| |

| |
| Names | |
|---|---|
|
Preferred IUPAC name
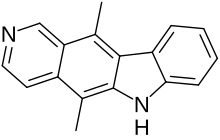
5,11-Dimethyl-6H-pyrido[4,3-b]carbazole | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.007.514 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C17H14N2 | |
| Molar mass | 246.313 g·mol−1 |
| Appearance | Yellow crystalline powder [1] |
| Density | 1.257±0.06 g/cm3 [2] |
| Melting point | 316–318 °C (601–604 °F; 589–591 K) [2] |
| Very low [3] | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
toxic |
| GHS labelling: | |
 [4]
[4]
| |
| H301 [4] | |
| P264, P270, P301+P310, P321, P330, P405, P501 [4] | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Ellipticine is a tetracyclic alkaloid first extracted from trees of the species Ochrosia elliptica and Rauvolfia sandwicensis, [5] [6] which inhibits the enzyme topoisomerase II via intercalative binding to DNA. [7]
Natural occurrence and synthesis
Ellipticine is an organic compound present in several trees within the genera Ochrosia, Rauvolfia, Aspidosperma, and Apocynaceae. [8] It was first isolated from Ochrosia elliptica Labill., a flowering tree native to Australia and New Caledonia which gives the alkaloid its name, in 1959, [5] and synthesised by Robert Burns Woodward later the same year. [6]
Biological activity
Ellipticine is a known intercalator, capable of entering a DNA strand between base pairs. In its intercalated state, ellipticine binds strongly [9] and lies parallel to the base pairs, [10] increasing the superhelical density of the DNA. [11] Intercalated ellipticine binds directly to topoisomerase II, an enzyme involved in DNA replication, [12] inhibiting the enzyme and resulting in powerful antitumour activity. [10] In clinical trials, ellipticine derivatives have been observed to induce remission of tumour growth, but are not used for medical purposes due to their high toxicity; side effects include nausea and vomiting, hypertension, cramp, pronounced fatigue, mouth dryness, and mycosis of the tongue and oesophagus. [13]
Further DNA damage results from the formation of covalent DNA adducts following enzymatic activation of ellipticine by with cytochromes P450 and peroxidases, meaning that ellipticine is classified as a prodrug. [14]
References
- ^ Miller, R B; Dugar, S (1989). "A regiospecific total synthesis of ellipticine via nitrene insertion". Tetrahedron Letters. 30 (3): 297–300. doi: 10.1016/S0040-4039(00)95184-0. ISSN 0040-4039.
- ^ a b "Ellipticine | 519-23-3". ChemicalBook. 2016. Retrieved 2017-05-30.
- ^ Sbai, M; Ait Lyazidi, S; Lerner, D A; del Castillo, B; Martin, M A (1996). "Use of micellar media for the fluorimetric determination of ellipticine in aqueous solutions". Journal of Pharmaceutical and Biomedical Analysis. 14 (8): 959–965. doi: 10.1016/S0731-7085(96)01759-1. ISSN 0731-7085. PMID 8818001.
- ^ a b c "Ellipticine | C17H14N2 - PubChem". PubChem. 2016. Retrieved 2017-05-30.
- ^ a b Goodwin, S; Smith, A F; Horning, E C (1959). "Alkaloids of Ochrosia elliptica Labill". Journal of the American Chemical Society. 81 (8): 1903–1908. doi: 10.1021/ja01517a031.
- ^ a b Woodward, R B; Iacobucci, G A; Hochstein, I A (1959). "The synthesis of ellipticine". Journal of the American Chemical Society. 81 (16): 4434–4435. doi: 10.1021/ja01525a085. ISSN 0002-7863.
- ^ Auclair, C (1987). "Multimodal action of antitumor agents on DNA: The ellipticine series". Archives of Biochemistry and Biophysics. 259 (1): 1–14. doi: 10.1016/0003-9861(87)90463-2. ISSN 0003-9861. PMID 3318697.
- ^ Isah, T (2016). "Anticancer Alkaloids from Trees: Development into Drugs". Pharmacognosy Reviews. 10 (20): 90–99. doi: 10.4103/0973-7847.194047. ISSN 0973-7847. PMC 5214563. PMID 28082790.
- ^ Kohn, K W; Waring, M J; Glaubiger, D; Friedman, C A (1975). "Intercalative Binding of Ellipticine to DNA". Cancer Research. 35 (1): 71–76. ISSN 0008-5472. PMID 1109798.
