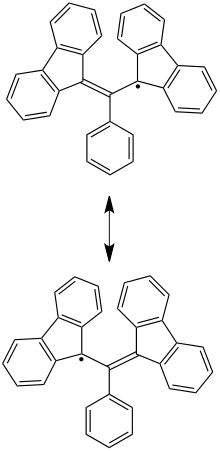
 Two resonance forms showing the predominant locations of the unpaired electron at the 1 and 3 positions
| |
| Names | |
|---|---|
|
Preferred IUPAC name
9-[(9H-Fluoren-9-ylidene)(phenyl)methyl]-9H-fluoren-9-yl | |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C33H21 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
The Koelsch radical (also known as Koelsch's radical and 1,3-bisdiphenylene-2-phenylallyl or α,γ-bisdiphenylene-β-phenylallyl, abbreviated BDPA) [1] [2] is a chemical compound that is an unusually stable carbon-centered radical, due to its resonance structures.
Properties
BDPA is an unusually stable radical compound due to the extent to which its electrons are delocalized through resonance structures. The unpaired electron is located predominantly at the 1 and 3 positions. [3] Steric effects from the biphenyl substituents also contribute to the compound's stability. [4]
Uses
BDPA and closely related compounds are used as molecular standards in electron paramagnetic resonance (EPR) and electron nuclear double resonance (ENDOR) experiments, [5] [6] and as a polarizing agent in dynamic nuclear polarization (DNP) nuclear magnetic resonance (NMR) experiments. [7] [8] Because BDPA itself is hydrophobic, derivatives have been developed that are more soluble in aqueous solution. [9]
History
The compound was first synthesized by C. Frederick Koelsch while he was a postdoctoral fellow at Harvard University in the 1930s. He attempted to publish a paper describing the compound, but the paper was rejected on the grounds that the described properties, particularly stability, were unlikely to be those of a radical. Subsequent experimental evidence and quantum mechanics calculations suggested his interpretation of the original experiment was correct, resulting in the publication of the paper in 1957, nearly 25 years after the original experiments. [1] [10] [11] Although the original report described stability on the order of years, modern experiments suggest that this family of compounds, while unusually stable for radicals, shows measurable degradation in months after preparation. [7]
References
- ^ a b Zard, Samir Z. (2003). Radical reactions in organic synthesis. Oxford: Oxford University Press. p. 3. ISBN 9780198502401.
- ^ Matsui, Yasunori; Shigemori, Minoru; Endo, Toshiyuki; Ogaki, Takuya; Ohta, Eisuke; Mizuno, Kazuhiko; Naito, Hiroyoshi; Ikeda, Hiroshi (August 2018). "Spectroscopic and electrical characterization of α,γ-bisdiphenylene-β-phenylallyl radical as an organic semiconductor". Research on Chemical Intermediates. 44 (8): 4765–4774. doi: 10.1007/s11164-018-3282-7.
- ^ Azuma, Nagao; Ozawa, Takehiro; Yamauchi, Jun (January 1994). "Molecular and Crystal Structures of Complexes of Stable Free Radical BDPA with Benzene and Acetone". Bulletin of the Chemical Society of Japan. 67 (1): 31–38. doi: 10.1246/bcsj.67.31.
- ^ Breslin, David T.; Fox, Marye Anne (December 1993). "Characterization of the excited-state reactivity of a persistent aryl-substituted allyl free radical". The Journal of Physical Chemistry. 97 (50): 13341–13347. doi: 10.1021/j100152a045.
- ^ Bennati, M.; Farrar, C.T.; Bryant, J.A.; Inati, S.J.; Weis, V.; Gerfen, G.J.; Riggs-Gelasco, P.; Stubbe, J.; Griffin, R.G. (June 1999). "Pulsed Electron-Nuclear Double Resonance (ENDOR) at 140 GHz". Journal of Magnetic Resonance. 138 (2): 232–243. doi: 10.1006/jmre.1999.1727.
