 | |
| Clinical data | |
|---|---|
| Trade names | Duvroq, Jesduvroq |
| Other names | GSK1278863 |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a623010 |
| License data |
|
|
Routes of administration | By mouth |
| Drug class | Hypoxia-inducible factor prolyl hydroxylase inhibitor |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.219.426 |
| Chemical and physical data | |
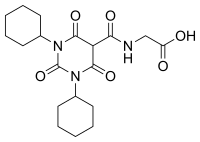
| Formula | C19H27N3O6 |
| Molar mass | 393.440 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Daprodustat, sold under the brand name Duvroq among others, is a medication that is used for the treatment of anemia due to chronic kidney disease. [2] It is a hypoxia-inducible factor prolyl hydroxylase inhibitor. [2] It is taken by mouth. [2] [3] [4]
The most common side effects include high blood pressure, thrombotic vascular events, abdominal pain, dizziness, and allergic reactions. [3] [4]
Daprodustat was approved for medical use in Japan in June 2020, [5] [6] and in the United States in February 2023. [2] [3] [7] [8] making it the first oral treatment for anemia caused by chronic kidney disease for adults in the US. [3] The US Food and Drug Administration (FDA) considers it to be a first-in-class medication. [9]
Medical uses
Daprodustat is indicated for the treatment of anemia due to chronic kidney disease. [2]
Daprodustat increases erythropoietin levels. [3]
Adverse effects
The FDA label for daprodustat has a boxed warning for an increased risk of thrombotic vascular (blood clotting) events including death, heart attack, stroke, and blood clots in the lung, legs, or dialysis access site. [4]
The most common side effects include high blood pressure, thrombotic vascular events, abdominal pain, dizziness, and allergic reactions. [3] [4]
History
The efficacy and safety of daprodustat were evaluated in 2,964 adults with anemia due to chronic kidney disease on dialysis and receiving an erythropoiesis-stimulating agent at the time of study entry in a randomized, sponsor-blind, active-controlled, global, multicenter, event-driven clinical trial (ASCEND-D; NCT02879305). [4] [10] Participants were stratified by dialysis type and were required to be on dialysis for at least four months prior to the first dose of daprodustat. [4] Participants on hemodialysis were randomized 1:1 to receive oral daprodustat (N=1,316) or intravenous epoetin alfa (N=1,308) while participants on peritoneal dialysis were randomized 1:1 to receive oral daprodustat (N=171) or subcutaneous darbepoetin alfa (N=169). [4] In this study, adults received either oral daprodustat or injected recombinant human erythropoietin (rhEPO) (a standard of care treatment for people with anemia due to chronic kidney disease). [3] [4] Daprodustat raised and maintained the hemoglobin (the protein in red blood cells that carries oxygen and is a common measure of anemia) within the target range of 10 to 11 g/dL, similar to that of the rhEPO treatment. [3] [4] The trial was conducted at 431 sites in 35 countries. [4]
The FDA granted the approval of Jesduvroq to GlaxoSmithKline LLC. [3]
Society and culture
Due to its potential applications in athletic doping, it has also been incorporated into screens for performance-enhancing drugs. [11]
Research
Daprodustat is in phase III clinical trials for the treatment of anemia caused by chronic kidney disease. [12] [13] [14]
References
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ a b c d e f "Jesduvroq- daprodustat tablet, film coated". DailyMed. 1 February 2023. Archived from the original on 11 February 2023. Retrieved 11 February 2023.
- ^
a
b
c
d
e
f
g
h
i
"FDA Approves First Oral Treatment for Anemia Caused by Chronic Kidney Disease for Adults on Dialysis". U.S.
Food and Drug Administration (FDA) (Press release). 1 February 2023.
Archived from the original on 4 February 2023. Retrieved 3 February 2023.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^
a
b
c
d
e
f
g
h
i
j
"Drug Trials Snapshots: Jesduvroq". U.S.
Food and Drug Administration (FDA). 1 February 2023.
Archived from the original on 24 February 2024. Retrieved 24 February 2024.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ Dhillon S (September 2020). "Daprodustat: First Approval". Drugs. 80 (14): 1491–1497. doi: 10.1007/s40265-020-01384-y. PMC 7471535. PMID 32880805.
- ^ "GSK receives first regulatory approval for Duvroq (daprodustat) in Japan for patients with anaemia due to chronic kidney disease" (Press release). GSK. 29 June 2020. Archived from the original on 4 February 2023. Retrieved 29 March 2021.
- ^ Harris E (15 February 2023). "FDA Approves First Oral Treatment for Kidney Disease–Induced Anemia". JAMA. 329 (9): 704. doi: 10.1001/jama.2023.1556. PMID 36790833.
