
| |

| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.636 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| Properties | |
| C18H15AuClP | |
| Molar mass | 494.71 g·mol−1 |
| Appearance | Colorless solid |
| Density | 1.97 g/cm3 |
| Melting point | 236–237 °C (457–459 °F; 509–510 K) |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Chloro(triphenylphosphine)gold(I) or triphenylphosphinegold(I) chloride is a coordination complex with the formula ( Ph3P)AuCl. This colorless solid is a common reagent for research on gold compounds.
Preparation and structure
The complex is prepared by reducing chloroauric acid with triphenylphosphine in 95% ethanol: [1] [2]
- HAuCl4 + H2O + 2 PPh3 → (Ph3P)AuCl + Ph3PO + 3 HCl
Ph3PAuCl can also be prepared by treating a thioether complex of gold like (dimethyl sulfide)gold(I) chloride, [(Me2S)AuCl], with triphenylphosphine. [3]
The complex adopts a linear coordination geometry, which is typical of most gold(I) compounds. [4] It crystallizes in the orthorhombic space group P212121 with a = 12.300(4) Å, b = 13.084(4) Å, c = 10.170(3) Å with Z = 4 formula units per unit cell. [5]
Reactivity
Triphenylphosphinegold(I) chloride is a popular stable precursor for a cationic gold(I) catalyst used in organic synthesis. [3] Typically, it is treated with silver(I) salts of weakly coordinating anions (e.g., X– = SbF6–, BF4–, TfO–, or Tf2N–) to generate a weakly bound Ph3PAu–X complex, in equilibrium with the catalytically-active species [Ph3PAu]+X– in solution. Among these, only the bistriflimide complex Ph3PAuNTf2 can be isolated as the pure compound. [6] The nitrate complex Ph3PAuONO2 and the oxonium species [(Ph3PAu)3O]+[BF4– are also prepared from the chloride. [7]
As shown in the scheme below, the methyl complex Ph3PAuMe is prepared from triphenylphosphinegold(I) chloride by transmetalation with a Grignard reagent. Further treatment of Ph3PAuMe with methyllithium displaces the phosphine ligand and generates lithium di- and tetramethylaurate, Li+[AuMe2– and Li+[AuMe4–, respectively. [8] [9]
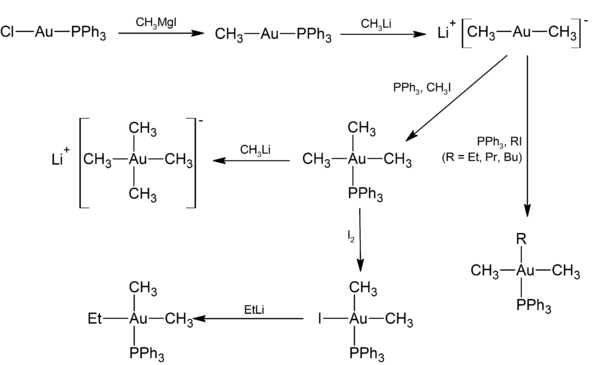
References
- ^ Pierre Braunstein; Hans Lehner; Dominique Matt (1990). A Platinum-Gold Cluster: Chloro-1κCl-Bis(Triethylphosphine-1κP)Bis(Triphenylphosphine)-2κP, 3κP-Triangulo- Digold-Platinum(1 +) Trifluoromethanesulfonate. Inorganic Syntheses. Vol. 27. pp. 218–221. doi: 10.1002/9780470132586.ch42.
- ^ M. I. Bruce; B. K. Nicholson; O. Bin Shawkataly (1989). "Synthesis of Gold-Containing Mixed-Metal Cluster Complexes". Inorganic Syntheses. Vol. 26. pp. 324–328. doi: 10.1002/9780470132579.ch59. ISBN 9780470132579.
- ^ a b Gorin, David J.; Sherry, Benjamin D.; Toste, F. Dean (2008), "Triphenylphosphinegold(I) chloride", Encyclopedia of Reagents for Organic Synthesis, American Cancer Society, doi: 10.1002/047084289x.rn00803, ISBN 9780470842898
- ^ Baenziger, N. C.; Bennett, W. E.; Soborofe, D. M. (1976). "Chloro(triphenylphosphine)gold(I)". Acta Crystallographica Section B. 32 (3): 962. doi: 10.1107/S0567740876004330.
