| |||

| |||
| Names | |||
|---|---|---|---|
|
IUPAC name
Hydrogen peroxide–urea (1/1)
| |||
|
Systematic IUPAC name
Peroxol–carbonyl diamide (1/1) | |||
| Other names
Urea peroxide, percarbamide, UHP
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.004.275 | ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| CH6N2O3 | |||
| Molar mass | 94.070 g·mol−1 | ||
| Appearance | White solid | ||
| Density | 1.50 g/cm3 | ||
| Melting point | 75 to 91.5 °C (167.0 to 196.7 °F; 348.1 to 364.6 K) (decomposes) | ||
| Pharmacology | |||
| D02AE01 ( WHO) | |||
| Hazards | |||
| GHS labelling: [1] | |||


| |||
| Danger | |||
| H272, H315, H318 | |||
| P210, P220, P264, P280, P302+P352, P305+P351+P338 | |||
| Flash point | 60 °C (140 °F; 333 K) | ||
| Safety data sheet (SDS) | External MSDS | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hydrogen peroxide–urea (also called Hyperol, artizone, urea hydrogen peroxide, and UHP) is a white crystalline solid chemical compound composed of equal amounts of hydrogen peroxide and urea. It contains solid and water-free hydrogen peroxide, which offers a higher stability and better controllability than liquid hydrogen peroxide when used as an oxidizing agent. Often called carbamide peroxide in dentistry, it is used as a source of hydrogen peroxide when dissolved in water for bleaching, disinfection and oxidation.
Production
For the preparation of the complex, urea is dissolved in 30% hydrogen peroxide (molar ratio 2:3) at temperatures below 60 °C. upon cooling this solution, hydrogen peroxide–urea precipitates in the form of small platelets. [2]
Akin to water of crystallization, hydrogen peroxide cocrystallizes with urea with the stoichiometry of 1:1. The compound is simply produced (on a scale of several hundred tonnes a year) by the dissolution of urea in excess concentrated hydrogen peroxide solution, followed by crystallization. [3] The laboratory synthesis is analogous. [4]
Structure and properties
The solid state structure of this adduct has been determined by neutron diffraction. [5]
Hydrogen peroxide–urea is a readily water-soluble, odorless, crystalline solid, which is available as white powder or colorless needles or platelets. [2] Upon dissolving in various solvents, the 1:1 complex dissociates back to urea and hydrogen peroxide. So just like hydrogen peroxide, the (erroneously) so-called adduct is an oxidizer but the release at room temperature in the presence of catalysts proceeds in a controlled manner. Thus the compound is suitable as a safe substitute for the unstable aqueous solution of hydrogen peroxide. Because of the tendency for thermal decomposition, which accelerates at temperatures above 82 °C, [6] it should not be heated above 60 °C, particularly in pure form.
The solubility of commercial samples varies from 0.05 g/mL [7] to more than 0.6 g/mL. [8]
Applications
Disinfectant and bleaching agent
Hydrogen peroxide–urea is mainly used as a disinfecting and bleaching agent in cosmetics and pharmaceuticals. [3] As a drug, this compound is used in some preparations for the whitening of teeth. [3] [9] [10] It is also used to relieve minor inflammation of gums, oral mucosal surfaces and lips including canker sores and dental irritation, [11] and to emulsify and disperse earwax. [12]
Carbamide peroxide is also suitable as a disinfectant, e.g. for germ reduction on contact lens surfaces or as an antiseptic for mouthwashes, ear drops or for superficial wounds and ulcers.
Reagent in organic synthesis
In the laboratory, it is used as a more easily handled replacement for hydrogen peroxide. [4] [13] [14] It has proven to be a stable, easy-to-handle and effective oxidizing agent which is readily controllable by a suitable choice of the reaction conditions. It delivers oxidation products in an environmentally friendly manner and often in high yields especially in the presence of organic catalysts such as cis-butenedioic anhydride [15] or inorganic catalysts such as sodium tungstate. [16]

It converts thiols selectively to disulfides, [15] secondary alcohols to ketones, [16] sulfides to sulfoxides and sulfones, [17] nitriles to amides, [17] [18] and N-heterocycles to amine oxides. [17] [19]

Hydroxybenzaldehydes are converted to dihydroxybenzenes ( Dakin reaction) [17] [20] and give, under suitable conditions, the corresponding benzoic acids. [20]
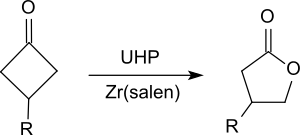
It oxidizes ketones to esters, in particular cyclic ketones, such as substituted cyclohexanones [21] or cyclobutanones [22] to give lactones ( Baeyer–Villiger oxidation).