- ^ a b Canals, A; Purciolas, M; Aymamí, J; Coll, M (2005). "The anticancer agent ellipticine unwinds DNA by intercalative binding in an orientation parallel to base pairs" (PDF). Acta Crystallographica D. 61 (7): 1009–1012. Bibcode: 2005AcCrD..61.1009C. doi: 10.1107/S0907444905015404. hdl: 10261/108793. ISSN 0907-4449. PMID 15983425.
- ^ Chu, Y; Hsu, M T (1992). "Ellipticine increases the superhelical density of intracellular SV40 DNA by intercalation". Nucleic Acids Research. 20 (15): 4033–4038. doi: 10.1093/nar/20.15.4033. ISSN 0305-1048. PMC 334084. PMID 1324474.
- ^ Froelich-Ammon, S J; Patchan, M W; Osheroff, N; Thompson, R B (1995). "Topoisomerase II binds to ellipticine in the absence or presence of DNA. Characterization of enzyme–drug interactions by fluorescence spectroscopy". Journal of Biological Chemistry. 270 (25): 14998–15004. doi: 10.1074/jbc.270.25.14998. ISSN 0021-9258. PMID 7797481.
-
^
Paoletti, C; Le Pecq, J B; Dat-Xuong, N; Juret, P; Garnier, H; Amiel, J L; Rouesse, J (1980). "Antitumor Activity, Pharmacology, and Toxicity of Ellipticines, Ellipticinium, and 9-Hydroxy Derivatives: Preliminary Clinical Trials of 2-Methyl-9-Hydroxy Ellipticinium (NSC 264-137)". Cancer Chemo- and Immunopharmacology. Recent Results in Cancer Research. Vol. 74. pp. 107–123.
doi:
10.1007/978-3-642-81488-4_15.
ISBN
978-3-642-81490-7.
ISSN
0080-0015.
PMID
7003658.
{{ cite book}}:|journal=ignored ( help) - ^ Stiborová, M; Poljaková, J; Martínková, E; Ulrichová, J; Šimánek, V; Dvořák, Z; Frei, E (2012). "Ellipticine oxidation and DNA adduct formation in human hepatocytes is catalyzed by human cytochromes P450 and enhanced by cytochrome b5". Toxicology. 302 (2–3): 233–241. doi: 10.1016/j.tox.2012.08.004. ISSN 0300-483X. PMID 22917556.

| |

| |
| Names | |
|---|---|
|
Preferred IUPAC name
5,11-Dimethyl-6H-pyrido[4,3-b]carbazole | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.007.514 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C17H14N2 | |
| Molar mass | 246.313 g·mol−1 |
| Appearance | Yellow crystalline powder [1] |
| Density | 1.257±0.06 g/cm3 [2] |
| Melting point | 316–318 °C (601–604 °F; 589–591 K) [2] |
| Very low [3] | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
toxic |
| GHS labelling: | |
 [4]
[4]
| |
| H301 [4] | |
| P264, P270, P301+P310, P321, P330, P405, P501 [4] | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Ellipticine is a tetracyclic alkaloid first extracted from trees of the species Ochrosia elliptica and Rauvolfia sandwicensis, [5] [6] which inhibits the enzyme topoisomerase II via intercalative binding to DNA. [7]
Natural occurrence and synthesis
Ellipticine is an organic compound present in several trees within the genera Ochrosia, Rauvolfia, Aspidosperma, and Apocynaceae. [8] It was first isolated from Ochrosia elliptica Labill., a flowering tree native to Australia and New Caledonia which gives the alkaloid its name, in 1959, [5] and synthesised by Robert Burns Woodward later the same year. [6]
Biological activity
Ellipticine is a known intercalator, capable of entering a DNA strand between base pairs. In its intercalated state, ellipticine binds strongly [9] and lies parallel to the base pairs, [10] increasing the superhelical density of the DNA. [11] Intercalated ellipticine binds directly to topoisomerase II, an enzyme involved in DNA replication, [12] inhibiting the enzyme and resulting in powerful antitumour activity. [10] In clinical trials, ellipticine derivatives have been observed to induce remission of tumour growth, but are not used for medical purposes due to their high toxicity; side effects include nausea and vomiting, hypertension, cramp, pronounced fatigue, mouth dryness, and mycosis of the tongue and oesophagus. [13]
Further DNA damage results from the formation of covalent DNA adducts following enzymatic activation of ellipticine by with cytochromes P450 and peroxidases, meaning that ellipticine is classified as a prodrug. [14]
References
- ^ Miller, R B; Dugar, S (1989). "A regiospecific total synthesis of ellipticine via nitrene insertion". Tetrahedron Letters. 30 (3): 297–300. doi: 10.1016/S0040-4039(00)95184-0. ISSN 0040-4039.
