| |
| Names | |
|---|---|
|
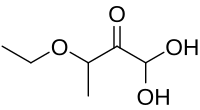
IUPAC name
3-Ethoxy-1,1-dihydroxy-2-butanone
| |
| Other names
3-Ethoxy-2-oxobutyraldehyde hydrate
| |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.215.334 |
| KEGG | |
| MeSH | C005135 |
PubChem
CID
|
|
| UNII |
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H12O4 | |
| Molar mass | 148.158 g·mol−1 |
| Appearance | Pale yellow syrup [1] |
| Boiling point | 145 °C (293 °F; 418 K) [1] [2] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Kethoxal (3-ethoxy-1,1-dihydroxy-2-butanone) is an organic compound that has antiviral and anaplasmosis properties. [1] [3] It also forms a stable covalent adduct with guanine, which makes it useful for nucleic acid structure determination. [4]
Nucleic acid binding
Kethoxal, as with other 1,2-di carbonyl compounds, [5] reacts with nucleic acids. It has high specificity for guanine over other ribonucleotides. In whole RNA, it reacts preferentially with guanine residues that are not involved in hydrogen-bonding. [6] [7] It can thus be used to probe the interactions involved with the secondary structure and other binding interactions of RNA [8] and help with nucleic acid sequence analysis. The binding is reversible, which allows the kethoxal to be removed and the original RNA recovered.[ citation needed]
References
- ^ a b c J. Elks (2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. p. 510. ISBN 9781475720853.
- ^ SciFinder record for CASRN 27762-78-3
- ^ "Kethoxal". Merriam-Webster.
- ^ Robert Shapiro; Bertram I. Cohen; Shian-Jan Shiuey; Hans Maurer (1969). "Reaction of guanine with glyoxal, pyruvaldehyde, and kethoxal, and the structure of the acylguanines. Synthesis of N2-alkylguanines". Biochemistry. 8 (1): 238–245. doi: 10.1021/bi00829a034. PMID 5777326.
- ^ Shapiro, Robert; Hachmann, John (1966). "The Reaction of Guanine Derivatives with 1,2-Dicarbonyl Compounds". Biochemistry. 5 (9): 2799–2807. doi: 10.1021/bi00873a004. PMID 5961865.
- ^ Litt, Michael; Hancock, Virginia (1967). "Kethoxal—A Potentially Useful Reagent for the Determination of Nucleotide Sequences in Single-Stranded Regions of Transfer Ribonucleic Acid". Biochemistry. 6 (6): 1848–1854. doi: 10.1021/bi00858a036. PMID 6035923.
- ^ Staehelin, Matthys (1959). "Inactivation of virus nucleic acid with glyoxal derivatives". Biochim. Biophys. Acta. 31 (2): 448–454. doi: 10.1016/0006-3002(59)90019-8. PMID 13628672.
- ^ Gopinath, S.C.B. (2009). "Mapping of RNA-protein interactions". Analytica Chimica Acta. 636 (2): 117–128. doi: 10.1016/j.aca.2009.01.052. PMID 19264161.

| |
| Names | |
|---|---|
|
IUPAC name
3-Ethoxy-1,1-dihydroxy-2-butanone
| |
| Other names
3-Ethoxy-2-oxobutyraldehyde hydrate
| |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.215.334 |
| KEGG | |
| MeSH | C005135 |
PubChem
CID
|
|
| UNII |
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H12O4 | |
| Molar mass | 148.158 g·mol−1 |
| Appearance | Pale yellow syrup [1] |
| Boiling point | 145 °C (293 °F; 418 K) [1] [2] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Kethoxal (3-ethoxy-1,1-dihydroxy-2-butanone) is an organic compound that has antiviral and anaplasmosis properties. [1] [3] It also forms a stable covalent adduct with guanine, which makes it useful for nucleic acid structure determination. [4]
Nucleic acid binding
Kethoxal, as with other 1,2-di carbonyl compounds, [5] reacts with nucleic acids. It has high specificity for guanine over other ribonucleotides. In whole RNA, it reacts preferentially with guanine residues that are not involved in hydrogen-bonding. [6] [7] It can thus be used to probe the interactions involved with the secondary structure and other binding interactions of RNA [8] and help with nucleic acid sequence analysis. The binding is reversible, which allows the kethoxal to be removed and the original RNA recovered.[ citation needed]
References
- ^ a b c J. Elks (2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. p. 510. ISBN 9781475720853.
- ^ SciFinder record for CASRN 27762-78-3
- ^ "Kethoxal". Merriam-Webster.
- ^ Robert Shapiro; Bertram I. Cohen; Shian-Jan Shiuey; Hans Maurer (1969). "Reaction of guanine with glyoxal, pyruvaldehyde, and kethoxal, and the structure of the acylguanines. Synthesis of N2-alkylguanines". Biochemistry. 8 (1): 238–245. doi: 10.1021/bi00829a034. PMID 5777326.
- ^ Shapiro, Robert; Hachmann, John (1966). "The Reaction of Guanine Derivatives with 1,2-Dicarbonyl Compounds". Biochemistry. 5 (9): 2799–2807. doi: 10.1021/bi00873a004. PMID 5961865.
- ^ Litt, Michael; Hancock, Virginia (1967). "Kethoxal—A Potentially Useful Reagent for the Determination of Nucleotide Sequences in Single-Stranded Regions of Transfer Ribonucleic Acid". Biochemistry. 6 (6): 1848–1854. doi: 10.1021/bi00858a036. PMID 6035923.
- ^ Staehelin, Matthys (1959). "Inactivation of virus nucleic acid with glyoxal derivatives". Biochim. Biophys. Acta. 31 (2): 448–454. doi: 10.1016/0006-3002(59)90019-8. PMID 13628672.
- ^ Gopinath, S.C.B. (2009). "Mapping of RNA-protein interactions". Analytica Chimica Acta. 636 (2): 117–128. doi: 10.1016/j.aca.2009.01.052. PMID 19264161.