The epoxidation of various alkenes in the presence of benzonitrile yields oxiranes in yields of 79 to 96%. [23]
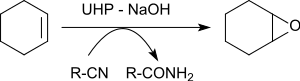
The oxygen atom transferred to the alkene originates from the peroxoimide acid formed intermediately from benzonitrile. The resulting imidic acid tautomerizes to the benzamide.
Safety
The compound acts as a strong oxidizing agent and can cause skin irritation and severe eye damage. [24] Urea–hydrogen peroxide was also found to be an insensitive high explosive, capable of detonation by strong impulse under heavy confinement. [25] [26]
See also
References
- ^ GHS: Sigma-Aldrich 289132
- ^ a b C.-S. Lu; E.W. Hughes; P.A. Giguère (1941), "The crystal structure of the urea-hydrogen peroxide addition compound CO(NH2)2 H2O2", J. Am. Chem. Soc., vol. 63, no. 6, pp. 1507–1513, doi: 10.1021/ja01851a007
- ^ a b c Harald Jakob; Stefan Leininger; Thomas Lehmann; Sylvia Jacobi; Sven Gutewort. "Peroxo Compounds, Inorganic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a19_177.pub2. ISBN 978-3527306732.
- ^ a b Yu, Lei; Meng, Bo; Huang, Xian (2008). "Urea-Hydrogen Peroxide Complex: A Selective Oxidant in the Synthesis of 2-Phenylselenyl-1,3-butadienes". Synthetic Communications. 38 (18): 3142. doi: 10.1080/00397910802109224. S2CID 98323467.
- ^ Fritchie, C. J. Jr.; McMullan, R. K. (1981). "Neutron Diffraction Study of the 1:1 Urea:Hydrogen Peroxide complex at 81 K". Acta Crystallographica Section B. 37 (5): 1086. doi: 10.1107/S0567740881005116.
-
^ H. Heaney; F. Cardona; A. Goti; A.L. Frederick (2013). "Hydrogen Peroxide-Urea". Encyclopedia of Reagents for Organic Synthesis.
doi:
10.1002/047084289X.rh047.pub3.
ISBN
978-0471936237.
{{ cite book}}:|periodical=ignored ( help) - ^ Sigma-Aldrich specification sheet
- ^ Chemicalland data sheet
- ^ Mokhlis, G. R.; Matis, B. A.; Cochran, M. A.; Eckert, G. J. (2000). "A Clinical Evaluation of Carbamide Peroxide and Hydrogen Peroxide Whitening Agents during Daytime Use". Journal of the American Dental Association. 131 (9): 1269–77. doi: 10.14219/jada.archive.2000.0380. PMID 10986827. Archived from the original on 2013-02-23.
- ^ Toothwhitening Archived 2008-03-17 at the Wayback Machine from the UMD of New Jersey website
- ^ Center for Integrative Medicine: Carbamide Peroxide from the University of Maryland Medical Center website Archived October 18, 2007, at the Wayback Machine
- ^ "Ear Drops GENERIC NAME(S): CARBAMIDE PEROXIDE". WebMD. Retrieved July 3, 2021.
- ^ Varma, Rajender S.; Naicker, Kannan P. (1999). "The Urea−Hydrogen Peroxide Complex: Solid-State Oxidative Protocols for Hydroxylated Aldehydes and Ketones (Dakin Reaction), Nitriles, Sulfides, and Nitrogen Heterocycles". Organic Letters. 1 (2): 189. doi: 10.1021/ol990522n.
