-
Testosterone levels during the first month of androgen deprivation therapy in men with prostate cancer treated with subcutaneous injections of a GnRH antagonist (degarelix) or agonist (leuprorelin). Doses were 240 then 80 mg/month and 7.5 mg/month, respectively. [26]
-
Testosterone levels in the long-term androgen deprivation therapy of men with prostate cancer by different GnRH agonists administered at 3 month intervals (goserelin, triptorelin and leuprorelin). Dotted line is the threshold for the castrate range. [27]
| Gonadotropin-releasing hormone agonist | |
|---|---|
| Drug class | |

Leuprorelin, one of the most widely used
GnRH agonists. | |
| Class identifiers | |
| Synonyms | GnRH receptor agonists; GnRH blockers; GnRH inhibitors; Antigonadotropins |
| Use | Fertility medicine; Prostate cancer; Breast cancer; Menorrhagia; Endometriosis; Uterine fibroids; Hyperandrogenism; Hirsutism; Precocious puberty; Transgender people; Chemical castration for paraphilias and sex offenders |
| Biological target | GnRH receptor |
| Chemical class | Peptides |
| Legal status | |
| In Wikidata | |
A gonadotropin-releasing hormone agonist (GnRH agonist) is a type of medication which affects gonadotropins and sex hormones. [1] They are used for a variety of indications including in fertility medicine and to lower sex hormone levels in the treatment of hormone-sensitive cancers such as prostate cancer and breast cancer, certain gynecological disorders like heavy periods and endometriosis, high testosterone levels in women, early puberty in children, as a part of transgender hormone therapy, and to delay puberty in transgender youth among other uses. It is also used in the suppression of spontaneous ovulation as part of controlled ovarian hyperstimulation, an essential component in IVF. GnRH agonists are given by injections into fat, as implants placed into fat, and as nasal sprays.
Side effects of GnRH agonists are related to sex hormone deficiency and include symptoms of low testosterone levels and low estrogen levels such as hot flashes, sexual dysfunction, vaginal atrophy, penile atrophy, osteoporosis, infertility, and diminished sex-specific physical characteristics. They are agonists of the GnRH receptor and work by increasing or decreasing the release of gonadotropins and the production of sex hormones by the gonads. When used to suppress gonadotropin release, GnRH agonists can lower sex hormone levels by 95% in both sexes. [2] [3] [4] [5]
GnRH was discovered in 1971, and GnRH analogues were introduced for medical use in the 1980s. [6] [7] Their nonproprietary names usually end in -relin. The most well-known and widely used GnRH analogues are leuprorelin (brand name Lupron) and triptorelin (brand name Decapeptyl). GnRH analogues are available as generic medications. Despite this, they continue to be very expensive.
Medical uses
GnRH agonists are useful in:
- Suppression of spontaneous ovulation as part of controlled ovarian hyperstimulation, which is an essential component in in vitro fertilisation (IVF). Typically, after GnRH agonists have induced a state of hypoestrogenism, exogenous FSH is given to stimulate ovarian follicle, followed by human chorionic gonadotropins (hCG) to trigger oocyte release. GnRH agonists routinely used for this purpose are: buserelin, leuprorelin, nafarelin, and triptorelin. [8]
- Final maturation induction after having performed controlled ovarian hyperstimulation. Usage of GnRH agonist for this purpose necessitates using a GnRH antagonist instead of a GnRH agonist for suppression of spontaneous ovulation, because using GnRH agonist for that purpose as well inactivates the axis for which it is intended to work for final maturation induction.
- Treatment of cancers that are hormonally sensitive and where a hypogonadal state decreases the chances of a recurrence. Thus they are commonly employed in the medical management of prostate cancer and have been used in patients with breast cancer.
- Delaying puberty in individuals with precocious puberty.
- Delaying puberty to delay development of secondary sex characteristics in children with gender dysphoria.
- Management of female disorders that are dependent on estrogen production. Women with menorrhagia, endometriosis, adenomyosis, or uterine fibroids may receive GnRH agonists to suppress ovarian activity and induce a hypoestrogenic state.
- Suppressing sex hormone levels in transgender people, especially transgender women.
- Severe cases of hyperandrogenism, such as in congenital adrenal hyperplasia.
- As part of the pharmacologic treatment of paraphilic disorders in sexual offenders or men with a high risk of sexual offending. [9]
Women of reproductive age who undergo cytotoxic chemotherapy have been pretreated with GnRH agonists to reduce the risk of oocyte loss during such therapy and preserve ovarian function. Further studies are necessary to prove that this approach is useful.
