 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Hexalen |
| Other names | 2,4,6-Tris(dimethylamino)-1,3,5-triazine |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a601200 |
| License data |
|
|
Pregnancy category |
|
|
Routes of administration | Oral ( capsules) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 94% |
| Metabolism | Extensive liver |
| Metabolites | Pentamethylmelamine, tetramethylmelamine |
| Elimination half-life | 4.7–10.2 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.010.391 |
| Chemical and physical data | |
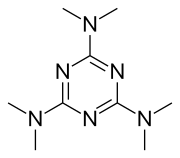
| Formula | C9H18N6 |
| Molar mass | 210.285 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Altretamine (trade name Hexalen), also called hexamethylmelamine, is an antineoplastic agent. It was approved by the U.S. FDA in 1990.
Uses
It is indicated for use as a single agent in the palliative treatment of patients with persistent or recurrent ovarian cancer following first-line therapy with cisplatin and/or alkylating agent-based combination. [1]
It is not considered a first-line treatment, [2] but it can be useful as salvage therapy. [3] It also has the advantage of being less toxic than other drugs used for treating refractory ovarian cancer. [4]
Mechanism
The precise mechanism by which altretamine exerts its anti-cancer effect is unknown but it is classified by MeSH as an alkylating antineoplastic agent. [5]
This unique structure is believed to damage tumor cells through the production of the weakly alkylating species formaldehyde, a product of CYP450-mediated N-demethylation. Administered orally, altretamine is extensively metabolized on first pass, producing primarily mono- and didemethylated metabolites. Additional demethylation reactions occur in tumor cells, releasing formaldehyde in situ before the drug is excreted in the urine. The carbinolamine (methylol) intermediates of CYP450-mediated metabolism also can generate electrophilic iminium species that are capable of reacting covalently with DNA guanine and cytosine residues as well as protein. Iminium-mediated DNA cross-linking and DNA-protein interstrand cross-linking, mediated through both the iminium intermediate and formaldehyde, have been demonstrated, although the significance of DNA cross-linking on altretamine antitumor activity is uncertain. [6]
Side effects
Side effects include nausea, vomiting, anemia and peripheral sensory neuropathy. [7]
Interactions
Combination with pyridoxine (vitamin B6) decreases neurotoxicity but has been found to reduce the effectiveness of an altretamine/ cisplatin regime. [8] MAO inhibitor can cause severe orthostatic hypotension when combined with altretamine; and cimetidine can increase its elimination half-life and toxicity. [7]
See also
References
- ^ "Hexalen (altretamine) Capsule. Human Prescription Drug Label". dailymed.nlm.nih.gov. Eisai Inc. Retrieved 24 August 2016.
- ^ Keldsen N, Havsteen H, Vergote I, Bertelsen K, Jakobsen A (February 2003). "Altretamine (hexamethylmelamine) in the treatment of platinum-resistant ovarian cancer: a phase II study". Gynecologic Oncology. 88 (2): 118–122. doi: 10.1016/S0090-8258(02)00103-8. PMID 12586589.
- ^ Chan JK, Loizzi V, Manetta A, Berman ML (January 2004). "Oral altretamine used as salvage therapy in recurrent ovarian cancer". Gynecologic Oncology. 92 (1): 368–371. doi: 10.1016/j.ygyno.2003.09.017. PMID 14751188.
- ^ Malik IA (February 2001). "Altretamine is an effective palliative therapy of patients with recurrent epithelial ovarian cancer". Japanese Journal of Clinical Oncology. 31 (2): 69–73. doi: 10.1093/jjco/hye012. PMID 11302345.
- ^ Damia G, D'Incalci M (June 1995). "Clinical pharmacokinetics of altretamine". Clinical Pharmacokinetics. 28 (6): 439–448. doi: 10.2165/00003088-199528060-00002. PMID 7656502. S2CID 28375029.
- ^ Lemke TL, Williams DA, eds. (2008). Foye's Principles of Medicinal Chemistry (6th ed.). Philadelphia: Lippincott Williams & Wilkins. ISBN 978-0-7817-6879-5.
- ^ a b "Altretamine Monograph". Drugs.com.
- ^ Wiernik PH, Yeap B, Vogl SE, Kaplan BH, Comis RL, Falkson G, et al. (1992). "Hexamethylmelamine and low or moderate dose cisplatin with or without pyridoxine for treatment of advanced ovarian carcinoma: a study of the Eastern Cooperative Oncology Group". Cancer Investigation. 10 (1): 1–9. doi: 10.3109/07357909209032783. PMID 1735009.