- ^ Harry Heaney, Francesca Cardona, Andrea Goti, "Hydrogen Peroxide–Urea" Encyclopedia of Reagents for Organic Synthesis 2008. doi: 10.1002/047084289X.rh047.pub2
- ^ a b B. Karami; M. Montazerozohori; M. H. Habibi (2005), "Urea-Hydrogen Peroxide (UHP) oxidation of thiols to the corresponding disulfides promoted by maleic anhydride as mediator" (PDF), Molecules (in German), vol. 10, no. 10, pp. 1358–1363, doi: 10.3390/10101385, PMC 6147623, PMID 18007530
- ^ a b M. Lukasiewicz; D. Bogdal; J. Pielichowski. "Microwave-assisted oxidation of alcohols using urea hydrogen peroxide". 8th International Electronic Conference on Synthetic Organic Chemistry. ECSOC-8. Retrieved 2016-05-10.
- ^ a b c d R.S. Varma, K.P. Naicker, "The Urea-Hydrogen Peroxide Complex: Solid-State Oxidative Protocols for Hydroxylated Aldehydes and Ketones (Dakin Reaction), Nitriles, Sulfides, and Nitrogen Heterocycles", Org. Lett. (in German), vol. 1, no. 2, pp. 189–191, doi: 10.1021/ol990522n
- ^ WO patent 2012069948, V. Mascitti, K.F. McClure, M.J. Munchhof, R.P. Robinson, Jr., "4-(5-Cyano-pyrazol-1-yl)-piperidine derivatives as GPR 119 modulators", issued 2012-5-31, assigned to Pfizer Inc.
- ^ D. Rong; V.A. Phillips; R.S. Rubio; M.A. Castro; R.T. Wheelhouse, "A safe, convenient and efficient method for the preparation of heterocyclic N-oxides using urea-hydrogen peroxide", Tetrahedron Lett. (in German), vol. 49, no. 48, pp. 6933–6935, doi: 10.1016/j.tetlet.2008.09.124
- ^ a b H. Heaney; A.J. Newbold (2001), "The oxidation of aromatic aldehydes by magnesium monoperoxyphthalate and urea-hydrogen peroxide", Tetrahedron Lett. (in German), vol. 42, no. 37, pp. 6607–6609, doi: 10.1016/S0040-4039(01)01332-6
- ^ M.Y. Rios; E. Salazar; H.F. Olivo (2007), "Baeyer–Villiger oxidation of substituted cyclohexanones via lipase-mediated perhydrolysis utilizing urea–hydrogen peroxide in ethyl acetate", Green Chem. (in German), vol. 9, no. 5, pp. 459–462, doi: 10.1039/B618175A
- ^ A. Watanabe; T. Uchida; K. Ito; T. Katsuki (2002), "Highly enantioselective Baeyer-Villiger oxidation using Zr(salen) complex as catalyst", Tetrahedron Lett. (in German), vol. 43, no. 25, pp. 4481–4485, doi: 10.1016/S0040-4039(02)00831-6
- ^ L. Ji; Y.-N. Wang; C. Qian; X.-Z. Chen (2013), "Nitrile-promoted alkene epoxidation with urea-hydrogen peroxide (UHP)", Synth. Commun. (in German), vol. 43, no. 16, pp. 2256–2264, doi: 10.1080/00397911.2012.699578, S2CID 93770740
- ^ "Hydrogen peroxide urea SDS". merckmillipore.com. 16 May 2023.
- ^ Halleux, Francis; Pons, Jean-François; Wilson, Ian; Van Riet, Romuald; Lefebvre, Michel (2022). "Small-Scale Detonation of Industrial Urea-Hydrogen Peroxide (UHP)". Propellants, Explosives, Pyrotechnics. 47 (2). doi: 10.1002/prep.202100250. hdl: 1826/17469. S2CID 244899815.
- ^ Halleux, Francis; Pons, Jean-François; Wilson, Ian; Simoens, Bart; Van Riet, Romuald; Lefebvre, Michel (2023). "Detonation performance of urea-hydrogen peroxide (UHP)". Propellants, Explosives, Pyrotechnics. 48 (6). doi: 10.1002/prep.202300011. S2CID 257196173.
