 Liquid hydrogen telluride in a test tube
| |
| |
 Tellurium, Te Hydrogen, H | |
| Names | |
|---|---|
|
IUPAC name
hydrogen telluride
| |
| Other names
hydrotelluric acid
tellane tellurium hydride dihydrogen telluride tellurane | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.029.073 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| H2Te | |
| Molar mass | 129.6158 g mol−1 |
| Appearance | colourless gas |
| Odor | Pungent, resembles rotting garlic or leeks |
| Density | 3.310 g/L, gas 2.57 g/cm3 (−20 °C, liquid) |
| Melting point | −49 °C (−56 °F; 224 K) [1] |
| Boiling point | −2.2 °C (28.0 °F; 270.9 K) (unstable above −2 °C) |
| 0.70 g/100 mL | |
| Acidity (pKa) | 2.6 |
| Conjugate acid | Telluronium |
| Conjugate base | Telluride |
| Structure | |
| bent | |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
0.7684 kJ/g |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
toxic |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other
anions
|
H2O H2S H2Se H2Po |
Other
cations
|
Na2Te Ag2Te K2Te Rb2Te Cs2Te |
Related compounds
|
telluric acid tellurous acid stibine |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
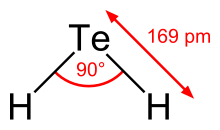
Hydrogen telluride is the inorganic compound with the formula H2 Te. A hydrogen chalcogenide and the simplest hydride of tellurium, it is a colorless gas. Although unstable in ambient air, the gas can exist long enough to be readily detected by the odour of rotting garlic at extremely low concentrations; or by the revolting odour of rotting leeks at somewhat higher concentrations. Most compounds with Te–H bonds ( tellurols) are unstable with respect to loss of H2. H2Te is chemically and structurally similar to hydrogen selenide, both are acidic. The H–Te–H angle is about 90°. Volatile tellurium compounds often have unpleasant odours, reminiscent of decayed leeks or garlic. [2]
Synthesis
Electrolytic methods have been developed. [3]
H2Te can also be prepared by hydrolysis of the telluride derivatives of electropositive metals. [4] The typical hydrolysis is that of aluminium telluride:
- Al2Te3 + 6 H2O → 2 Al(OH)3 + 3 H2Te
Other salts of Te2− such as MgTe and
sodium telluride can also be used. Na2Te can be made by the reaction of Na and Te in anhydrous
ammonia.
[5] The intermediate in the hydrolysis, HTe−
, can be isolated as salts as well. NaHTe can be made by reducing tellurium with
NaBH
4.
[5]
Hydrogen telluride cannot be efficiently prepared from its constituent elements, in contrast to H2Se. [3]
Properties
H
2Te is an
endothermic compound, degrading to the elements at room temperature:
- H
2Te → H
2 + Te
Light accelerates the decomposition. It is unstable in air, being oxidized to water and elemental tellurium: [6]
- 2 H
2Te + O
2 → 2 H
2O + 2 Te
It is almost as acidic as phosphoric acid (Ka = 8.1×10−3), having a Ka value of about 2.3×10−3. [6] It reacts with many metals to form tellurides. [7]
See also
References
- ^ Lide, David R., ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, FL: CRC Press. ISBN 0-8493-0487-3.
- ^ Greenwood, N. N.; & Earnshaw, A. (1997). Chemistry of the Elements (2nd Edn.), Oxford:Butterworth-Heinemann. ISBN 0-7506-3365-4.
- ^ a b F. Fehér, "Hydrogen Telluride" in Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. pp. 438.
- ^ Shriver, Atkins. Inorganic Chemistry, Fifth Edition. W. H. Freeman and Company, New York, 2010; pp 407.
- ^ a b Nicola Petragnani; Hélio A. Stefani (2007). Tellurium in organic synthesis. Best synthetic methods (2nd ed.). Academic Press. p. 6. ISBN 978-0-08-045310-1.
- ^ a b Egon Wiberg; Arnold Frederick Holleman (2001). Nils Wiberg (ed.). Inorganic chemistry. Translated by Mary Eagleson. Academic Press. p. 589. ISBN 0-12-352651-5.
- ^ Henry Enfield Roscoe; Carl Schorlemmer (1878). A treatise on chemistry. Vol. 1. Appleton. pp. 367–368.
