Folate receptor 1 (Folate receptor alpha, FOLR1) is a protein that in humans is encoded by the FOLR1 gene. [5] [6]
The protein encoded by this gene is a member of the folate receptor (FOLR) family. Members of this family have a high affinity for folic acid and for several reduced folic acid derivatives, and mediate delivery of 5-methyltetrahydrofolate to the interior of cells.
Functions
This receptor is responsible for binding to folic acid and its derivatives, which becomes crucial during fetal development. By adding folate supplementation during pregnancy, neural tube defects in the fetus are prevented. Folate derivatives are necessary for important metabolic processes such as DNA, protein and lipid methylation. More importantly, folate plays a major role in DNA replication and cell division, which are common characteristics of rapid growth. Even though it is unclear how folate affects neural tube formation, scientists are certain that without appropriate folate levels, neural tube defects can develop through human and mice studies. Neural tube defects refer to the improper development of the neural tube by not being sealed correctly. This results in exencephaly or spina bifida, both nervous system abnormalities. [7]
This gene is composed of 7 exons; exons 1 through 4 encode the 5' UTR and exons 4 through 7 encode the open reading frame. Due to the presence of 2 promoters, multiple transcription start sites, and alternative splicing of exons, several transcript variants are derived from this gene. These variants differ in the lengths of 5' and 3' UTR, but they encode an identical amino acid sequence. [6]
Clinical significance
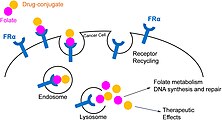
FRα, due to its high expression in some tumors, is an attractive therapeutic target for the development of novel anti-cancer agents in order to limit toxic side-effects on off-target tissues. [8]
FRa can be overexpressed by a number of epithelial-derived tumors including ovarian, breast, renal, lung, colorectal, and brain. According to a review published in 2020, elevated expression of FRa was noted in mesotheliomas (72-100% of cases), triple-negative breast cancer (35-68% of cases) and epithelial ovarian cancer (76-89% of cases). [9]
Therefore, antibodies to FRa are being developed for use in targeted therapies, with one example being farletuzumab, in a phase III trial for ovarian cancer. Further, FRa-binding markers have been created in an attempt to visualise FRa-expressing tumors. In 2021, the fluorescent marker pafolacianine was approved for identification of malignant lesions during surgeries.
Autoantibodies to the FRA have been linked to neurodevelopmental diseases, [10] particularly cerebral folate deficiency [11] schizophrenia [11] and autism spectrum disorder. [12] Recent studies have shown that these neurodevelopmental disorders can be treated with folinic acid. [12] [13]
Figures

See also
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000110195 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000001827 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Campbell IG, Jones TA, Foulkes WD, Trowsdale J (October 1991). "Folate-binding protein is a marker for ovarian cancer". Cancer Research. 51 (19): 5329–38. PMID 1717147.
- ^ a b "Entrez Gene: FOLR1 folate receptor 1 (adult)".
- ^ Balashova OA, Visina O, Borodinsky LN (2017-04-15). "Folate receptor 1 is necessary for neural plate cell apical constriction during Xenopus neural tube formation". Development. 144 (8): 1518–1530. doi: 10.1242/dev.137315. ISSN 0950-1991. PMC 5399658. PMID 28255006.
- ^ a b Razaghi A, Zickler AM, Spallholz J, Kirsch G, Björnstedt M (June 2021). "Selenofolate inhibits the proliferation of IGROV1 cancer cells independently from folate receptor alpha". Heliyon. 7 (6): e07254. Bibcode: 2021Heliy...707254R. doi: 10.1016/j.heliyon.2021.e07254. PMC 8209087. PMID 34169173.
- ^ Scaranti M, Cojocaru E, Banerjee S, Banerji U (June 2020). "Exploiting the folate receptor α in oncology". Nature Reviews. Clinical Oncology. 17 (6): 349–359. doi: 10.1038/s41571-020-0339-5. PMID 32152484. S2CID 212641989.