External links
- "Hydrogen peroxide urea adduct, UHP". Organic Chemistry Portal.
- "Carbamide Peroxide Monograph". Drugs.com.
| |||

| |||
| Names | |||
|---|---|---|---|
|
IUPAC name
Hydrogen peroxide–urea (1/1)
| |||
|
Systematic IUPAC name
Peroxol–carbonyl diamide (1/1) | |||
| Other names
Urea peroxide, percarbamide, UHP
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.004.275 | ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| CH6N2O3 | |||
| Molar mass | 94.070 g·mol−1 | ||
| Appearance | White solid | ||
| Density | 1.50 g/cm3 | ||
| Melting point | 75 to 91.5 °C (167.0 to 196.7 °F; 348.1 to 364.6 K) (decomposes) | ||
| Pharmacology | |||
| D02AE01 ( WHO) | |||
| Hazards | |||
| GHS labelling: [1] | |||


| |||
| Danger | |||
| H272, H315, H318 | |||
| P210, P220, P264, P280, P302+P352, P305+P351+P338 | |||
| Flash point | 60 °C (140 °F; 333 K) | ||
| Safety data sheet (SDS) | External MSDS | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hydrogen peroxide–urea (also called Hyperol, artizone, urea hydrogen peroxide, and UHP) is a white crystalline solid chemical compound composed of equal amounts of hydrogen peroxide and urea. It contains solid and water-free hydrogen peroxide, which offers a higher stability and better controllability than liquid hydrogen peroxide when used as an oxidizing agent. Often called carbamide peroxide in dentistry, it is used as a source of hydrogen peroxide when dissolved in water for bleaching, disinfection and oxidation.
Production
For the preparation of the complex, urea is dissolved in 30% hydrogen peroxide (molar ratio 2:3) at temperatures below 60 °C. upon cooling this solution, hydrogen peroxide–urea precipitates in the form of small platelets. [2]
Akin to water of crystallization, hydrogen peroxide cocrystallizes with urea with the stoichiometry of 1:1. The compound is simply produced (on a scale of several hundred tonnes a year) by the dissolution of urea in excess concentrated hydrogen peroxide solution, followed by crystallization. [3] The laboratory synthesis is analogous. [4]
Structure and properties
The solid state structure of this adduct has been determined by neutron diffraction. [5]
Hydrogen peroxide–urea is a readily water-soluble, odorless, crystalline solid, which is available as white powder or colorless needles or platelets. [2] Upon dissolving in various solvents, the 1:1 complex dissociates back to urea and hydrogen peroxide. So just like hydrogen peroxide, the (erroneously) so-called adduct is an oxidizer but the release at room temperature in the presence of catalysts proceeds in a controlled manner. Thus the compound is suitable as a safe substitute for the unstable aqueous solution of hydrogen peroxide. Because of the tendency for thermal decomposition, which accelerates at temperatures above 82 °C, [6] it should not be heated above 60 °C, particularly in pure form.
The solubility of commercial samples varies from 0.05 g/mL [7] to more than 0.6 g/mL. [8]
Applications
Disinfectant and bleaching agent
Hydrogen peroxide–urea is mainly used as a disinfecting and bleaching agent in cosmetics and pharmaceuticals. [3] As a drug, this compound is used in some preparations for the whitening of teeth. [3] [9] [10] It is also used to relieve minor inflammation of gums, oral mucosal surfaces and lips including canker sores and dental irritation, [11] and to emulsify and disperse earwax. [12]
Carbamide peroxide is also suitable as a disinfectant, e.g. for germ reduction on contact lens surfaces or as an antiseptic for mouthwashes, ear drops or for superficial wounds and ulcers.