Available forms
| Name | Brand names | Approved uses | Routes | Launch | Hits |
|---|---|---|---|---|---|
| Azagly-nafarelin | Gonazon | Veterinary medicine (assisted reproduction; chemical castration) | Implant; Injection | 2005a | 9,190 |
| Buserelin | Suprefact | Breast cancer; Endometrial hyperplasia; Endometriosis; Female infertility (assisted reproduction); Prostate cancer; Uterine fibroids | Nasal spray; Injection; Implant | 1984 | 253,000 |
| Deslorelin | Ovuplant; Suprelorin | Veterinary medicine (assisted reproduction; chemical castration) | Implant; Injection | 1994 | 85,100 |
| Fertirelin | Ovalyse | Veterinary medicine (assisted reproduction) | Injection | 1981 | 41,000 |
| Gonadorelin | Factrel; Others | Cryptorchidism; Delayed puberty; Diagnostic agent (pituitary disorders); Hypogonadotropic hypogonadism; Veterinary medicine (assisted reproduction) | Injection; Infusion pump; Nasal spray | 1978 | 259,000 |
| Goserelin | Zoladex | Breast cancer; Endometriosis; Female infertility (assisted reproduction); Prostate cancer; Uterine diseases (endometrial thinning agent); Uterine fibroids; Uterine hemorrhage | Implant | 1989 | 400,000 |
| Histrelin | Vantas; Supprelin LA | Precocious puberty; Prostate cancer | Implant | 1993 | 283,000 |
| Lecirelin | Dalmarelin | Veterinary medicine (assisted reproduction) | Injection | 2000a | 19,700 |
| Leuprorelin | Lupron; Eligard; Procren; Prostap; Staladex | Breast cancer; Endometriosis; Menorrhagia; Precocious puberty; Prostate cancer; Uterine fibroids | Injection; Implant | 1985 | 536,000 |
| Nafarelin | Synarel | Precocious puberty; Endometriosis | Nasal spray | 1990 | 117,000 |
| Peforelin | Maprelin | Veterinary medicine (assisted reproduction) | Injection | 2001a | 3,240 |
| Triptorelin | Decapeptyl | Breast cancer; Endometriosis; Female infertility (assisted reproduction); Paraphilias; Precocious puberty; Prostate cancer; Uterine fibroids | Injection | 1986 | 302,000 |
| Notes: Hits = Google Search hits (as of February 2018). Footnotes: a = Launched by this year. | |||||
GnRH agonists that have been marketed and are available for medical use include buserelin, gonadorelin, goserelin, histrelin, leuprorelin, nafarelin, and triptorelin. GnRH agonists that are used mostly or exclusively in veterinary medicine include deslorelin and fertirelin. GnRH agonists can be administered by injection, by implant, or intranasally as a nasal spray. Injectables have been formulated for daily, monthly, and quarterly use, and implants are available that can last from one month to a year. With the exception of gonadorelin, which is used as a progonadotropin, all approved GnRH agonists are used as antigonadotropins.
The clinically used desensitizing GnRH agonists are available in the following pharmaceutical formulations: [10] [11] [12] [13]
- Short-acting injection (once per day): buserelin, histrelin, leuprorelin, triptorelin
- Long-acting depot injection or injected pellet (once every one to six months): leuprorelin, triptorelin
- Injected implant (once every one to three months): buserelin, goserelin, leuprorelin
- Surgically implanted pellet (once per year): histrelin, leuprorelin
- Nasal spray (two to three times per day): buserelin, nafarelin
Contraindications
GnRH agonists are pregnancy category X drugs.
Side effects
Common side effects of the GnRH agonists and antagonists include symptoms of hypogonadism such as hot flashes, gynecomastia, fatigue, weight gain, fluid retention, erectile dysfunction and decreased libido. Long term therapy can result in metabolic abnormalities, weight gain, worsening of diabetes and osteoporosis. Rare, but potentially serious adverse events include transient worsening of prostate cancer due to surge in testosterone with initial injection of GnRH agonists and pituitary apoplexy in patients with pituitary adenoma. Single instances of clinically apparent liver injury have been reported with some GnRH agonists (histrelin, goserelin), but the reports were not very convincing. There is no evidence to indicate that there is cross sensitivity to liver injury among the various GnRH analogues despite their similarity in structure. [14] There is also a report that GnRH agonists used in the treatment of advanced prostate cancer may increase the risk of heart problems by 30%. [15]
Pharmacology
GnRH agonists act as agonists of the GnRH receptor, the biological target of gonadotropin-releasing hormone (GnRH). These drugs can be both peptides and small-molecules. They are modeled after the hypothalamic neurohormone GnRH, which interacts with the GnRH receptor to elicit its biologic response, the release of the pituitary hormones follicle-stimulating hormone (FSH) and luteinizing hormone (LH). However, after the initial "flare" response, continued stimulation with GnRH agonists desensitizes the pituitary gland (by causing GnRH receptor downregulation) to GnRH. Pituitary desensitization reduces the secretion of LH and FSH and thus induces a state of hypogonadotropic hypogonadal anovulation, sometimes referred to as "pseudomenopause" or "medical oophorectomy". [1] GnRH agonists are able to completely shutdown gonadal testosterone production and thereby suppress circulating testosterone levels by 95% or into the castrate/female range in men. [5]
