
| |
| Names | |
|---|---|
|
IUPAC name
(2S,3R,4S)-3,4-dihydroxy-2-[(2R,3R,4S,5R)-4-hydroxy-2,5-dimethoxyoxan-3-yl]oxy-3,4-dihydro-2H-pyran-6-carboxylic acid
| |
| Other names
4-deoxy-4-hexenopyranosyluronic acid
4-deoxy-beta-L-threo-hex-4-enopyranosyluronic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| Abbreviations | HexA |
| MeSH | C095263 |
PubChem
CID
|
|
| |
| |
| Properties | |
| C13H20O10 | |
| Molar mass | 336.293 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Hexenuronic acid (HexA) is a bicyclic organic compound with the formula C13H20O10. [1] [2] It is an unsaturated sugar produced during the kraft process in the creation of wood pulp. [2]
Kraft process
During the kraft process, which is the turning of wood into wood pulp for papermaking, wood chips are treated with sodium hydroxide and sodium sulfide. Sodium hydroxide catalyzes the demethylation of 4-O-methyl-D-glucuronoxylan, which is found at the ends of the polysaccharide xylan. [2] [1] [3]
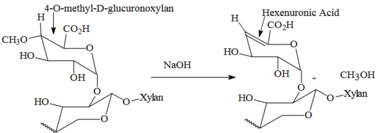
Hexenuronic acid decreases a wood's kappa number, which is a measure of bleachability of wood pulp, by 3-7. [3] It readily reacts with common wood pulp bleaching agents like ozone, peracetic acid, and chlorine dioxide. [2] [1] Consequently, research has focused on ways to break down hexenuronic acid prior to bleaching to decrease dangerous waste products and costs. [3]
The main method of destroying hexenuronic acid is to treat the wood pulp post kraft processing with strong acids at high temperatures. HexA is hydrolyzed and broken down into aldehydes and alcohols like 2-furoic acid and 5-carboxy-2-furaldehdye. [3] [2] This process has led to a 50% reduction in bleaching costs of the wood pulp in some cases. [2]
In microbes
Polysaccharide lyases (PL) are a type of enzyme that is found in numerous microorganisms including bacteriophages that break down parts of wood. [4] PL catalyzses β-elimination of uronic acid-containing polysaccharides into HexA. [4] [5]
References
- ^ a b c Cadena EM, Vidal T, Torres AL (May 2010). "Influence of the hexenuronic acid content on refining and ageing in eucalyptus TCF pulp". Bioresource Technology. 101 (10): 3554–3560. doi: 10.1016/j.biortech.2009.11.105. PMID 20074936.
- ^ a b c d e f Chakar FS, Allison L, Ragauskas AJ, McDonough TJ, Sezgi US (November 2000). "Influence of hexenuronic acids on us bleaching operations". TAPPI Journal. 83 (11).
- ^ a b c d Petit-Breuilh X, Zaror C, Melo R (December 2004). "Hexenuronic acid removal from unbleached kraft eucalyptus pulp by peroxymonosulfuric acid". Journal of the Chilean Chemical Society. 49 (4): 355–360. doi: 10.4067/S0717-97072004000400016. ISSN 0717-9707.
- ^ a b Sutherland IW (July 1995). "Polysaccharide lyases". FEMS Microbiology Reviews. 16 (4): 323–347. doi: 10.1111/j.1574-6976.1995.tb00179.x. PMID 7654407.
- ^ Quan Y, da Silva NM, de Souza Lima BJ, de Hoog S, Vicente VA, Mayer V, et al. (March 2022). "Black fungi and ants: a genomic comparison of species inhabiting carton nests versus domatia". IMA Fungus. 13 (1): 4. doi: 10.1186/s43008-022-00091-5. PMC 8900376. PMID 35256015.

| |
| Names | |
|---|---|
|
IUPAC name
(2S,3R,4S)-3,4-dihydroxy-2-[(2R,3R,4S,5R)-4-hydroxy-2,5-dimethoxyoxan-3-yl]oxy-3,4-dihydro-2H-pyran-6-carboxylic acid
| |
| Other names
4-deoxy-4-hexenopyranosyluronic acid
4-deoxy-beta-L-threo-hex-4-enopyranosyluronic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| Abbreviations | HexA |
| MeSH | C095263 |
PubChem
CID
|
|
| |
| |
| Properties | |
| C13H20O10 | |
| Molar mass | 336.293 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Hexenuronic acid (HexA) is a bicyclic organic compound with the formula C13H20O10. [1] [2] It is an unsaturated sugar produced during the kraft process in the creation of wood pulp. [2]
Kraft process
During the kraft process, which is the turning of wood into wood pulp for papermaking, wood chips are treated with sodium hydroxide and sodium sulfide. Sodium hydroxide catalyzes the demethylation of 4-O-methyl-D-glucuronoxylan, which is found at the ends of the polysaccharide xylan. [2] [1] [3]

Hexenuronic acid decreases a wood's kappa number, which is a measure of bleachability of wood pulp, by 3-7. [3] It readily reacts with common wood pulp bleaching agents like ozone, peracetic acid, and chlorine dioxide. [2] [1] Consequently, research has focused on ways to break down hexenuronic acid prior to bleaching to decrease dangerous waste products and costs. [3]
The main method of destroying hexenuronic acid is to treat the wood pulp post kraft processing with strong acids at high temperatures. HexA is hydrolyzed and broken down into aldehydes and alcohols like 2-furoic acid and 5-carboxy-2-furaldehdye. [3] [2] This process has led to a 50% reduction in bleaching costs of the wood pulp in some cases. [2]
In microbes
Polysaccharide lyases (PL) are a type of enzyme that is found in numerous microorganisms including bacteriophages that break down parts of wood. [4] PL catalyzses β-elimination of uronic acid-containing polysaccharides into HexA. [4] [5]
References
- ^ a b c Cadena EM, Vidal T, Torres AL (May 2010). "Influence of the hexenuronic acid content on refining and ageing in eucalyptus TCF pulp". Bioresource Technology. 101 (10): 3554–3560. doi: 10.1016/j.biortech.2009.11.105. PMID 20074936.
- ^ a b c d e f Chakar FS, Allison L, Ragauskas AJ, McDonough TJ, Sezgi US (November 2000). "Influence of hexenuronic acids on us bleaching operations". TAPPI Journal. 83 (11).
- ^ a b c d Petit-Breuilh X, Zaror C, Melo R (December 2004). "Hexenuronic acid removal from unbleached kraft eucalyptus pulp by peroxymonosulfuric acid". Journal of the Chilean Chemical Society. 49 (4): 355–360. doi: 10.4067/S0717-97072004000400016. ISSN 0717-9707.
- ^ a b Sutherland IW (July 1995). "Polysaccharide lyases". FEMS Microbiology Reviews. 16 (4): 323–347. doi: 10.1111/j.1574-6976.1995.tb00179.x. PMID 7654407.
- ^ Quan Y, da Silva NM, de Souza Lima BJ, de Hoog S, Vicente VA, Mayer V, et al. (March 2022). "Black fungi and ants: a genomic comparison of species inhabiting carton nests versus domatia". IMA Fungus. 13 (1): 4. doi: 10.1186/s43008-022-00091-5. PMC 8900376. PMID 35256015.