saunders, Francis J.; Drill, Victor A. (1956). "The Myotrophic and Androgenic Effects of 17-Ethyl-19-Nortestosterone and Related Compounds". Endocrinology. 58 (5): 567–572. doi: 10.1210/endo-58-5-567. ISSN 0013-7227. PMID 13317831.
- ^ LEWIS RA, DeMAJO S, ROSEMBERG E (1949). "The effects of 17-vinyl testosterone upon the rat adrenal". Endocrinology. 45 (6): 564–70. doi: 10.1210/endo-45-6-564. PMID 15402199.
-
^ Paulsen, C. Alvin; Leach, Robert B.; Lanman, John; Goldston, Norman; Maddock, W. O.; Heller, Carl G. (1962). "Inherent Estrogenicity of Norethindrone and Norethynodrel: Comparison with Other Synthetic Progestins and Progesterone1". The Journal of Clinical Endocrinology & Metabolism. 22 (10): 1033–1039.
doi:
10.1210/jcem-22-10-1033.
ISSN
0021-972X.
PMID
13942007.
Androgenic effects were absent for each of the compounds in the doses administered as judged by: (a) marked decrease in libido and sexual potentia in each of 21 normal male subjects receiving norethynodrel, norethindrone and norethandrolone; (b) failure to increase libido and sexual potentia in each of four hypogonadotrophic eunuchoidal men receiving norethandrolone (each had previously responded to testosterone administration); (c) no virilization of 14 of 15 postmenopausal women receiving the three progestins (one who was taking norethandrolone at the dose level of 30 mg daily noted lowering in the pitch of her voice during the second month of therapy).
Further reading
- Kicman, A T (2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–521. doi: 10.1038/bjp.2008.165. ISSN 0007-1188. PMC 2439524. PMID 18500378.
- Fragkaki AG, Angelis YS, Koupparis M, Tsantili-Kakoulidou A, Kokotos G, Georgakopoulos C (2009). "Structural characteristics of anabolic androgenic steroids contributing to binding to the androgen receptor and to their anabolic and androgenic activities. Applied modifications in the steroidal structure". Steroids. 74 (2): 172–97. doi: 10.1016/j.steroids.2008.10.016. PMID 19028512. S2CID 41356223.
- McRobb L, Handelsman DJ, Kazlauskas R, Wilkinson S, McLeod MD, Heather AK (2008). "Structure-activity relationships of synthetic progestins in a yeast-based in vitro androgen bioassay". J. Steroid Biochem. Mol. Biol. 110 (1–2): 39–47. doi: 10.1016/j.jsbmb.2007.10.008. PMID 18395441. S2CID 5612000.