- ^ Borissova, Alexandra O.; Korlyukov, Alexander A.; Antipin, Mikhail Yu.; Lyssenko, Konstantin A. (2008). "Estimation of Dissociation Energy in Donor−Acceptor Complex AuCl·PPh3via Topological Analysis of the Experimental Electron Density Distribution Function". The Journal of Physical Chemistry A. 112 (46): 11519–22. doi: 10.1021/jp807258d. PMID 18959385.
- ^ Mézailles, Nicolas; Ricard, Louis; Gagosz, Fabien (2005-09-01). "Phosphine Gold(I) Bis-(trifluoromethanesulfonyl)imidate Complexes as New Highly Efficient and Air-Stable Catalysts for the Cycloisomerization of Enynes". Organic Letters. 7 (19): 4133–4136. doi: 10.1021/ol0515917. ISSN 1523-7060. PMID 16146370.
- ^ A. M. Mueting, B. D. Alexander, P. D. Boyle, A. L. Casalnuovo, L. N. Ito, B. J. Johnson, L. H. Pignolet "Mixed-Metal-Gold Phosphine Cluster Compounds" Inorganic Syntheses, 1992, Volume 29, Pages 279–298, 2007. doi: 10.1002/9780470132609.ch63
- ^ Rice, Gary W.; Tobias, R. Stuart (1976). "Isolation of thermally stable compounds containing the dimethylaurate(I) and tetramethylaurate(III) anions". Inorganic Chemistry. 15 (2): 489–490. doi: 10.1021/ic50156a058. ISSN 0020-1669.
- ^ Zhu, Dunming; Lindeman, Sergey V.; Kochi, Jay K. (1999). "X-ray Crystal Structures and the Facile Oxidative (Au−C) Cleavage of the Dimethylaurate(I) and Tetramethylaurate(III) Homologues". Organometallics. 18 (11): 2241–2248. doi: 10.1021/om990043s. ISSN 0276-7333.

| |

| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.636 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| Properties | |
| C18H15AuClP | |
| Molar mass | 494.71 g·mol−1 |
| Appearance | Colorless solid |
| Density | 1.97 g/cm3 |
| Melting point | 236–237 °C (457–459 °F; 509–510 K) |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Chloro(triphenylphosphine)gold(I) or triphenylphosphinegold(I) chloride is a coordination complex with the formula ( Ph3P)AuCl. This colorless solid is a common reagent for research on gold compounds.
Preparation and structure
The complex is prepared by reducing chloroauric acid with triphenylphosphine in 95% ethanol: [1] [2]
- HAuCl4 + H2O + 2 PPh3 → (Ph3P)AuCl + Ph3PO + 3 HCl
Ph3PAuCl can also be prepared by treating a thioether complex of gold like (dimethyl sulfide)gold(I) chloride, [(Me2S)AuCl], with triphenylphosphine. [3]
The complex adopts a linear coordination geometry, which is typical of most gold(I) compounds. [4] It crystallizes in the orthorhombic space group P212121 with a = 12.300(4) Å, b = 13.084(4) Å, c = 10.170(3) Å with Z = 4 formula units per unit cell. [5]
Reactivity
Triphenylphosphinegold(I) chloride is a popular stable precursor for a cationic gold(I) catalyst used in organic synthesis. [3] Typically, it is treated with silver(I) salts of weakly coordinating anions (e.g., X– = SbF6–, BF4–, TfO–, or Tf2N–) to generate a weakly bound Ph3PAu–X complex, in equilibrium with the catalytically-active species [Ph3PAu]+X– in solution. Among these, only the bistriflimide complex Ph3PAuNTf2 can be isolated as the pure compound. [6] The nitrate complex Ph3PAuONO2 and the oxonium species [(Ph3PAu)3O]+[BF4– are also prepared from the chloride. [7]