- ^ Frye RE, Slattery JC, Quadros EV (August 2017). "Folate metabolism abnormalities in autism: potential biomarkers". Biomarkers in Medicine. 11 (8): 687–699. doi: 10.2217/bmm-2017-0109. PMID 28770615.
- ^ a b Ramaekers VT, Rothenberg SP, Sequeira JM, Opladen T, Blau N, Quadros EV, Selhub J (May 2005). "Autoantibodies to folate receptors in the cerebral folate deficiency syndrome". The New England Journal of Medicine. 352 (19): 1985–91. doi: 10.1056/NEJMoa043160. PMID 15888699.
- ^ a b Frye RE, Sequeira JM, Quadros EV, James SJ, Rossignol DA (March 2013). "Cerebral folate receptor autoantibodies in autism spectrum disorder". Molecular Psychiatry. 18 (3): 369–81. doi: 10.1038/mp.2011.175. PMC 3578948. PMID 22230883.
- ^ Frye RE, Slattery J, Delhey L, Furgerson B, Strickland T, Tippett M, et al. (February 2018). "Folinic acid improves verbal communication in children with autism and language impairment: a randomized double-blind placebo-controlled trial". Molecular Psychiatry. 23 (2): 247–256. doi: 10.1038/mp.2016.168. PMC 5794882. PMID 27752075.
- ^ Mafi S, Laroche-Raynaud C, Chazelas P, Lia AS, Derouault P, Sturtz F, Baaj Y, Froget R, Rio M, Benoist JF, Poumeaud F, Favreau F, Faye PA (October 2020). "Pharmacoresistant Epilepsy in Childhood: Think of the Cerebral Folate Deficiency, a Treatable Disease". Brain Sciences. 10 (11): 762. doi: 10.3390/brainsci10110762. PMC 7690394. PMID 33105619.
- ^ Tummers QR, Hoogstins CE, Gaarenstroom KN, de Kroon CD, van Poelgeest MI, Vuyk J, Bosse T, Smit VT, van de Velde CJ, Cohen AF, Low PS, Burggraaf J, Vahrmeijer AL (May 2016). "Intraoperative imaging of folate receptor alpha positive ovarian and breast cancer using the tumor specific agent EC17". Oncotarget. 7 (22): 32144–55. doi: 10.18632/oncotarget.8282. PMC 5078003. PMID 27014973.
Further reading
- Henderson GB (1990). "Folate-binding proteins". Annual Review of Nutrition. 10: 319–35. doi: 10.1146/annurev.nu.10.070190.001535. PMID 2166548.
- Kelemen LE (July 2006). "The role of folate receptor alpha in cancer development, progression and treatment: cause, consequence or innocent bystander?". International Journal of Cancer. 119 (2): 243–50. doi: 10.1002/ijc.21712. PMID 16453285. S2CID 1966455.
- Ragoussis J, Senger G, Trowsdale J, Campbell IG (October 1992). "Genomic organization of the human folate receptor genes on chromosome 11q13". Genomics. 14 (2): 423–30. doi: 10.1016/S0888-7543(05)80236-8. PMID 1330883.
- Sadasivan E, Cedeno M, Rothenberg SP (May 1992). "Genomic organization of the gene and a related pseudogene for a human folate binding protein". Biochimica et Biophysica Acta (BBA) - Gene Structure and Expression. 1131 (1): 91–4. doi: 10.1016/0167-4781(92)90103-7. PMID 1581364.
- Coney LR, Tomassetti A, Carayannopoulos L, Frasca V, Kamen BA, Colnaghi MI, Zurawski VR (November 1991). "Cloning of a tumor-associated antigen: MOv18 and MOv19 antibodies recognize a folate-binding protein". Cancer Research. 51 (22): 6125–32. PMID 1840502.
- Lacey SW, Sanders JM, Rothberg KG, Anderson RG, Kamen BA (August 1989). "Complementary DNA for the folate binding protein correctly predicts anchoring to the membrane by glycosyl-phosphatidylinositol". The Journal of Clinical Investigation. 84 (2): 715–20. doi: 10.1172/JCI114220. PMC 548937. PMID 2527252.