Agonists do not quickly dissociate from the GnRH receptor. As a result, initially there is an increase in FSH and LH secretion (so-called "flare effect"). Levels of LH may increase by up to 10-fold, [16] [17] while levels of testosterone generally increase to 140 to 200% of baseline values. [18] However, after continuous administration, a profound hypogonadal effect (i.e. decrease in FSH and LH) is achieved through receptor downregulation by internalization of receptors. [16] Generally this induced and reversible hypogonadism is the therapeutic goal. During the flare, peak levels of testosterone occur after 2 to 4 days, baseline testosterone levels are returned to by 7 to 8 days, and castrate levels of testosterone are achieved by two to four weeks. [18] [16] A 7 day study of infertile women found that restoration of normal gonadotropin secretion takes 5 to 8 days after cessation of exogenous GnRH agonists. [19]
Various medications can be used to prevent the testosterone flare and/or its effects at the initiation of GnRH agonist therapy. [17] [20] [21] These include antigonadotropins such as progestogens like cyproterone acetate and chlormadinone acetate and estrogens like diethylstilbestrol, fosfestrol (diethylstilbestrol diphosphate), and estramustine phosphate; antiandrogens such as nonsteroidal antiandrogens like flutamide, nilutamide, and bicalutamide; and androgen synthesis inhibitors such as ketoconazole and abiraterone acetate. [17] [20] [21] [22] [23] [24] [25]
Chemistry
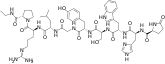
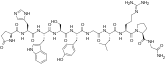
GnRH agonists are synthetically modeled after the natural GnRH decapeptide with specific modifications, usually double and single substitutions and typically in position 6 (amino acid substitution), 9 (alkylation) and 10 (deletion). These substitutions inhibit rapid degradation. Agonists with two substitutions include: leuprorelin, buserelin, histrelin, goserelin, and deslorelin. The agents nafarelin and triptorelin are agonists with single substitutions at position 6.
Veterinary uses
GnRH analogues are also used in veterinary medicine. Uses include:
- Temporary suppression of fertility in female dogs
- Induction of ovulation in mares
See also
References
- ^ a b Magon N (October 2011). "Gonadotropin releasing hormone agonists: Expanding vistas". Indian Journal of Endocrinology and Metabolism. 15 (4): 261–7. doi: 10.4103/2230-8210.85575. PMC 3193774. PMID 22028996.
- ^ Hemat RA (2 March 2003). Andropathy. Urotext. pp. 120–. ISBN 978-1-903737-08-8.
- ^ Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 973–. ISBN 978-0-7817-1750-2.
- ^ Corson SL, Derman RJ (15 December 1995). Fertility Control. CRC Press. pp. 249–250. ISBN 978-0-9697978-0-7.
- ^ a b Novara G, Galfano A, Secco S, Ficarra V, Artibani W (2009). "Impact of surgical and medical castration on serum testosterone level in prostate cancer patients". Urologia Internationalis. 82 (3): 249–55. doi: 10.1159/000209352. PMID 19440008.
- ^ Gardner DK, Simón C (26 June 2017). Handbook of In Vitro Fertilization (Fourth ed.). CRC Press. pp. 131–. ISBN 978-1-4987-2947-5.
- ^ Jameson JL, De Groot LJ (25 February 2015). Endocrinology: Adult and Pediatric E-Book. Elsevier Health Sciences. pp. 2135–. ISBN 978-0-323-32195-2.
- ^ van Loenen AC, Huirne JA, Schats R, Hompes PG, Lambalk CB (November 2002). "GnRH agonists, antagonists, and assisted conception". Seminars in Reproductive Medicine. 20 (4): 349–64. doi: 10.1055/s-2002-36713. PMID 12536358. S2CID 45436435.
- ^ Turner D, Briken P (January 2018). "Treatment of Paraphilic Disorders in Sexual Offenders or Men With a Risk of Sexual Offending With Luteinizing Hormone-Releasing Hormone Agonists: An Updated Systematic Review". The Journal of Sexual Medicine. 15 (1): 77–93. doi: 10.1016/j.jsxm.2017.11.013. PMID 29289377.
- ^ Richard A. Lehne, Laura Rosenthal (25 June 2014). Pharmacology for Nursing Care - E-Book. Elsevier Health Sciences. pp. 1296–. ISBN 978-0-323-29354-9.
- ^ James L. Gulley (20 December 2011). Prostate Cancer. Demos Medical Publishing. pp. 503–. ISBN 978-1-936287-46-8.
- ^ Charles G. D. Brook, Peter Clayton, Rosalind Brown (22 September 2011). Brook's Clinical Pediatric Endocrinology. John Wiley & Sons. pp. 242–. ISBN 978-1-4443-1673-5.
- ^ Surveen Ghumman (22 September 2015). Principles and Practice of Controlled Ovarian Stimulation in ART. Springer. pp. 96–. ISBN 978-81-322-1686-5.
- ^ LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Gonadotropin Releasing Hormone (GnRH) Analogues. [Updated 2018 Mar 20]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547863/
- ^ "Researchers Suggest Hormone Therapy for Prostate Cancer Can Cause Serious Heart Problems and Death". Genetic Engineering & Biotechnology News. 22 September 2009.
- ^ a b c Wein AJ, Kavoussi LR, Novick AC, Partin AW, Peters CA (25 August 2011). Campbell-Walsh Urology: Expert Consult Premium Edition: Enhanced Online Features and Print, 4-Volume Set. Elsevier Health Sciences. pp. 2939–. ISBN 978-1-4160-6911-9.
