| |
| Names | |
|---|---|
|
Preferred IUPAC name
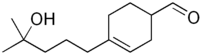
4-(4-Hydroxy-4-methylpentyl)cyclohex-3-ene-1-carbaldehyde | |
| Other names
Lyral, Kovanol, Mugonal, Landolal
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.046.225 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C13H22O2 | |
| Molar mass | 210.317 g·mol−1 |
| Density | 0.995 g/mL at 20 °C |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Hydroxymethylpentylcyclohexenecarboxaldehyde is a synthetic fragrance known by the trade names Lyral, Kovanol, Mugonal, Landolal. It is found in some soaps, eau de toilettes, aftershaves and deodorants.
Synthesis
Typical synthesis starts from myrcene [2] and involves a Diels–Alder reaction with acrolein to produce the cyclohexenecarbaldehyde group, this species is marketed as a fragrance in its own right, most commonly under the name 'myrac aldehyde'. Acid-catalyzed hydration of this completes the synthesis by forming the tertiary alcohol.
Safety
Lyral is known to act as a skin allergen and is listed as such in EU Directive 76/768/EEC. [3] It is commonly tested for in patients undergoing patch testing. [4]
References
- ^ 4-(4-Hydroxy-4-methylpentyl)-3-cyclohexene-1-carboxaldehyde at Sigma-Aldrich
- ^ Fahlbusch, K.-G.; Hammerschmidt, F.-J.; Panten, J.; Pickenhagen, W.; Schatkowski, D.; Bauer, K.; Garbe, D.; Surburg, H. (2002). "Flavors and Fragrances". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a11_141. ISBN 3527306730.
- ^ Allergens in Cosmetics & Personal Care Products
- ^ Bruze, Magnus; Andersen, Klaus Ejner; Goossens, An (March 2008). "Recommendation to include fragrance mix 2 and hydroxyisohexyl 3-cyclohexene carboxaldehyde (Lyral) in the European baseline patch test series". Contact Dermatitis. 58 (3): 129–133. doi: 10.1111/j.1600-0536.2007.01292.x. PMID 18279149. S2CID 205813259.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
4-(4-Hydroxy-4-methylpentyl)cyclohex-3-ene-1-carbaldehyde | |
| Other names
Lyral, Kovanol, Mugonal, Landolal
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.046.225 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C13H22O2 | |
| Molar mass | 210.317 g·mol−1 |
| Density | 0.995 g/mL at 20 °C |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Hydroxymethylpentylcyclohexenecarboxaldehyde is a synthetic fragrance known by the trade names Lyral, Kovanol, Mugonal, Landolal. It is found in some soaps, eau de toilettes, aftershaves and deodorants.
Synthesis
Typical synthesis starts from myrcene [2] and involves a Diels–Alder reaction with acrolein to produce the cyclohexenecarbaldehyde group, this species is marketed as a fragrance in its own right, most commonly under the name 'myrac aldehyde'. Acid-catalyzed hydration of this completes the synthesis by forming the tertiary alcohol.
Safety
Lyral is known to act as a skin allergen and is listed as such in EU Directive 76/768/EEC. [3] It is commonly tested for in patients undergoing patch testing. [4]
References
- ^ 4-(4-Hydroxy-4-methylpentyl)-3-cyclohexene-1-carboxaldehyde at Sigma-Aldrich
- ^ Fahlbusch, K.-G.; Hammerschmidt, F.-J.; Panten, J.; Pickenhagen, W.; Schatkowski, D.; Bauer, K.; Garbe, D.; Surburg, H. (2002). "Flavors and Fragrances". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a11_141. ISBN 3527306730.
- ^ Allergens in Cosmetics & Personal Care Products
- ^ Bruze, Magnus; Andersen, Klaus Ejner; Goossens, An (March 2008). "Recommendation to include fragrance mix 2 and hydroxyisohexyl 3-cyclohexene carboxaldehyde (Lyral) in the European baseline patch test series". Contact Dermatitis. 58 (3): 129–133. doi: 10.1111/j.1600-0536.2007.01292.x. PMID 18279149. S2CID 205813259.