- Sadasivan E, Rothenberg SP (April 1989). "The complete amino acid sequence of a human folate binding protein from KB cells determined from the cDNA". The Journal of Biological Chemistry. 264 (10): 5806–11. doi: 10.1016/S0021-9258(18)83621-3. PMID 2538429.
- Elwood PC (September 1989). "Molecular cloning and characterization of the human folate-binding protein cDNA from placenta and malignant tissue culture (KB) cells". The Journal of Biological Chemistry. 264 (25): 14893–901. doi: 10.1016/S0021-9258(18)63786-X. PMID 2768245.
- Sadasivan E, Rothenberg SP (November 1988). "Molecular cloning of the complementary DNA for a human folate binding protein". Proceedings of the Society for Experimental Biology and Medicine. 189 (2): 240–4. doi: 10.3181/00379727-189-42804. PMID 3194438. S2CID 36960775.
- Luhrs CA, Pitiranggon P, da Costa M, Rothenberg SP, Slomiany BL, Brink L, et al. (September 1987). "Purified membrane and soluble folate binding proteins from cultured KB cells have similar amino acid compositions and molecular weights but differ in fatty acid acylation". Proceedings of the National Academy of Sciences of the United States of America. 84 (18): 6546–9. Bibcode: 1987PNAS...84.6546L. doi: 10.1073/pnas.84.18.6546. PMC 299115. PMID 3476960.
- Yan W, Ratnam M (November 1995). "Preferred sites of glycosylphosphatidylinositol modification in folate receptors and constraints in the primary structure of the hydrophobic portion of the signal". Biochemistry. 34 (44): 14594–600. doi: 10.1021/bi00044a039. PMID 7578066.
- Saikawa Y, Price K, Hance KW, Chen TY, Elwood PC (August 1995). "Structural and functional analysis of the human KB cell folate receptor gene P4 promoter: cooperation of three clustered Sp1-binding sites with initiator region for basal promoter activity". Biochemistry. 34 (31): 9951–61. doi: 10.1021/bi00031a018. PMID 7632694.
- Prasad PD, Ramamoorthy S, Moe AJ, Smith CH, Leibach FH, Ganapathy V (August 1994). "Selective expression of the high-affinity isoform of the folate receptor (FR-alpha) in the human placental syncytiotrophoblast and choriocarcinoma cells". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 1223 (1): 71–5. doi: 10.1016/0167-4889(94)90074-4. PMID 8061055.
- Maruyama K, Sugano S (January 1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi: 10.1016/0378-1119(94)90802-8. PMID 8125298.
- Elwood PC, Nachmanoff K, Saikawa Y, Page ST, Pacheco P, Roberts S, Chung KN (February 1997). "The divergent 5' termini of the alpha human folate receptor (hFR) mRNAs originate from two tissue-specific promoters and alternative splicing: characterization of the alpha hFR gene structure". Biochemistry. 36 (6): 1467–78. doi: 10.1021/bi962070h. PMID 9063895.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (October 1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi: 10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Barber RC, Shaw GM, Lammer EJ, Greer KA, Biela TA, Lacey SW, et al. (April 1998). "Lack of association between mutations in the folate receptor-alpha gene and spina bifida". American Journal of Medical Genetics. 76 (4): 310–7. doi: 10.1002/(SICI)1096-8628(19980401)76:4<310::AID-AJMG6>3.0.CO;2-T. PMID 9545095.