- ^ a b c Thompson IM (2001). "Flare Associated with LHRH-Agonist Therapy". Rev Urol. 3 (Suppl 3): S10–4. PMC 1476081. PMID 16986003.
- ^
a
b Krakowsky Y, Morgentaler A (July 2017). "Risk of Testosterone Flare in the Era of the Saturation Model: One More Historical Myth". Eur Urol Focus. 5 (1): 81–89.
doi:
10.1016/j.euf.2017.06.008.
PMID
28753828.
S2CID
10011200.
Initial administration of LHRH agonists reliably causes a transient rise in serum T, with peak T values observed at 2–4 d followed by a reduction to baseline values by 7–8 d, and achievement of castrate levels by 2–4 wk [10]. Most studies demonstrate an increase in peak serum T concentrations by 40–100% above baseline during T flare.
- ^ Cedrin-Durnerin I, Bidart JM, Robert P, Wolf JP, Uzan M, Hugues JN (May 2000). "Consequences on gonadotrophin secretion of an early discontinuation of gonadotrophin-releasing hormone agonist administration in short-term protocol for in-vitro fertilization". Human Reproduction. 15 (5): 1009–14. doi: 10.1093/humrep/15.5.1009. PMID 10783343.
- ^ a b Scaletscky R, Smith JA (April 1993). "Disease flare with gonadotrophin-releasing hormone (GnRH) analogues. How serious is it?". Drug Saf. 8 (4): 265–70. doi: 10.2165/00002018-199308040-00001. PMID 8481213. S2CID 36964191.
- ^ a b Vis AN, van der Sluis TM, Al-Itejawi HH, van Moorselaar RJ, Meuleman EJ (January 2015). "Risk of disease flare with LHRH agonist therapy in men with prostate cancer: myth or fact?". Urol. Oncol. 33 (1): 7–15. doi: 10.1016/j.urolonc.2014.04.016. PMID 25159013.
- ^ Kotake T, Usami M, Akaza H, Koiso K, Homma Y, Kawabe K, Aso Y, Orikasa S, Shimazaki J, Isaka S, Yoshida O, Hirao Y, Okajima E, Naito S, Kumazawa J, Kanetake H, Saito Y, Ohi Y, Ohashi Y (November 1999). "Goserelin acetate with or without antiandrogen or estrogen in the treatment of patients with advanced prostate cancer: a multicenter, randomized, controlled trial in Japan. Zoladex Study Group". Jpn. J. Clin. Oncol. 29 (11): 562–70. doi: 10.1093/jjco/29.11.562. PMID 10678560.
- ^ Shimizu TS, Shibata Y, Jinbo H, Satoh J, Yamanaka H (1995). "Estramustine phosphate for preventing flare-up in luteinizing hormone-releasing hormone analogue depot therapy". Eur. Urol. 27 (3): 192–5. doi: 10.1159/000475159. PMID 7541359.
- ^ Sugiono M, Winkler MH, Okeke AA, Benney M, Gillatt DA (2005). "Bicalutamide vs cyproterone acetate in preventing flare with LHRH analogue therapy for prostate cancer--a pilot study". Prostate Cancer Prostatic Dis. 8 (1): 91–4. doi: 10.1038/sj.pcan.4500784. PMID 15711607. S2CID 9151853.
- ^ Pokuri VK, Nourkeyhani H, Betsy B, Herbst L, Sikorski M, Spangenthal E, Fabiano A, George S (July 2015). "Strategies to Circumvent Testosterone Surge and Disease Flare in Advanced Prostate Cancer: Emerging Treatment Paradigms". J Natl Compr Canc Netw. 13 (7): e49–55. doi: 10.6004/jnccn.2015.0109. PMID 26150586.
- ^ Klotz L, Boccon-Gibod L, Shore ND, Andreou C, Persson BE, Cantor P, Jensen JK, Olesen TK, Schröder FH (December 2008). "The efficacy and safety of degarelix: a 12-month, comparative, randomized, open-label, parallel-group phase III study in patients with prostate cancer". BJU Int. 102 (11): 1531–8. doi: 10.1111/j.1464-410X.2008.08183.x. PMID 19035858.
- ^ Shim M, Bang WJ, Oh CY, Lee YS, Cho JS (July 2019). "Effectiveness of three different luteinizing hormone-releasing hormone agonists in the chemical castration of patients with prostate cancer: Goserelin versus triptorelin versus leuprolide". Investig Clin Urol. 60 (4): 244–250. doi: 10.4111/icu.2019.60.4.244. PMC 6607074. PMID 31294133.