Reagent in organic synthesis
In the laboratory, it is used as a more easily handled replacement for hydrogen peroxide. [4] [13] [14] It has proven to be a stable, easy-to-handle and effective oxidizing agent which is readily controllable by a suitable choice of the reaction conditions. It delivers oxidation products in an environmentally friendly manner and often in high yields especially in the presence of organic catalysts such as cis-butenedioic anhydride [15] or inorganic catalysts such as sodium tungstate. [16]

It converts thiols selectively to disulfides, [15] secondary alcohols to ketones, [16] sulfides to sulfoxides and sulfones, [17] nitriles to amides, [17] [18] and N-heterocycles to amine oxides. [17] [19]

Hydroxybenzaldehydes are converted to dihydroxybenzenes ( Dakin reaction) [17] [20] and give, under suitable conditions, the corresponding benzoic acids. [20]

It oxidizes ketones to esters, in particular cyclic ketones, such as substituted cyclohexanones [21] or cyclobutanones [22] to give lactones ( Baeyer–Villiger oxidation).
The epoxidation of various alkenes in the presence of benzonitrile yields oxiranes in yields of 79 to 96%. [23]

The oxygen atom transferred to the alkene originates from the peroxoimide acid formed intermediately from benzonitrile. The resulting imidic acid tautomerizes to the benzamide.
Safety
The compound acts as a strong oxidizing agent and can cause skin irritation and severe eye damage. [24] Urea–hydrogen peroxide was also found to be an insensitive high explosive, capable of detonation by strong impulse under heavy confinement. [25] [26]
See also
References
- ^ GHS: Sigma-Aldrich 289132
- ^ a b C.-S. Lu; E.W. Hughes; P.A. Giguère (1941), "The crystal structure of the urea-hydrogen peroxide addition compound CO(NH2)2 H2O2", J. Am. Chem. Soc., vol. 63, no. 6, pp. 1507–1513, doi: 10.1021/ja01851a007
- ^ a b c Harald Jakob; Stefan Leininger; Thomas Lehmann; Sylvia Jacobi; Sven Gutewort. "Peroxo Compounds, Inorganic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a19_177.pub2. ISBN 978-3527306732.
- ^ a b Yu, Lei; Meng, Bo; Huang, Xian (2008). "Urea-Hydrogen Peroxide Complex: A Selective Oxidant in the Synthesis of 2-Phenylselenyl-1,3-butadienes". Synthetic Communications. 38 (18): 3142. doi: 10.1080/00397910802109224. S2CID 98323467.
- ^ Fritchie, C. J. Jr.; McMullan, R. K. (1981). "Neutron Diffraction Study of the 1:1 Urea:Hydrogen Peroxide complex at 81 K". Acta Crystallographica Section B. 37 (5): 1086. doi: 10.1107/S0567740881005116.
-
^ H. Heaney; F. Cardona; A. Goti; A.L. Frederick (2013). "Hydrogen Peroxide-Urea". Encyclopedia of Reagents for Organic Synthesis.
doi:
10.1002/047084289X.rh047.pub3.
ISBN
978-0471936237.
{{ cite book}}:|periodical=ignored ( help) - ^ Sigma-Aldrich specification sheet
- ^ Chemicalland data sheet
- ^ Mokhlis, G. R.; Matis, B. A.; Cochran, M. A.; Eckert, G. J. (2000). "A Clinical Evaluation of Carbamide Peroxide and Hydrogen Peroxide Whitening Agents during Daytime Use". Journal of the American Dental Association. 131 (9): 1269–77. doi: 10.14219/jada.archive.2000.0380. PMID 10986827. Archived from the original on 2013-02-23.
- ^ Toothwhitening Archived 2008-03-17 at the Wayback Machine from the UMD of New Jersey website
- ^ Center for Integrative Medicine: Carbamide Peroxide from the University of Maryland Medical Center website Archived October 18, 2007, at the Wayback Machine
- ^ "Ear Drops GENERIC NAME(S): CARBAMIDE PEROXIDE". WebMD. Retrieved July 3, 2021.
- ^ Varma, Rajender S.; Naicker, Kannan P. (1999). "The Urea−Hydrogen Peroxide Complex: Solid-State Oxidative Protocols for Hydroxylated Aldehydes and Ketones (Dakin Reaction), Nitriles, Sulfides, and Nitrogen Heterocycles". Organic Letters. 1 (2): 189. doi: 10.1021/ol990522n.