 Liquid hydrogen telluride in a test tube
| |

| |
 Tellurium, Te Hydrogen, H | |
| Names | |
|---|---|
|
IUPAC name
hydrogen telluride
| |
| Other names
hydrotelluric acid
tellane tellurium hydride dihydrogen telluride tellurane | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.029.073 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| H2Te | |
| Molar mass | 129.6158 g mol−1 |
| Appearance | colourless gas |
| Odor | Pungent, resembles rotting garlic or leeks |
| Density | 3.310 g/L, gas 2.57 g/cm3 (−20 °C, liquid) |
| Melting point | −49 °C (−56 °F; 224 K) [1] |
| Boiling point | −2.2 °C (28.0 °F; 270.9 K) (unstable above −2 °C) |
| 0.70 g/100 mL | |
| Acidity (pKa) | 2.6 |
| Conjugate acid | Telluronium |
| Conjugate base | Telluride |
| Structure | |
| bent | |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
0.7684 kJ/g |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
toxic |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other
anions
|
H2O H2S H2Se H2Po |
Other
cations
|
Na2Te Ag2Te K2Te Rb2Te Cs2Te |
Related compounds
|
telluric acid tellurous acid stibine |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Hydrogen telluride is the inorganic compound with the formula H2 Te. A hydrogen chalcogenide and the simplest hydride of tellurium, it is a colorless gas. Although unstable in ambient air, the gas can exist long enough to be readily detected by the odour of rotting garlic at extremely low concentrations; or by the revolting odour of rotting leeks at somewhat higher concentrations. Most compounds with Te–H bonds ( tellurols) are unstable with respect to loss of H2. H2Te is chemically and structurally similar to hydrogen selenide, both are acidic. The H–Te–H angle is about 90°. Volatile tellurium compounds often have unpleasant odours, reminiscent of decayed leeks or garlic. [2]
Synthesis
Electrolytic methods have been developed. [3]
H2Te can also be prepared by hydrolysis of the telluride derivatives of electropositive metals. [4] The typical hydrolysis is that of aluminium telluride:
- Al2Te3 + 6 H2O → 2 Al(OH)3 + 3 H2Te
Other salts of Te2− such as MgTe and
sodium telluride can also be used. Na2Te can be made by the reaction of Na and Te in anhydrous
ammonia.
[5] The intermediate in the hydrolysis, HTe−
, can be isolated as salts as well. NaHTe can be made by reducing tellurium with
NaBH
4.
[5]
Hydrogen telluride cannot be efficiently prepared from its constituent elements, in contrast to H2Se. [3]
Properties
H
2Te is an
endothermic compound, degrading to the elements at room temperature:
- H
2Te → H
2 + Te
Light accelerates the decomposition. It is unstable in air, being oxidized to water and elemental tellurium: [6]
- 2 H
2Te + O
2 → 2 H
2O + 2 Te
It is almost as acidic as phosphoric acid (Ka = 8.1×10−3), having a Ka value of about 2.3×10−3. [6] It reacts with many metals to form tellurides. [7]
See also
References
- ^ Lide, David R., ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, FL: CRC Press. ISBN 0-8493-0487-3.
- ^ Greenwood, N. N.; & Earnshaw, A. (1997). Chemistry of the Elements (2nd Edn.), Oxford:Butterworth-Heinemann. ISBN 0-7506-3365-4.
- ^ a b F. Fehér, "Hydrogen Telluride" in Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. pp. 438.
- ^ Shriver, Atkins. Inorganic Chemistry, Fifth Edition. W. H. Freeman and Company, New York, 2010; pp 407.
- ^ a b Nicola Petragnani; Hélio A. Stefani (2007). Tellurium in organic synthesis. Best synthetic methods (2nd ed.). Academic Press. p. 6. ISBN 978-0-08-045310-1.
- ^ a b Egon Wiberg; Arnold Frederick Holleman (2001). Nils Wiberg (ed.). Inorganic chemistry. Translated by Mary Eagleson. Academic Press. p. 589. ISBN 0-12-352651-5.
- ^ Henry Enfield Roscoe; Carl Schorlemmer (1878). A treatise on chemistry. Vol. 1. Appleton. pp. 367–368.
