| |||

AFM image of hexabenzocoronene
| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
Hexabenzo[bc,ef,hi,kl,no,qr]coronene | |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C42H18 | |||
| Molar mass | 522.606 g·mol−1 | ||
| Appearance | dark yellow [1] | ||
| Density | 1.54 g/cm3 (calc.) [1] | ||
| -346.0·10−6 cm3/mol [2] | |||
| Structure [1] | |||
| monoclinic, P21/a | |||
a = 1.8431(3) nm, b = 0.5119(1) nm, c = 1.2929(2) nm α = 90°, β = 112.57(1)°, γ = 90°
| |||
Formula units (Z)
|
2 | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
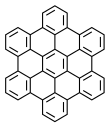
Hexa-peri-hexabenzocoronene (HBC) is a polycyclic aromatic hydrocarbon with the molecular formula C42H18. It consists of a central coronene molecule, with an additional benzene ring fused between each adjacent pair of rings around the periphery. It is sometimes simply called hexabenzocoronene, however, there are other chemicals that share this less-specific name, such as hexa-cata-hexabenzocoronene.
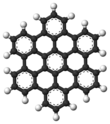
Hexa-peri-hexabenzocoronene has been imaged by atomic force microscopy (AFM) providing the first example of a molecule in which differences in bond order and bond lengths of the individual bonds can be distinguished by a measurement in direct space. [3]
Supramolecular structures
Various hexabenzocoronenes have been investigated in
supramolecular electronics. They are known to
self-assemble into a
columnar phase. One derivative in particular forms
carbon nanotubes with interesting electrical properties.
[4] The columnar phase in this compound further organises itself into sheets, which ultimately roll up like a carpet to form multi-walled nanotubes with an outer diameter of 20
nanometers and a wall thickness of 3 nm. In this geometry, the stacks of coronene disks are aligned with the length of the tube. The nanotubes have sufficient length to fit between two
platinum nanogap
electrodes produced by
scanning probe nanofabrication and are 180 nm apart. The nanotubes as such are insulating, but, after one-electron
oxidation with
nitrosonium tetrafluoroborate (NOBF
4), they conduct electricity.
[5]
Synthesis
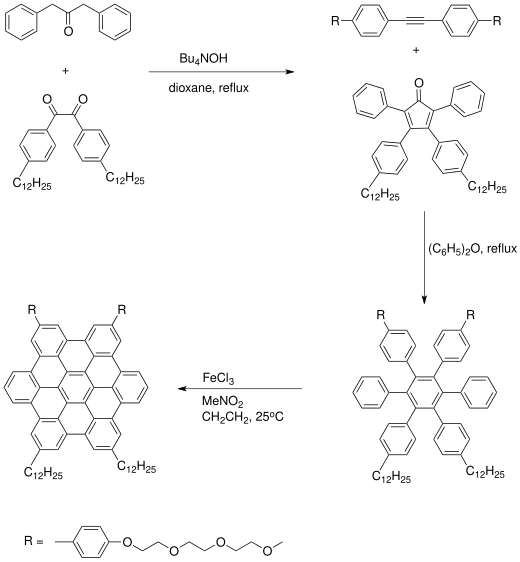
Organic synthesis of a hexabenzocoronene starts with an Aldol condensation reaction of dibenzyl ketone with a benzil derivative to give a substituted cyclopentadienone. A Diels–Alder reaction with alkyne and subsequent expulsion of carbon monoxide gives a hexaphenylbenzene. The adjacent pairs of benzene rings undergo oxidative electrocyclic reactions and aromatization by oxidation by iron(III) chloride in nitromethane.
References
- ^ a b c Goddard, Richard; Haenel, Matthias W.; Herndon, William C.; Krueger, Carl; Zander, Maximilian (1995). "Crystallization of Large Planar Polycyclic Aromatic Hydrocarbons: The Molecular and Crystal Structures of Hexabenzo[bc,ef,hi,kl,no,qr]coronene and Benzo[1,2,3-bc:4,5,6-b'c']dicoronene". Journal of the American Chemical Society. 117: 30–41. doi: 10.1021/ja00106a004.
- ^ Akamatu, Hideo; Kinoshita, Minoru (1959). "On the Magnetic Susceptibility of Hexabenzocoronene and Triphenylene". Bulletin of the Chemical Society of Japan. 32 (7): 773–774. doi: 10.1246/bcsj.32.773.
- ^ Gross, L.; Mohn, F.; Moll, N.; Schuler, B.; Criado, A.; Guitian, E.; Pena, D.; Gourdon, A.; Meyer, G. (2012). "Bond-Order Discrimination by Atomic Force Microscopy". Science. 337 (6100): 1326–9. Bibcode: 2012Sci...337.1326G. doi: 10.1126/science.1225621. PMID 22984067. S2CID 206542919.
- ^ Jonathan P. Hill; Wusong Jin; Atsuko Kosaka; Takanori Fukushima; Hideki Ichihara; Takeshi Shimomura; Kohzo Ito; Tomihiro Hashizume; Noriyuki Ishii; Takuzo Aida (2004). "Self–Assembled Hexa-peri-hexabenzocoronene Graphitic Nanotube". Science. 304 (5676): 1481–1483. Bibcode: 2004Sci...304.1481H. doi: 10.1126/science.1097789. PMID 15178796. S2CID 39674411.
