
Size of this preview:
800 × 456 pixels. Other resolutions:
320 × 182 pixels |
640 × 365 pixels |
1,024 × 584 pixels |
1,280 × 730 pixels |
3,000 × 1,710 pixels.
Original file (3,000 × 1,710 pixels, file size: 1,017 KB, MIME type: image/png)
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 12:53, 29 November 2018 |
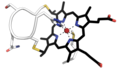 | 3,000 × 1,710 (1,017 KB) | Antibob | {{Information |description ={{en|1=Cysteine-X-X-Cysteine-Histidine (CXXCH) binding motif in cytochrome c proteins. Amino acid side chains are shown in white, the heme in black. The heme iron is coordinated in the heme plane by four nitrogen atoms and axially by a histidine side chain and a methionine side chain of the surrounding protein. Model used for preparation of the image: human recombinant cytochrome c, {{PDB|3ZCF}}.}} |date =2018-11-29 |source ={{own}} |author... |
File usage
The following pages on the English Wikipedia use this file (pages on other projects are not listed):
Global file usage
The following other wikis use this file:
- Usage on sk.wikipedia.org
Metadata

Size of this preview:
800 × 456 pixels. Other resolutions:
320 × 182 pixels |
640 × 365 pixels |
1,024 × 584 pixels |
1,280 × 730 pixels |
3,000 × 1,710 pixels.
Original file (3,000 × 1,710 pixels, file size: 1,017 KB, MIME type: image/png)
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 12:53, 29 November 2018 |
 | 3,000 × 1,710 (1,017 KB) | Antibob | {{Information |description ={{en|1=Cysteine-X-X-Cysteine-Histidine (CXXCH) binding motif in cytochrome c proteins. Amino acid side chains are shown in white, the heme in black. The heme iron is coordinated in the heme plane by four nitrogen atoms and axially by a histidine side chain and a methionine side chain of the surrounding protein. Model used for preparation of the image: human recombinant cytochrome c, {{PDB|3ZCF}}.}} |date =2018-11-29 |source ={{own}} |author... |
File usage
The following pages on the English Wikipedia use this file (pages on other projects are not listed):
Global file usage
The following other wikis use this file:
- Usage on sk.wikipedia.org