- ^ Goldfarb, Daniella; Lipkin, Yaakov; Potapov, Alexey; Gorodetsky, Yehoshua; Epel, Boris; Raitsimring, Arnold M.; Radoul, Marina; Kaminker, Ilia (September 2008). "HYSCORE and DEER with an upgraded 95GHz pulse EPR spectrometer". Journal of Magnetic Resonance. 194 (1): 8–15. doi: 10.1016/j.jmr.2008.05.019.
- ^ a b Mandal, Sucharita; Sigurdsson, Snorri Th. (10 June 2020). "On the Limited Stability of BDPA Radicals". Chemistry – A European Journal. 26 (33): 7486–7491. doi: 10.1002/chem.202001084.
- ^ Lumata, Lloyd; Ratnakar, S. James; Jindal, Ashish; Merritt, Matthew; Comment, Arnaud; Malloy, Craig; Sherry, A. Dean; Kovacs, Zoltan (19 September 2011). "BDPA: An Efficient Polarizing Agent for Fast Dissolution Dynamic Nuclear Polarization NMR Spectroscopy". Chemistry - A European Journal. 17 (39): 10825–10827. doi: 10.1002/chem.201102037. PMC 3366484.
- ^ Dane, Eric L.; Swager, Timothy M. (21 May 2010). "Synthesis of a Water-Soluble 1,3-Bis(diphenylene)-2-phenylallyl Radical". The Journal of Organic Chemistry. 75 (10): 3533–3536. doi: 10.1021/jo100577g. PMC 2882179.
- ^ Koelsch, C. F. (August 1957). "Syntheses with Triarylvinylmagnesium Bromides. α,γ-Bisdiphenylene-β-phenylallyl, a Stable Free Radical". Journal of the American Chemical Society. 79 (16): 4439–4441. doi: 10.1021/ja01573a053.
- ^ Noland, Wayland E. (November 1975). "The Life and Times of Professor Emeritus C. Frederick Koelsch". The Minnesota Chemist. 27 (7): 7–17. Retrieved 29 December 2020.
 Two resonance forms showing the predominant locations of the unpaired electron at the 1 and 3 positions
| |
| Names | |
|---|---|
|
Preferred IUPAC name
9-[(9H-Fluoren-9-ylidene)(phenyl)methyl]-9H-fluoren-9-yl | |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C33H21 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
The Koelsch radical (also known as Koelsch's radical and 1,3-bisdiphenylene-2-phenylallyl or α,γ-bisdiphenylene-β-phenylallyl, abbreviated BDPA) [1] [2] is a chemical compound that is an unusually stable carbon-centered radical, due to its resonance structures.
Properties
BDPA is an unusually stable radical compound due to the extent to which its electrons are delocalized through resonance structures. The unpaired electron is located predominantly at the 1 and 3 positions. [3] Steric effects from the biphenyl substituents also contribute to the compound's stability. [4]
Uses
BDPA and closely related compounds are used as molecular standards in electron paramagnetic resonance (EPR) and electron nuclear double resonance (ENDOR) experiments, [5] [6] and as a polarizing agent in dynamic nuclear polarization (DNP) nuclear magnetic resonance (NMR) experiments. [7] [8] Because BDPA itself is hydrophobic, derivatives have been developed that are more soluble in aqueous solution. [9]
History
The compound was first synthesized by C. Frederick Koelsch while he was a postdoctoral fellow at Harvard University in the 1930s. He attempted to publish a paper describing the compound, but the paper was rejected on the grounds that the described properties, particularly stability, were unlikely to be those of a radical. Subsequent experimental evidence and quantum mechanics calculations suggested his interpretation of the original experiment was correct, resulting in the publication of the paper in 1957, nearly 25 years after the original experiments. [1] [10] [11] Although the original report described stability on the order of years, modern experiments suggest that this family of compounds, while unusually stable for radicals, shows measurable degradation in months after preparation. [7]
References
- ^ a b Zard, Samir Z. (2003). Radical reactions in organic synthesis. Oxford: Oxford University Press. p. 3. ISBN 9780198502401.