- Tomassetti A, Bottero F, Mazzi M, Miotti S, Colnaghi MI, Canevari S (January 1999). "Molecular requirements for attachment of the glycosylphosphatidylinositol anchor to the human alpha folate receptor". Journal of Cellular Biochemistry. 72 (1): 111–8. doi: 10.1002/(SICI)1097-4644(19990101)72:1<111::AID-JCB12>3.0.CO;2-1. PMID 10025672. S2CID 23174724.
| FOLR1 | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | FOLR1, FBP, FOLR, Folate receptor 1, folate receptor 1 (adult), folate receptor alpha, FRalpha, NCFTD | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 136430; MGI: 95568; HomoloGene: 7322; GeneCards: FOLR1; OMA: FOLR1 - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Folate receptor 1 (Folate receptor alpha, FOLR1) is a protein that in humans is encoded by the FOLR1 gene. [5] [6]
The protein encoded by this gene is a member of the folate receptor (FOLR) family. Members of this family have a high affinity for folic acid and for several reduced folic acid derivatives, and mediate delivery of 5-methyltetrahydrofolate to the interior of cells.
Functions
This receptor is responsible for binding to folic acid and its derivatives, which becomes crucial during fetal development. By adding folate supplementation during pregnancy, neural tube defects in the fetus are prevented. Folate derivatives are necessary for important metabolic processes such as DNA, protein and lipid methylation. More importantly, folate plays a major role in DNA replication and cell division, which are common characteristics of rapid growth. Even though it is unclear how folate affects neural tube formation, scientists are certain that without appropriate folate levels, neural tube defects can develop through human and mice studies. Neural tube defects refer to the improper development of the neural tube by not being sealed correctly. This results in exencephaly or spina bifida, both nervous system abnormalities. [7]
This gene is composed of 7 exons; exons 1 through 4 encode the 5' UTR and exons 4 through 7 encode the open reading frame. Due to the presence of 2 promoters, multiple transcription start sites, and alternative splicing of exons, several transcript variants are derived from this gene. These variants differ in the lengths of 5' and 3' UTR, but they encode an identical amino acid sequence. [6]
Clinical significance

FRα, due to its high expression in some tumors, is an attractive therapeutic target for the development of novel anti-cancer agents in order to limit toxic side-effects on off-target tissues. [8]
FRa can be overexpressed by a number of epithelial-derived tumors including ovarian, breast, renal, lung, colorectal, and brain. According to a review published in 2020, elevated expression of FRa was noted in mesotheliomas (72-100% of cases), triple-negative breast cancer (35-68% of cases) and epithelial ovarian cancer (76-89% of cases). [9]
Therefore, antibodies to FRa are being developed for use in targeted therapies, with one example being farletuzumab, in a phase III trial for ovarian cancer. Further, FRa-binding markers have been created in an attempt to visualise FRa-expressing tumors. In 2021, the fluorescent marker pafolacianine was approved for identification of malignant lesions during surgeries.
Autoantibodies to the FRA have been linked to neurodevelopmental diseases, [10] particularly cerebral folate deficiency [11] schizophrenia [11] and autism spectrum disorder. [12] Recent studies have shown that these neurodevelopmental disorders can be treated with folinic acid. [12] [13]
Figures


See also
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000110195 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000001827 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Campbell IG, Jones TA, Foulkes WD, Trowsdale J (October 1991). "Folate-binding protein is a marker for ovarian cancer". Cancer Research. 51 (19): 5329–38. PMID 1717147.
- ^ a b "Entrez Gene: FOLR1 folate receptor 1 (adult)".
- ^ Balashova OA, Visina O, Borodinsky LN (2017-04-15). "Folate receptor 1 is necessary for neural plate cell apical constriction during Xenopus neural tube formation". Development. 144 (8): 1518–1530. doi: 10.1242/dev.137315. ISSN 0950-1991. PMC 5399658. PMID 28255006.
- ^ a b Razaghi A, Zickler AM, Spallholz J, Kirsch G, Björnstedt M (June 2021). "Selenofolate inhibits the proliferation of IGROV1 cancer cells independently from folate receptor alpha". Heliyon. 7 (6): e07254. Bibcode: 2021Heliy...707254R. doi: 10.1016/j.heliyon.2021.e07254. PMC 8209087. PMID 34169173.
- ^ Scaranti M, Cojocaru E, Banerjee S, Banerji U (June 2020). "Exploiting the folate receptor α in oncology". Nature Reviews. Clinical Oncology. 17 (6): 349–359. doi: 10.1038/s41571-020-0339-5. PMID 32152484. S2CID 212641989.