External links
| Gonadotropin-releasing hormone agonist | |
|---|---|
| Drug class | |

Leuprorelin, one of the most widely used
GnRH agonists. | |
| Class identifiers | |
| Synonyms | GnRH receptor agonists; GnRH blockers; GnRH inhibitors; Antigonadotropins |
| Use | Fertility medicine; Prostate cancer; Breast cancer; Menorrhagia; Endometriosis; Uterine fibroids; Hyperandrogenism; Hirsutism; Precocious puberty; Transgender people; Chemical castration for paraphilias and sex offenders |
| Biological target | GnRH receptor |
| Chemical class | Peptides |
| Legal status | |
| In Wikidata | |
A gonadotropin-releasing hormone agonist (GnRH agonist) is a type of medication which affects gonadotropins and sex hormones. [1] They are used for a variety of indications including in fertility medicine and to lower sex hormone levels in the treatment of hormone-sensitive cancers such as prostate cancer and breast cancer, certain gynecological disorders like heavy periods and endometriosis, high testosterone levels in women, early puberty in children, as a part of transgender hormone therapy, and to delay puberty in transgender youth among other uses. It is also used in the suppression of spontaneous ovulation as part of controlled ovarian hyperstimulation, an essential component in IVF. GnRH agonists are given by injections into fat, as implants placed into fat, and as nasal sprays.
Side effects of GnRH agonists are related to sex hormone deficiency and include symptoms of low testosterone levels and low estrogen levels such as hot flashes, sexual dysfunction, vaginal atrophy, penile atrophy, osteoporosis, infertility, and diminished sex-specific physical characteristics. They are agonists of the GnRH receptor and work by increasing or decreasing the release of gonadotropins and the production of sex hormones by the gonads. When used to suppress gonadotropin release, GnRH agonists can lower sex hormone levels by 95% in both sexes. [2] [3] [4] [5]
GnRH was discovered in 1971, and GnRH analogues were introduced for medical use in the 1980s. [6] [7] Their nonproprietary names usually end in -relin. The most well-known and widely used GnRH analogues are leuprorelin (brand name Lupron) and triptorelin (brand name Decapeptyl). GnRH analogues are available as generic medications. Despite this, they continue to be very expensive.
Medical uses
GnRH agonists are useful in:
- Suppression of spontaneous ovulation as part of controlled ovarian hyperstimulation, which is an essential component in in vitro fertilisation (IVF). Typically, after GnRH agonists have induced a state of hypoestrogenism, exogenous FSH is given to stimulate ovarian follicle, followed by human chorionic gonadotropins (hCG) to trigger oocyte release. GnRH agonists routinely used for this purpose are: buserelin, leuprorelin, nafarelin, and triptorelin. [8]
- Final maturation induction after having performed controlled ovarian hyperstimulation. Usage of GnRH agonist for this purpose necessitates using a GnRH antagonist instead of a GnRH agonist for suppression of spontaneous ovulation, because using GnRH agonist for that purpose as well inactivates the axis for which it is intended to work for final maturation induction.
- Treatment of cancers that are hormonally sensitive and where a hypogonadal state decreases the chances of a recurrence. Thus they are commonly employed in the medical management of prostate cancer and have been used in patients with breast cancer.
- Delaying puberty in individuals with precocious puberty.
- Delaying puberty to delay development of secondary sex characteristics in children with gender dysphoria.
- Management of female disorders that are dependent on estrogen production. Women with menorrhagia, endometriosis, adenomyosis, or uterine fibroids may receive GnRH agonists to suppress ovarian activity and induce a hypoestrogenic state.
- Suppressing sex hormone levels in transgender people, especially transgender women.
- Severe cases of hyperandrogenism, such as in congenital adrenal hyperplasia.
- As part of the pharmacologic treatment of paraphilic disorders in sexual offenders or men with a high risk of sexual offending. [9]
Women of reproductive age who undergo cytotoxic chemotherapy have been pretreated with GnRH agonists to reduce the risk of oocyte loss during such therapy and preserve ovarian function. Further studies are necessary to prove that this approach is useful.
Available forms
| Name | Brand names | Approved uses | Routes | Launch | Hits |
|---|---|---|---|---|---|
| Azagly-nafarelin | Gonazon | Veterinary medicine (assisted reproduction; chemical castration) | Implant; Injection | 2005a | 9,190 |
| Buserelin | Suprefact | Breast cancer; Endometrial hyperplasia; Endometriosis; Female infertility (assisted reproduction); Prostate cancer; Uterine fibroids | Nasal spray; Injection; Implant | 1984 | 253,000 |
| Deslorelin | Ovuplant; Suprelorin | Veterinary medicine (assisted reproduction; chemical castration) | Implant; Injection | 1994 | 85,100 |
| Fertirelin | Ovalyse | Veterinary medicine (assisted reproduction) | Injection | 1981 | 41,000 |
| Gonadorelin | Factrel; Others | Cryptorchidism; Delayed puberty; Diagnostic agent (pituitary disorders); Hypogonadotropic hypogonadism; Veterinary medicine (assisted reproduction) | Injection; Infusion pump; Nasal spray | 1978 | 259,000 |
| Goserelin | Zoladex | Breast cancer; Endometriosis; Female infertility (assisted reproduction); Prostate cancer; Uterine diseases (endometrial thinning agent); Uterine fibroids; Uterine hemorrhage | Implant | 1989 | 400,000 |
| Histrelin | Vantas; Supprelin LA | Precocious puberty; Prostate cancer | Implant | 1993 | 283,000 |
| Lecirelin | Dalmarelin | Veterinary medicine (assisted reproduction) | Injection | 2000a | 19,700 |
| Leuprorelin | Lupron; Eligard; Procren; Prostap; Staladex | Breast cancer; Endometriosis; Menorrhagia; Precocious puberty; Prostate cancer; Uterine fibroids | Injection; Implant | 1985 | 536,000 |
| Nafarelin | Synarel | Precocious puberty; Endometriosis | Nasal spray | 1990 | 117,000 |
| Peforelin | Maprelin | Veterinary medicine (assisted reproduction) | Injection | 2001a | 3,240 |
| Triptorelin | Decapeptyl | Breast cancer; Endometriosis; Female infertility (assisted reproduction); Paraphilias; Precocious puberty; Prostate cancer; Uterine fibroids | Injection | 1986 | 302,000 |
| Notes: Hits = Google Search hits (as of February 2018). Footnotes: a = Launched by this year. | |||||
GnRH agonists that have been marketed and are available for medical use include buserelin, gonadorelin, goserelin, histrelin, leuprorelin, nafarelin, and triptorelin. GnRH agonists that are used mostly or exclusively in veterinary medicine include deslorelin and fertirelin. GnRH agonists can be administered by injection, by implant, or intranasally as a nasal spray. Injectables have been formulated for daily, monthly, and quarterly use, and implants are available that can last from one month to a year. With the exception of gonadorelin, which is used as a progonadotropin, all approved GnRH agonists are used as antigonadotropins.