- ^ Harry Heaney, Francesca Cardona, Andrea Goti, "Hydrogen Peroxide–Urea" Encyclopedia of Reagents for Organic Synthesis 2008. doi: 10.1002/047084289X.rh047.pub2
- ^ a b B. Karami; M. Montazerozohori; M. H. Habibi (2005), "Urea-Hydrogen Peroxide (UHP) oxidation of thiols to the corresponding disulfides promoted by maleic anhydride as mediator" (PDF), Molecules (in German), vol. 10, no. 10, pp. 1358–1363, doi: 10.3390/10101385, PMC 6147623, PMID 18007530
- ^ a b M. Lukasiewicz; D. Bogdal; J. Pielichowski. "Microwave-assisted oxidation of alcohols using urea hydrogen peroxide". 8th International Electronic Conference on Synthetic Organic Chemistry. ECSOC-8. Retrieved 2016-05-10.
- ^ a b c d R.S. Varma, K.P. Naicker, "The Urea-Hydrogen Peroxide Complex: Solid-State Oxidative Protocols for Hydroxylated Aldehydes and Ketones (Dakin Reaction), Nitriles, Sulfides, and Nitrogen Heterocycles", Org. Lett. (in German), vol. 1, no. 2, pp. 189–191, doi: 10.1021/ol990522n
- ^ WO patent 2012069948, V. Mascitti, K.F. McClure, M.J. Munchhof, R.P. Robinson, Jr., "4-(5-Cyano-pyrazol-1-yl)-piperidine derivatives as GPR 119 modulators", issued 2012-5-31, assigned to Pfizer Inc.
- ^ D. Rong; V.A. Phillips; R.S. Rubio; M.A. Castro; R.T. Wheelhouse, "A safe, convenient and efficient method for the preparation of heterocyclic N-oxides using urea-hydrogen peroxide", Tetrahedron Lett. (in German), vol. 49, no. 48, pp. 6933–6935, doi: 10.1016/j.tetlet.2008.09.124
- ^ a b H. Heaney; A.J. Newbold (2001), "The oxidation of aromatic aldehydes by magnesium monoperoxyphthalate and urea-hydrogen peroxide", Tetrahedron Lett. (in German), vol. 42, no. 37, pp. 6607–6609, doi: 10.1016/S0040-4039(01)01332-6
- ^ M.Y. Rios; E. Salazar; H.F. Olivo (2007), "Baeyer–Villiger oxidation of substituted cyclohexanones via lipase-mediated perhydrolysis utilizing urea–hydrogen peroxide in ethyl acetate", Green Chem. (in German), vol. 9, no. 5, pp. 459–462, doi: 10.1039/B618175A
- ^ A. Watanabe; T. Uchida; K. Ito; T. Katsuki (2002), "Highly enantioselective Baeyer-Villiger oxidation using Zr(salen) complex as catalyst", Tetrahedron Lett. (in German), vol. 43, no. 25, pp. 4481–4485, doi: 10.1016/S0040-4039(02)00831-6
- ^ L. Ji; Y.-N. Wang; C. Qian; X.-Z. Chen (2013), "Nitrile-promoted alkene epoxidation with urea-hydrogen peroxide (UHP)", Synth. Commun. (in German), vol. 43, no. 16, pp. 2256–2264, doi: 10.1080/00397911.2012.699578, S2CID 93770740
- ^ "Hydrogen peroxide urea SDS". merckmillipore.com. 16 May 2023.
- ^ Halleux, Francis; Pons, Jean-François; Wilson, Ian; Van Riet, Romuald; Lefebvre, Michel (2022). "Small-Scale Detonation of Industrial Urea-Hydrogen Peroxide (UHP)". Propellants, Explosives, Pyrotechnics. 47 (2). doi: 10.1002/prep.202100250. hdl: 1826/17469. S2CID 244899815.
- ^ Halleux, Francis; Pons, Jean-François; Wilson, Ian; Simoens, Bart; Van Riet, Romuald; Lefebvre, Michel (2023). "Detonation performance of urea-hydrogen peroxide (UHP)". Propellants, Explosives, Pyrotechnics. 48 (6). doi: 10.1002/prep.202300011. S2CID 257196173.
External links
- "Hydrogen peroxide urea adduct, UHP". Organic Chemistry Portal.
- "Carbamide Peroxide Monograph". Drugs.com.