- ^ Frye RE, Slattery JC, Quadros EV (August 2017). "Folate metabolism abnormalities in autism: potential biomarkers". Biomarkers in Medicine. 11 (8): 687–699. doi: 10.2217/bmm-2017-0109. PMID 28770615.
- ^ a b Ramaekers VT, Rothenberg SP, Sequeira JM, Opladen T, Blau N, Quadros EV, Selhub J (May 2005). "Autoantibodies to folate receptors in the cerebral folate deficiency syndrome". The New England Journal of Medicine. 352 (19): 1985–91. doi: 10.1056/NEJMoa043160. PMID 15888699.
- ^ a b Frye RE, Sequeira JM, Quadros EV, James SJ, Rossignol DA (March 2013). "Cerebral folate receptor autoantibodies in autism spectrum disorder". Molecular Psychiatry. 18 (3): 369–81. doi: 10.1038/mp.2011.175. PMC 3578948. PMID 22230883.
- ^ Frye RE, Slattery J, Delhey L, Furgerson B, Strickland T, Tippett M, et al. (February 2018). "Folinic acid improves verbal communication in children with autism and language impairment: a randomized double-blind placebo-controlled trial". Molecular Psychiatry. 23 (2): 247–256. doi: 10.1038/mp.2016.168. PMC 5794882. PMID 27752075.
- ^ Mafi S, Laroche-Raynaud C, Chazelas P, Lia AS, Derouault P, Sturtz F, Baaj Y, Froget R, Rio M, Benoist JF, Poumeaud F, Favreau F, Faye PA (October 2020). "Pharmacoresistant Epilepsy in Childhood: Think of the Cerebral Folate Deficiency, a Treatable Disease". Brain Sciences. 10 (11): 762. doi: 10.3390/brainsci10110762. PMC 7690394. PMID 33105619.
- ^ Tummers QR, Hoogstins CE, Gaarenstroom KN, de Kroon CD, van Poelgeest MI, Vuyk J, Bosse T, Smit VT, van de Velde CJ, Cohen AF, Low PS, Burggraaf J, Vahrmeijer AL (May 2016). "Intraoperative imaging of folate receptor alpha positive ovarian and breast cancer using the tumor specific agent EC17". Oncotarget. 7 (22): 32144–55. doi: 10.18632/oncotarget.8282. PMC 5078003. PMID 27014973.
Further reading
- Henderson GB (1990). "Folate-binding proteins". Annual Review of Nutrition. 10: 319–35. doi: 10.1146/annurev.nu.10.070190.001535. PMID 2166548.
- Kelemen LE (July 2006). "The role of folate receptor alpha in cancer development, progression and treatment: cause, consequence or innocent bystander?". International Journal of Cancer. 119 (2): 243–50. doi: 10.1002/ijc.21712. PMID 16453285. S2CID 1966455.
- Ragoussis J, Senger G, Trowsdale J, Campbell IG (October 1992). "Genomic organization of the human folate receptor genes on chromosome 11q13". Genomics. 14 (2): 423–30. doi: 10.1016/S0888-7543(05)80236-8. PMID 1330883.
- Sadasivan E, Cedeno M, Rothenberg SP (May 1992). "Genomic organization of the gene and a related pseudogene for a human folate binding protein". Biochimica et Biophysica Acta (BBA) - Gene Structure and Expression. 1131 (1): 91–4. doi: 10.1016/0167-4781(92)90103-7. PMID 1581364.
- Coney LR, Tomassetti A, Carayannopoulos L, Frasca V, Kamen BA, Colnaghi MI, Zurawski VR (November 1991). "Cloning of a tumor-associated antigen: MOv18 and MOv19 antibodies recognize a folate-binding protein". Cancer Research. 51 (22): 6125–32. PMID 1840502.
- Lacey SW, Sanders JM, Rothberg KG, Anderson RG, Kamen BA (August 1989). "Complementary DNA for the folate binding protein correctly predicts anchoring to the membrane by glycosyl-phosphatidylinositol". The Journal of Clinical Investigation. 84 (2): 715–20. doi: 10.1172/JCI114220. PMC 548937. PMID 2527252.