The clinically used desensitizing GnRH agonists are available in the following pharmaceutical formulations: [10] [11] [12] [13]
- Short-acting injection (once per day): buserelin, histrelin, leuprorelin, triptorelin
- Long-acting depot injection or injected pellet (once every one to six months): leuprorelin, triptorelin
- Injected implant (once every one to three months): buserelin, goserelin, leuprorelin
- Surgically implanted pellet (once per year): histrelin, leuprorelin
- Nasal spray (two to three times per day): buserelin, nafarelin
Contraindications
GnRH agonists are pregnancy category X drugs.
Side effects
Common side effects of the GnRH agonists and antagonists include symptoms of hypogonadism such as hot flashes, gynecomastia, fatigue, weight gain, fluid retention, erectile dysfunction and decreased libido. Long term therapy can result in metabolic abnormalities, weight gain, worsening of diabetes and osteoporosis. Rare, but potentially serious adverse events include transient worsening of prostate cancer due to surge in testosterone with initial injection of GnRH agonists and pituitary apoplexy in patients with pituitary adenoma. Single instances of clinically apparent liver injury have been reported with some GnRH agonists (histrelin, goserelin), but the reports were not very convincing. There is no evidence to indicate that there is cross sensitivity to liver injury among the various GnRH analogues despite their similarity in structure. [14] There is also a report that GnRH agonists used in the treatment of advanced prostate cancer may increase the risk of heart problems by 30%. [15]
Pharmacology
GnRH agonists act as agonists of the GnRH receptor, the biological target of gonadotropin-releasing hormone (GnRH). These drugs can be both peptides and small-molecules. They are modeled after the hypothalamic neurohormone GnRH, which interacts with the GnRH receptor to elicit its biologic response, the release of the pituitary hormones follicle-stimulating hormone (FSH) and luteinizing hormone (LH). However, after the initial "flare" response, continued stimulation with GnRH agonists desensitizes the pituitary gland (by causing GnRH receptor downregulation) to GnRH. Pituitary desensitization reduces the secretion of LH and FSH and thus induces a state of hypogonadotropic hypogonadal anovulation, sometimes referred to as "pseudomenopause" or "medical oophorectomy". [1] GnRH agonists are able to completely shutdown gonadal testosterone production and thereby suppress circulating testosterone levels by 95% or into the castrate/female range in men. [5]
Agonists do not quickly dissociate from the GnRH receptor. As a result, initially there is an increase in FSH and LH secretion (so-called "flare effect"). Levels of LH may increase by up to 10-fold, [16] [17] while levels of testosterone generally increase to 140 to 200% of baseline values. [18] However, after continuous administration, a profound hypogonadal effect (i.e. decrease in FSH and LH) is achieved through receptor downregulation by internalization of receptors. [16] Generally this induced and reversible hypogonadism is the therapeutic goal. During the flare, peak levels of testosterone occur after 2 to 4 days, baseline testosterone levels are returned to by 7 to 8 days, and castrate levels of testosterone are achieved by two to four weeks. [18] [16] A 7 day study of infertile women found that restoration of normal gonadotropin secretion takes 5 to 8 days after cessation of exogenous GnRH agonists. [19]
Various medications can be used to prevent the testosterone flare and/or its effects at the initiation of GnRH agonist therapy. [17] [20] [21] These include antigonadotropins such as progestogens like cyproterone acetate and chlormadinone acetate and estrogens like diethylstilbestrol, fosfestrol (diethylstilbestrol diphosphate), and estramustine phosphate; antiandrogens such as nonsteroidal antiandrogens like flutamide, nilutamide, and bicalutamide; and androgen synthesis inhibitors such as ketoconazole and abiraterone acetate. [17] [20] [21] [22] [23] [24] [25]
-
Testosterone levels during the first month of androgen deprivation therapy in men with prostate cancer treated with subcutaneous injections of a GnRH antagonist (degarelix) or agonist (leuprorelin). Doses were 240 then 80 mg/month and 7.5 mg/month, respectively. [26]
-
Testosterone levels in the long-term androgen deprivation therapy of men with prostate cancer by different GnRH agonists administered at 3 month intervals (goserelin, triptorelin and leuprorelin). Dotted line is the threshold for the castrate range. [27]
Chemistry
GnRH agonists are synthetically modeled after the natural GnRH decapeptide with specific modifications, usually double and single substitutions and typically in position 6 (amino acid substitution), 9 (alkylation) and 10 (deletion). These substitutions inhibit rapid degradation. Agonists with two substitutions include: leuprorelin, buserelin, histrelin, goserelin, and deslorelin. The agents nafarelin and triptorelin are agonists with single substitutions at position 6.