- ^ a b "Ellipticine | 519-23-3". ChemicalBook. 2016. Retrieved 2017-05-30.
- ^ Sbai, M; Ait Lyazidi, S; Lerner, D A; del Castillo, B; Martin, M A (1996). "Use of micellar media for the fluorimetric determination of ellipticine in aqueous solutions". Journal of Pharmaceutical and Biomedical Analysis. 14 (8): 959–965. doi: 10.1016/S0731-7085(96)01759-1. ISSN 0731-7085. PMID 8818001.
- ^ a b c "Ellipticine | C17H14N2 - PubChem". PubChem. 2016. Retrieved 2017-05-30.
- ^ a b Goodwin, S; Smith, A F; Horning, E C (1959). "Alkaloids of Ochrosia elliptica Labill". Journal of the American Chemical Society. 81 (8): 1903–1908. doi: 10.1021/ja01517a031.
- ^ a b Woodward, R B; Iacobucci, G A; Hochstein, I A (1959). "The synthesis of ellipticine". Journal of the American Chemical Society. 81 (16): 4434–4435. doi: 10.1021/ja01525a085. ISSN 0002-7863.
- ^ Auclair, C (1987). "Multimodal action of antitumor agents on DNA: The ellipticine series". Archives of Biochemistry and Biophysics. 259 (1): 1–14. doi: 10.1016/0003-9861(87)90463-2. ISSN 0003-9861. PMID 3318697.
- ^ Isah, T (2016). "Anticancer Alkaloids from Trees: Development into Drugs". Pharmacognosy Reviews. 10 (20): 90–99. doi: 10.4103/0973-7847.194047. ISSN 0973-7847. PMC 5214563. PMID 28082790.
- ^ Kohn, K W; Waring, M J; Glaubiger, D; Friedman, C A (1975). "Intercalative Binding of Ellipticine to DNA". Cancer Research. 35 (1): 71–76. ISSN 0008-5472. PMID 1109798.
- ^ a b Canals, A; Purciolas, M; Aymamí, J; Coll, M (2005). "The anticancer agent ellipticine unwinds DNA by intercalative binding in an orientation parallel to base pairs" (PDF). Acta Crystallographica D. 61 (7): 1009–1012. Bibcode: 2005AcCrD..61.1009C. doi: 10.1107/S0907444905015404. hdl: 10261/108793. ISSN 0907-4449. PMID 15983425.
- ^ Chu, Y; Hsu, M T (1992). "Ellipticine increases the superhelical density of intracellular SV40 DNA by intercalation". Nucleic Acids Research. 20 (15): 4033–4038. doi: 10.1093/nar/20.15.4033. ISSN 0305-1048. PMC 334084. PMID 1324474.
- ^ Froelich-Ammon, S J; Patchan, M W; Osheroff, N; Thompson, R B (1995). "Topoisomerase II binds to ellipticine in the absence or presence of DNA. Characterization of enzyme–drug interactions by fluorescence spectroscopy". Journal of Biological Chemistry. 270 (25): 14998–15004. doi: 10.1074/jbc.270.25.14998. ISSN 0021-9258. PMID 7797481.
-
^
Paoletti, C; Le Pecq, J B; Dat-Xuong, N; Juret, P; Garnier, H; Amiel, J L; Rouesse, J (1980). "Antitumor Activity, Pharmacology, and Toxicity of Ellipticines, Ellipticinium, and 9-Hydroxy Derivatives: Preliminary Clinical Trials of 2-Methyl-9-Hydroxy Ellipticinium (NSC 264-137)". Cancer Chemo- and Immunopharmacology. Recent Results in Cancer Research. Vol. 74. pp. 107–123.
doi:
10.1007/978-3-642-81488-4_15.
ISBN
978-3-642-81490-7.
ISSN
0080-0015.
PMID
7003658.
{{ cite book}}:|journal=ignored ( help) - ^ Stiborová, M; Poljaková, J; Martínková, E; Ulrichová, J; Šimánek, V; Dvořák, Z; Frei, E (2012). "Ellipticine oxidation and DNA adduct formation in human hepatocytes is catalyzed by human cytochromes P450 and enhanced by cytochrome b5". Toxicology. 302 (2–3): 233–241. doi: 10.1016/j.tox.2012.08.004. ISSN 0300-483X. PMID 22917556.