- Sadasivan E, Rothenberg SP (April 1989). "The complete amino acid sequence of a human folate binding protein from KB cells determined from the cDNA". The Journal of Biological Chemistry. 264 (10): 5806–11. doi: 10.1016/S0021-9258(18)83621-3. PMID 2538429.
- Elwood PC (September 1989). "Molecular cloning and characterization of the human folate-binding protein cDNA from placenta and malignant tissue culture (KB) cells". The Journal of Biological Chemistry. 264 (25): 14893–901. doi: 10.1016/S0021-9258(18)63786-X. PMID 2768245.
- Sadasivan E, Rothenberg SP (November 1988). "Molecular cloning of the complementary DNA for a human folate binding protein". Proceedings of the Society for Experimental Biology and Medicine. 189 (2): 240–4. doi: 10.3181/00379727-189-42804. PMID 3194438. S2CID 36960775.
- Luhrs CA, Pitiranggon P, da Costa M, Rothenberg SP, Slomiany BL, Brink L, et al. (September 1987). "Purified membrane and soluble folate binding proteins from cultured KB cells have similar amino acid compositions and molecular weights but differ in fatty acid acylation". Proceedings of the National Academy of Sciences of the United States of America. 84 (18): 6546–9. Bibcode: 1987PNAS...84.6546L. doi: 10.1073/pnas.84.18.6546. PMC 299115. PMID 3476960.
- Yan W, Ratnam M (November 1995). "Preferred sites of glycosylphosphatidylinositol modification in folate receptors and constraints in the primary structure of the hydrophobic portion of the signal". Biochemistry. 34 (44): 14594–600. doi: 10.1021/bi00044a039. PMID 7578066.
- Saikawa Y, Price K, Hance KW, Chen TY, Elwood PC (August 1995). "Structural and functional analysis of the human KB cell folate receptor gene P4 promoter: cooperation of three clustered Sp1-binding sites with initiator region for basal promoter activity". Biochemistry. 34 (31): 9951–61. doi: 10.1021/bi00031a018. PMID 7632694.
- Prasad PD, Ramamoorthy S, Moe AJ, Smith CH, Leibach FH, Ganapathy V (August 1994). "Selective expression of the high-affinity isoform of the folate receptor (FR-alpha) in the human placental syncytiotrophoblast and choriocarcinoma cells". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 1223 (1): 71–5. doi: 10.1016/0167-4889(94)90074-4. PMID 8061055.
- Maruyama K, Sugano S (January 1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi: 10.1016/0378-1119(94)90802-8. PMID 8125298.
- Elwood PC, Nachmanoff K, Saikawa Y, Page ST, Pacheco P, Roberts S, Chung KN (February 1997). "The divergent 5' termini of the alpha human folate receptor (hFR) mRNAs originate from two tissue-specific promoters and alternative splicing: characterization of the alpha hFR gene structure". Biochemistry. 36 (6): 1467–78. doi: 10.1021/bi962070h. PMID 9063895.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (October 1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi: 10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Barber RC, Shaw GM, Lammer EJ, Greer KA, Biela TA, Lacey SW, et al. (April 1998). "Lack of association between mutations in the folate receptor-alpha gene and spina bifida". American Journal of Medical Genetics. 76 (4): 310–7. doi: 10.1002/(SICI)1096-8628(19980401)76:4<310::AID-AJMG6>3.0.CO;2-T. PMID 9545095.
- Tomassetti A, Bottero F, Mazzi M, Miotti S, Colnaghi MI, Canevari S (January 1999). "Molecular requirements for attachment of the glycosylphosphatidylinositol anchor to the human alpha folate receptor". Journal of Cellular Biochemistry. 72 (1): 111–8. doi: 10.1002/(SICI)1097-4644(19990101)72:1<111::AID-JCB12>3.0.CO;2-1. PMID 10025672. S2CID 23174724.