- ^ Sasaki, Tatsuya; Yamada, Yasuhiro; Sato, Satoshi (2018-09-18). "Quantitative Analysis of Zigzag and Armchair Edges on Carbon Materials with and without Pentagons Using Infrared Spectroscopy". Analytical Chemistry. 90 (18): 10724–10731. doi: 10.1021/acs.analchem.8b00949. PMID 30079720. S2CID 51920955.
External links
![]() Media related to
Hexabenzocoronene at Wikimedia Commons
Media related to
Hexabenzocoronene at Wikimedia Commons
| |||

AFM image of hexabenzocoronene
| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
Hexabenzo[bc,ef,hi,kl,no,qr]coronene | |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C42H18 | |||
| Molar mass | 522.606 g·mol−1 | ||
| Appearance | dark yellow [1] | ||
| Density | 1.54 g/cm3 (calc.) [1] | ||
| -346.0·10−6 cm3/mol [2] | |||
| Structure [1] | |||
| monoclinic, P21/a | |||
a = 1.8431(3) nm, b = 0.5119(1) nm, c = 1.2929(2) nm α = 90°, β = 112.57(1)°, γ = 90°
| |||
Formula units (Z)
|
2 | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hexa-peri-hexabenzocoronene (HBC) is a polycyclic aromatic hydrocarbon with the molecular formula C42H18. It consists of a central coronene molecule, with an additional benzene ring fused between each adjacent pair of rings around the periphery. It is sometimes simply called hexabenzocoronene, however, there are other chemicals that share this less-specific name, such as hexa-cata-hexabenzocoronene.
Hexa-peri-hexabenzocoronene has been imaged by atomic force microscopy (AFM) providing the first example of a molecule in which differences in bond order and bond lengths of the individual bonds can be distinguished by a measurement in direct space. [3]
Supramolecular structures
Various hexabenzocoronenes have been investigated in
supramolecular electronics. They are known to
self-assemble into a
columnar phase. One derivative in particular forms
carbon nanotubes with interesting electrical properties.
[4] The columnar phase in this compound further organises itself into sheets, which ultimately roll up like a carpet to form multi-walled nanotubes with an outer diameter of 20
nanometers and a wall thickness of 3 nm. In this geometry, the stacks of coronene disks are aligned with the length of the tube. The nanotubes have sufficient length to fit between two
platinum nanogap
electrodes produced by
scanning probe nanofabrication and are 180 nm apart. The nanotubes as such are insulating, but, after one-electron
oxidation with
nitrosonium tetrafluoroborate (NOBF
4), they conduct electricity.
[5]
Synthesis
Organic synthesis of a hexabenzocoronene starts with an Aldol condensation reaction of dibenzyl ketone with a benzil derivative to give a substituted cyclopentadienone. A Diels–Alder reaction with alkyne and subsequent expulsion of carbon monoxide gives a hexaphenylbenzene. The adjacent pairs of benzene rings undergo oxidative electrocyclic reactions and aromatization by oxidation by iron(III) chloride in nitromethane.
References
- ^ a b c Goddard, Richard; Haenel, Matthias W.; Herndon, William C.; Krueger, Carl; Zander, Maximilian (1995). "Crystallization of Large Planar Polycyclic Aromatic Hydrocarbons: The Molecular and Crystal Structures of Hexabenzo[bc,ef,hi,kl,no,qr]coronene and Benzo[1,2,3-bc:4,5,6-b'c']dicoronene". Journal of the American Chemical Society. 117: 30–41. doi: 10.1021/ja00106a004.
- ^ Akamatu, Hideo; Kinoshita, Minoru (1959). "On the Magnetic Susceptibility of Hexabenzocoronene and Triphenylene". Bulletin of the Chemical Society of Japan. 32 (7): 773–774. doi: 10.1246/bcsj.32.773.
- ^ Gross, L.; Mohn, F.; Moll, N.; Schuler, B.; Criado, A.; Guitian, E.; Pena, D.; Gourdon, A.; Meyer, G. (2012). "Bond-Order Discrimination by Atomic Force Microscopy". Science. 337 (6100): 1326–9. Bibcode: 2012Sci...337.1326G. doi: 10.1126/science.1225621. PMID 22984067. S2CID 206542919.
- ^ Jonathan P. Hill; Wusong Jin; Atsuko Kosaka; Takanori Fukushima; Hideki Ichihara; Takeshi Shimomura; Kohzo Ito; Tomihiro Hashizume; Noriyuki Ishii; Takuzo Aida (2004). "Self–Assembled Hexa-peri-hexabenzocoronene Graphitic Nanotube". Science. 304 (5676): 1481–1483. Bibcode: 2004Sci...304.1481H. doi: 10.1126/science.1097789. PMID 15178796. S2CID 39674411.
- ^ Sasaki, Tatsuya; Yamada, Yasuhiro; Sato, Satoshi (2018-09-18). "Quantitative Analysis of Zigzag and Armchair Edges on Carbon Materials with and without Pentagons Using Infrared Spectroscopy". Analytical Chemistry. 90 (18): 10724–10731. doi: 10.1021/acs.analchem.8b00949. PMID 30079720. S2CID 51920955.
External links
![]() Media related to
Hexabenzocoronene at Wikimedia Commons
Media related to
Hexabenzocoronene at Wikimedia Commons