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Hexalen |
| Other names | 2,4,6-Tris(dimethylamino)-1,3,5-triazine |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a601200 |
| License data |
|
|
Pregnancy category |
|
|
Routes of administration | Oral ( capsules) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 94% |
| Metabolism | Extensive liver |
| Metabolites | Pentamethylmelamine, tetramethylmelamine |
| Elimination half-life | 4.7–10.2 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.010.391 |
| Chemical and physical data | |
| Formula | C9H18N6 |
| Molar mass | 210.285 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Altretamine (trade name Hexalen), also called hexamethylmelamine, is an antineoplastic agent. It was approved by the U.S. FDA in 1990.
Uses
It is indicated for use as a single agent in the palliative treatment of patients with persistent or recurrent ovarian cancer following first-line therapy with cisplatin and/or alkylating agent-based combination. [1]
It is not considered a first-line treatment, [2] but it can be useful as salvage therapy. [3] It also has the advantage of being less toxic than other drugs used for treating refractory ovarian cancer. [4]
Mechanism
The precise mechanism by which altretamine exerts its anti-cancer effect is unknown but it is classified by MeSH as an alkylating antineoplastic agent. [5]
This unique structure is believed to damage tumor cells through the production of the weakly alkylating species formaldehyde, a product of CYP450-mediated N-demethylation. Administered orally, altretamine is extensively metabolized on first pass, producing primarily mono- and didemethylated metabolites. Additional demethylation reactions occur in tumor cells, releasing formaldehyde in situ before the drug is excreted in the urine. The carbinolamine (methylol) intermediates of CYP450-mediated metabolism also can generate electrophilic iminium species that are capable of reacting covalently with DNA guanine and cytosine residues as well as protein. Iminium-mediated DNA cross-linking and DNA-protein interstrand cross-linking, mediated through both the iminium intermediate and formaldehyde, have been demonstrated, although the significance of DNA cross-linking on altretamine antitumor activity is uncertain. [6]
Side effects
Side effects include nausea, vomiting, anemia and peripheral sensory neuropathy. [7]
Interactions
Combination with pyridoxine (vitamin B6) decreases neurotoxicity but has been found to reduce the effectiveness of an altretamine/ cisplatin regime. [8] MAO inhibitor can cause severe orthostatic hypotension when combined with altretamine; and cimetidine can increase its elimination half-life and toxicity. [7]
See also
References
- ^ "Hexalen (altretamine) Capsule. Human Prescription Drug Label". dailymed.nlm.nih.gov. Eisai Inc. Retrieved 24 August 2016.
- ^ Keldsen N, Havsteen H, Vergote I, Bertelsen K, Jakobsen A (February 2003). "Altretamine (hexamethylmelamine) in the treatment of platinum-resistant ovarian cancer: a phase II study". Gynecologic Oncology. 88 (2): 118–122. doi: 10.1016/S0090-8258(02)00103-8. PMID 12586589.
- ^ Chan JK, Loizzi V, Manetta A, Berman ML (January 2004). "Oral altretamine used as salvage therapy in recurrent ovarian cancer". Gynecologic Oncology. 92 (1): 368–371. doi: 10.1016/j.ygyno.2003.09.017. PMID 14751188.
- ^ Malik IA (February 2001). "Altretamine is an effective palliative therapy of patients with recurrent epithelial ovarian cancer". Japanese Journal of Clinical Oncology. 31 (2): 69–73. doi: 10.1093/jjco/hye012. PMID 11302345.
- ^ Damia G, D'Incalci M (June 1995). "Clinical pharmacokinetics of altretamine". Clinical Pharmacokinetics. 28 (6): 439–448. doi: 10.2165/00003088-199528060-00002. PMID 7656502. S2CID 28375029.
- ^ Lemke TL, Williams DA, eds. (2008). Foye's Principles of Medicinal Chemistry (6th ed.). Philadelphia: Lippincott Williams & Wilkins. ISBN 978-0-7817-6879-5.
- ^ a b "Altretamine Monograph". Drugs.com.
- ^ Wiernik PH, Yeap B, Vogl SE, Kaplan BH, Comis RL, Falkson G, et al. (1992). "Hexamethylmelamine and low or moderate dose cisplatin with or without pyridoxine for treatment of advanced ovarian carcinoma: a study of the Eastern Cooperative Oncology Group". Cancer Investigation. 10 (1): 1–9. doi: 10.3109/07357909209032783. PMID 1735009.