Veterinary uses
GnRH analogues are also used in veterinary medicine. Uses include:
- Temporary suppression of fertility in female dogs
- Induction of ovulation in mares
See also
References
- ^ a b Magon N (October 2011). "Gonadotropin releasing hormone agonists: Expanding vistas". Indian Journal of Endocrinology and Metabolism. 15 (4): 261–7. doi: 10.4103/2230-8210.85575. PMC 3193774. PMID 22028996.
- ^ Hemat RA (2 March 2003). Andropathy. Urotext. pp. 120–. ISBN 978-1-903737-08-8.
- ^ Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 973–. ISBN 978-0-7817-1750-2.
- ^ Corson SL, Derman RJ (15 December 1995). Fertility Control. CRC Press. pp. 249–250. ISBN 978-0-9697978-0-7.
- ^ a b Novara G, Galfano A, Secco S, Ficarra V, Artibani W (2009). "Impact of surgical and medical castration on serum testosterone level in prostate cancer patients". Urologia Internationalis. 82 (3): 249–55. doi: 10.1159/000209352. PMID 19440008.
- ^ Gardner DK, Simón C (26 June 2017). Handbook of In Vitro Fertilization (Fourth ed.). CRC Press. pp. 131–. ISBN 978-1-4987-2947-5.
- ^ Jameson JL, De Groot LJ (25 February 2015). Endocrinology: Adult and Pediatric E-Book. Elsevier Health Sciences. pp. 2135–. ISBN 978-0-323-32195-2.
- ^ van Loenen AC, Huirne JA, Schats R, Hompes PG, Lambalk CB (November 2002). "GnRH agonists, antagonists, and assisted conception". Seminars in Reproductive Medicine. 20 (4): 349–64. doi: 10.1055/s-2002-36713. PMID 12536358. S2CID 45436435.
- ^ Turner D, Briken P (January 2018). "Treatment of Paraphilic Disorders in Sexual Offenders or Men With a Risk of Sexual Offending With Luteinizing Hormone-Releasing Hormone Agonists: An Updated Systematic Review". The Journal of Sexual Medicine. 15 (1): 77–93. doi: 10.1016/j.jsxm.2017.11.013. PMID 29289377.
- ^ Richard A. Lehne, Laura Rosenthal (25 June 2014). Pharmacology for Nursing Care - E-Book. Elsevier Health Sciences. pp. 1296–. ISBN 978-0-323-29354-9.
- ^ James L. Gulley (20 December 2011). Prostate Cancer. Demos Medical Publishing. pp. 503–. ISBN 978-1-936287-46-8.
- ^ Charles G. D. Brook, Peter Clayton, Rosalind Brown (22 September 2011). Brook's Clinical Pediatric Endocrinology. John Wiley & Sons. pp. 242–. ISBN 978-1-4443-1673-5.
- ^ Surveen Ghumman (22 September 2015). Principles and Practice of Controlled Ovarian Stimulation in ART. Springer. pp. 96–. ISBN 978-81-322-1686-5.
- ^ LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Gonadotropin Releasing Hormone (GnRH) Analogues. [Updated 2018 Mar 20]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547863/
- ^ "Researchers Suggest Hormone Therapy for Prostate Cancer Can Cause Serious Heart Problems and Death". Genetic Engineering & Biotechnology News. 22 September 2009.
- ^ a b c Wein AJ, Kavoussi LR, Novick AC, Partin AW, Peters CA (25 August 2011). Campbell-Walsh Urology: Expert Consult Premium Edition: Enhanced Online Features and Print, 4-Volume Set. Elsevier Health Sciences. pp. 2939–. ISBN 978-1-4160-6911-9.
- ^ a b c Thompson IM (2001). "Flare Associated with LHRH-Agonist Therapy". Rev Urol. 3 (Suppl 3): S10–4. PMC 1476081. PMID 16986003.
- ^
a
b Krakowsky Y, Morgentaler A (July 2017). "Risk of Testosterone Flare in the Era of the Saturation Model: One More Historical Myth". Eur Urol Focus. 5 (1): 81–89.
doi:
10.1016/j.euf.2017.06.008.
PMID
28753828.
S2CID
10011200.
