| |
| Names | |
|---|---|
|
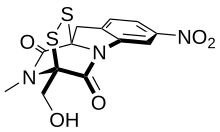
IUPAC name
(3R,10aR)-3-(hydroxymethyl)-2-methyl-7-nitro-2,3-dihydro-10H-3,10a-epidithiopyrazino[1,2-a]indole-1,4-dione
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C13H11N3O5S2 | |
| Molar mass | 353.37 g·mol−1 |
| Appearance | Yellow powder |
| Melting point | 181-182 °C |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Glionitrin A is an antibiotic-anticancer compound made by microbes found in an abandoned mine. [1] The total synthesis of (−)-glionitrin A was reported by Daniel Strand and co-workers in 2021. [2]
- ^ Park, H. B.; Kwon, H. C.; Lee, C. H.; Yang, H. O. (February 2009). "Glionitrin A, an antibiotic-antitumor metabolite derived from competitive interaction between abandoned mine microbes". J. Nat. Prod. 72 (2): 248–52. doi: 10.1021/np800606e. PMID 19159274.
- ^ Koning, N.R.; Sundin, A.P.; Strand, D. (22 November 2021). "Total Synthesis of (−)-Glionitrin A and B Enabled by an Asymmetric Oxidative Sulfenylation of Triketopiperazines". J. Am. Chem. Soc. 143 (50): 21218–21222. doi: 10.1021/jacs.1c10364. PMC 8704193. PMID 34808045.

| |
| Names | |
|---|---|
|
IUPAC name
(3R,10aR)-3-(hydroxymethyl)-2-methyl-7-nitro-2,3-dihydro-10H-3,10a-epidithiopyrazino[1,2-a]indole-1,4-dione
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C13H11N3O5S2 | |
| Molar mass | 353.37 g·mol−1 |
| Appearance | Yellow powder |
| Melting point | 181-182 °C |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Glionitrin A is an antibiotic-anticancer compound made by microbes found in an abandoned mine. [1] The total synthesis of (−)-glionitrin A was reported by Daniel Strand and co-workers in 2021. [2]
- ^ Park, H. B.; Kwon, H. C.; Lee, C. H.; Yang, H. O. (February 2009). "Glionitrin A, an antibiotic-antitumor metabolite derived from competitive interaction between abandoned mine microbes". J. Nat. Prod. 72 (2): 248–52. doi: 10.1021/np800606e. PMID 19159274.
- ^ Koning, N.R.; Sundin, A.P.; Strand, D. (22 November 2021). "Total Synthesis of (−)-Glionitrin A and B Enabled by an Asymmetric Oxidative Sulfenylation of Triketopiperazines". J. Am. Chem. Soc. 143 (50): 21218–21222. doi: 10.1021/jacs.1c10364. PMC 8704193. PMID 34808045.