- ^ Matsui, Yasunori; Shigemori, Minoru; Endo, Toshiyuki; Ogaki, Takuya; Ohta, Eisuke; Mizuno, Kazuhiko; Naito, Hiroyoshi; Ikeda, Hiroshi (August 2018). "Spectroscopic and electrical characterization of α,γ-bisdiphenylene-β-phenylallyl radical as an organic semiconductor". Research on Chemical Intermediates. 44 (8): 4765–4774. doi: 10.1007/s11164-018-3282-7.
- ^ Azuma, Nagao; Ozawa, Takehiro; Yamauchi, Jun (January 1994). "Molecular and Crystal Structures of Complexes of Stable Free Radical BDPA with Benzene and Acetone". Bulletin of the Chemical Society of Japan. 67 (1): 31–38. doi: 10.1246/bcsj.67.31.
- ^ Breslin, David T.; Fox, Marye Anne (December 1993). "Characterization of the excited-state reactivity of a persistent aryl-substituted allyl free radical". The Journal of Physical Chemistry. 97 (50): 13341–13347. doi: 10.1021/j100152a045.
- ^ Bennati, M.; Farrar, C.T.; Bryant, J.A.; Inati, S.J.; Weis, V.; Gerfen, G.J.; Riggs-Gelasco, P.; Stubbe, J.; Griffin, R.G. (June 1999). "Pulsed Electron-Nuclear Double Resonance (ENDOR) at 140 GHz". Journal of Magnetic Resonance. 138 (2): 232–243. doi: 10.1006/jmre.1999.1727.
- ^ Goldfarb, Daniella; Lipkin, Yaakov; Potapov, Alexey; Gorodetsky, Yehoshua; Epel, Boris; Raitsimring, Arnold M.; Radoul, Marina; Kaminker, Ilia (September 2008). "HYSCORE and DEER with an upgraded 95GHz pulse EPR spectrometer". Journal of Magnetic Resonance. 194 (1): 8–15. doi: 10.1016/j.jmr.2008.05.019.
- ^ a b Mandal, Sucharita; Sigurdsson, Snorri Th. (10 June 2020). "On the Limited Stability of BDPA Radicals". Chemistry – A European Journal. 26 (33): 7486–7491. doi: 10.1002/chem.202001084.
- ^ Lumata, Lloyd; Ratnakar, S. James; Jindal, Ashish; Merritt, Matthew; Comment, Arnaud; Malloy, Craig; Sherry, A. Dean; Kovacs, Zoltan (19 September 2011). "BDPA: An Efficient Polarizing Agent for Fast Dissolution Dynamic Nuclear Polarization NMR Spectroscopy". Chemistry - A European Journal. 17 (39): 10825–10827. doi: 10.1002/chem.201102037. PMC 3366484.
- ^ Dane, Eric L.; Swager, Timothy M. (21 May 2010). "Synthesis of a Water-Soluble 1,3-Bis(diphenylene)-2-phenylallyl Radical". The Journal of Organic Chemistry. 75 (10): 3533–3536. doi: 10.1021/jo100577g. PMC 2882179.
- ^ Koelsch, C. F. (August 1957). "Syntheses with Triarylvinylmagnesium Bromides. α,γ-Bisdiphenylene-β-phenylallyl, a Stable Free Radical". Journal of the American Chemical Society. 79 (16): 4439–4441. doi: 10.1021/ja01573a053.
- ^ Noland, Wayland E. (November 1975). "The Life and Times of Professor Emeritus C. Frederick Koelsch". The Minnesota Chemist. 27 (7): 7–17. Retrieved 29 December 2020.