Initial administration of LHRH agonists reliably causes a transient rise in serum T, with peak T values observed at 2–4 d followed by a reduction to baseline values by 7–8 d, and achievement of castrate levels by 2–4 wk [10]. Most studies demonstrate an increase in peak serum T concentrations by 40–100% above baseline during T flare.
- ^ Cedrin-Durnerin I, Bidart JM, Robert P, Wolf JP, Uzan M, Hugues JN (May 2000). "Consequences on gonadotrophin secretion of an early discontinuation of gonadotrophin-releasing hormone agonist administration in short-term protocol for in-vitro fertilization". Human Reproduction. 15 (5): 1009–14. doi: 10.1093/humrep/15.5.1009. PMID 10783343.
- ^ a b Scaletscky R, Smith JA (April 1993). "Disease flare with gonadotrophin-releasing hormone (GnRH) analogues. How serious is it?". Drug Saf. 8 (4): 265–70. doi: 10.2165/00002018-199308040-00001. PMID 8481213. S2CID 36964191.
- ^ a b Vis AN, van der Sluis TM, Al-Itejawi HH, van Moorselaar RJ, Meuleman EJ (January 2015). "Risk of disease flare with LHRH agonist therapy in men with prostate cancer: myth or fact?". Urol. Oncol. 33 (1): 7–15. doi: 10.1016/j.urolonc.2014.04.016. PMID 25159013.
- ^ Kotake T, Usami M, Akaza H, Koiso K, Homma Y, Kawabe K, Aso Y, Orikasa S, Shimazaki J, Isaka S, Yoshida O, Hirao Y, Okajima E, Naito S, Kumazawa J, Kanetake H, Saito Y, Ohi Y, Ohashi Y (November 1999). "Goserelin acetate with or without antiandrogen or estrogen in the treatment of patients with advanced prostate cancer: a multicenter, randomized, controlled trial in Japan. Zoladex Study Group". Jpn. J. Clin. Oncol. 29 (11): 562–70. doi: 10.1093/jjco/29.11.562. PMID 10678560.
- ^ Shimizu TS, Shibata Y, Jinbo H, Satoh J, Yamanaka H (1995). "Estramustine phosphate for preventing flare-up in luteinizing hormone-releasing hormone analogue depot therapy". Eur. Urol. 27 (3): 192–5. doi: 10.1159/000475159. PMID 7541359.
- ^ Sugiono M, Winkler MH, Okeke AA, Benney M, Gillatt DA (2005). "Bicalutamide vs cyproterone acetate in preventing flare with LHRH analogue therapy for prostate cancer--a pilot study". Prostate Cancer Prostatic Dis. 8 (1): 91–4. doi: 10.1038/sj.pcan.4500784. PMID 15711607. S2CID 9151853.
- ^ Pokuri VK, Nourkeyhani H, Betsy B, Herbst L, Sikorski M, Spangenthal E, Fabiano A, George S (July 2015). "Strategies to Circumvent Testosterone Surge and Disease Flare in Advanced Prostate Cancer: Emerging Treatment Paradigms". J Natl Compr Canc Netw. 13 (7): e49–55. doi: 10.6004/jnccn.2015.0109. PMID 26150586.
- ^ Klotz L, Boccon-Gibod L, Shore ND, Andreou C, Persson BE, Cantor P, Jensen JK, Olesen TK, Schröder FH (December 2008). "The efficacy and safety of degarelix: a 12-month, comparative, randomized, open-label, parallel-group phase III study in patients with prostate cancer". BJU Int. 102 (11): 1531–8. doi: 10.1111/j.1464-410X.2008.08183.x. PMID 19035858.
- ^ Shim M, Bang WJ, Oh CY, Lee YS, Cho JS (July 2019). "Effectiveness of three different luteinizing hormone-releasing hormone agonists in the chemical castration of patients with prostate cancer: Goserelin versus triptorelin versus leuprolide". Investig Clin Urol. 60 (4): 244–250. doi: 10.4111/icu.2019.60.4.244. PMC 6607074. PMID 31294133.
![Testosterone levels during the first month of androgen deprivation therapy in men with prostate cancer treated with subcutaneous injections of a GnRH antagonist (degarelix) or agonist (leuprorelin). Doses were 240 then 80 mg/month and 7.5 mg/month, respectively.[26]](https://upload.wikimedia.org/wikipedia/commons/thumb/f/f8/Testosterone_levels_in_men_with_prostate_cancer_treated_with_GnRH_agonists_and_antagonists.png/300px-Testosterone_levels_in_men_with_prostate_cancer_treated_with_GnRH_agonists_and_antagonists.png)
![Testosterone levels in the long-term androgen deprivation therapy of men with prostate cancer by different GnRH agonists administered at 3 month intervals (goserelin, triptorelin and leuprorelin). Dotted line is the threshold for the castrate range.[27]](https://upload.wikimedia.org/wikipedia/commons/thumb/3/39/Testosterone_levels_in_long-term_androgen_deprivation_therapy_with_different_GnRH_agonists.png/300px-Testosterone_levels_in_long-term_androgen_deprivation_therapy_with_different_GnRH_agonists.png)