As shown in the scheme below, the methyl complex Ph3PAuMe is prepared from triphenylphosphinegold(I) chloride by transmetalation with a Grignard reagent. Further treatment of Ph3PAuMe with methyllithium displaces the phosphine ligand and generates lithium di- and tetramethylaurate, Li+[AuMe2– and Li+[AuMe4–, respectively. [8] [9]
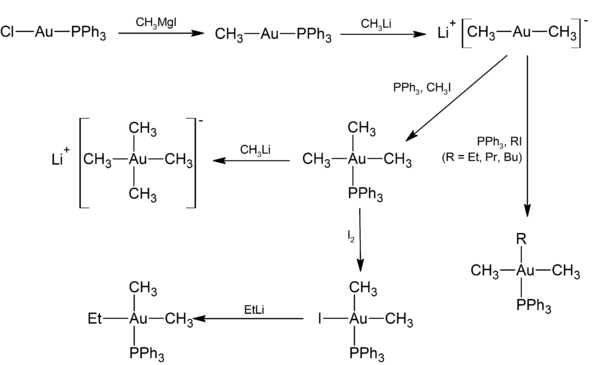
References
- ^ Pierre Braunstein; Hans Lehner; Dominique Matt (1990). A Platinum-Gold Cluster: Chloro-1κCl-Bis(Triethylphosphine-1κP)Bis(Triphenylphosphine)-2κP, 3κP-Triangulo- Digold-Platinum(1 +) Trifluoromethanesulfonate. Inorganic Syntheses. Vol. 27. pp. 218–221. doi: 10.1002/9780470132586.ch42.
- ^ M. I. Bruce; B. K. Nicholson; O. Bin Shawkataly (1989). "Synthesis of Gold-Containing Mixed-Metal Cluster Complexes". Inorganic Syntheses. Vol. 26. pp. 324–328. doi: 10.1002/9780470132579.ch59. ISBN 9780470132579.
- ^ a b Gorin, David J.; Sherry, Benjamin D.; Toste, F. Dean (2008), "Triphenylphosphinegold(I) chloride", Encyclopedia of Reagents for Organic Synthesis, American Cancer Society, doi: 10.1002/047084289x.rn00803, ISBN 9780470842898
- ^ Baenziger, N. C.; Bennett, W. E.; Soborofe, D. M. (1976). "Chloro(triphenylphosphine)gold(I)". Acta Crystallographica Section B. 32 (3): 962. doi: 10.1107/S0567740876004330.
- ^ Borissova, Alexandra O.; Korlyukov, Alexander A.; Antipin, Mikhail Yu.; Lyssenko, Konstantin A. (2008). "Estimation of Dissociation Energy in Donor−Acceptor Complex AuCl·PPh3via Topological Analysis of the Experimental Electron Density Distribution Function". The Journal of Physical Chemistry A. 112 (46): 11519–22. doi: 10.1021/jp807258d. PMID 18959385.
- ^ Mézailles, Nicolas; Ricard, Louis; Gagosz, Fabien (2005-09-01). "Phosphine Gold(I) Bis-(trifluoromethanesulfonyl)imidate Complexes as New Highly Efficient and Air-Stable Catalysts for the Cycloisomerization of Enynes". Organic Letters. 7 (19): 4133–4136. doi: 10.1021/ol0515917. ISSN 1523-7060. PMID 16146370.
- ^ A. M. Mueting, B. D. Alexander, P. D. Boyle, A. L. Casalnuovo, L. N. Ito, B. J. Johnson, L. H. Pignolet "Mixed-Metal-Gold Phosphine Cluster Compounds" Inorganic Syntheses, 1992, Volume 29, Pages 279–298, 2007. doi: 10.1002/9780470132609.ch63
- ^ Rice, Gary W.; Tobias, R. Stuart (1976). "Isolation of thermally stable compounds containing the dimethylaurate(I) and tetramethylaurate(III) anions". Inorganic Chemistry. 15 (2): 489–490. doi: 10.1021/ic50156a058. ISSN 0020-1669.
- ^ Zhu, Dunming; Lindeman, Sergey V.; Kochi, Jay K. (1999). "X-ray Crystal Structures and the Facile Oxidative (Au−C) Cleavage of the Dimethylaurate(I) and Tetramethylaurate(III) Homologues". Organometallics. 18 (11): 2241–2248. doi: 10.1021/om990043s. ISSN 0276-7333.