- ^ "Jesduvroq (daprodustat) approved by US FDA for anemia of chronic kidney disease in adults on dialysis" (Press release). GSK US. 1 February 2023. Archived from the original on 4 February 2023. Retrieved 24 February 2024.
- ^ New Drug Therapy Approvals 2023 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2024. Archived from the original on 10 January 2024. Retrieved 9 January 2024.
- ^ Clinical trial number NCT02879305 for "Anemia Studies in Chronic Kidney Disease: Erythropoiesis Via a Novel Prolyl Hydroxylase Inhibitor Daprodustat-Dialysis (ASCEND-D)" at ClinicalTrials.gov
- ^ Thevis M, Milosovich S, Licea-Perez H, Knecht D, Cavalier T, Schänzer W (August 2016). "Mass spectrometric characterization of a prolyl hydroxylase inhibitor GSK1278863, its bishydroxylated metabolite, and its implementation into routine doping controls". Drug Testing and Analysis. 8 (8): 858–63. doi: 10.1002/dta.1870. PMID 26361079.
- ^ Schmid H, Jelkmann W (August 2016). "Investigational therapies for renal disease-induced anemia". Expert Opinion on Investigational Drugs. 25 (8): 901–16. doi: 10.1080/13543784.2016.1182981. PMID 27122198. S2CID 32493057.
- ^ Ariazi JL, Duffy KJ, Adams DF, Fitch DM, Luo L, Pappalardi M, et al. (December 2017). "Discovery and Preclinical Characterization of GSK1278863 (Daprodustat), a Small Molecule Hypoxia Inducible Factor-Prolyl Hydroxylase Inhibitor for Anemia". The Journal of Pharmacology and Experimental Therapeutics. 363 (3): 336–347. doi: 10.1124/jpet.117.242503. PMID 28928122.
- ^ Singh AK, Carroll K, McMurray JJ, Solomon S, Jha V, Johansen KL, et al. (December 2021). "Daprodustat for the Treatment of Anemia in Patients Not Undergoing Dialysis". The New England Journal of Medicine. 385 (25): 2313–2324. doi: 10.1056/NEJMoa2113380. PMID 34739196. S2CID 243761990.
 | |
| Clinical data | |
|---|---|
| Trade names | Duvroq, Jesduvroq |
| Other names | GSK1278863 |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a623010 |
| License data |
|
|
Routes of administration | By mouth |
| Drug class | Hypoxia-inducible factor prolyl hydroxylase inhibitor |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.219.426 |
| Chemical and physical data | |
| Formula | C19H27N3O6 |
| Molar mass | 393.440 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Daprodustat, sold under the brand name Duvroq among others, is a medication that is used for the treatment of anemia due to chronic kidney disease. [2] It is a hypoxia-inducible factor prolyl hydroxylase inhibitor. [2] It is taken by mouth. [2] [3] [4]
The most common side effects include high blood pressure, thrombotic vascular events, abdominal pain, dizziness, and allergic reactions. [3] [4]
Daprodustat was approved for medical use in Japan in June 2020, [5] [6] and in the United States in February 2023. [2] [3] [7] [8] making it the first oral treatment for anemia caused by chronic kidney disease for adults in the US. [3] The US Food and Drug Administration (FDA) considers it to be a first-in-class medication. [9]
Medical uses
Daprodustat is indicated for the treatment of anemia due to chronic kidney disease. [2]
Daprodustat increases erythropoietin levels. [3]
Adverse effects
The FDA label for daprodustat has a boxed warning for an increased risk of thrombotic vascular (blood clotting) events including death, heart attack, stroke, and blood clots in the lung, legs, or dialysis access site. [4]
The most common side effects include high blood pressure, thrombotic vascular events, abdominal pain, dizziness, and allergic reactions. [3] [4]
History
The efficacy and safety of daprodustat were evaluated in 2,964 adults with anemia due to chronic kidney disease on dialysis and receiving an erythropoiesis-stimulating agent at the time of study entry in a randomized, sponsor-blind, active-controlled, global, multicenter, event-driven clinical trial (ASCEND-D; NCT02879305). [4] [10] Participants were stratified by dialysis type and were required to be on dialysis for at least four months prior to the first dose of daprodustat. [4] Participants on hemodialysis were randomized 1:1 to receive oral daprodustat (N=1,316) or intravenous epoetin alfa (N=1,308) while participants on peritoneal dialysis were randomized 1:1 to receive oral daprodustat (N=171) or subcutaneous darbepoetin alfa (N=169). [4] In this study, adults received either oral daprodustat or injected recombinant human erythropoietin (rhEPO) (a standard of care treatment for people with anemia due to chronic kidney disease). [3] [4] Daprodustat raised and maintained the hemoglobin (the protein in red blood cells that carries oxygen and is a common measure of anemia) within the target range of 10 to 11 g/dL, similar to that of the rhEPO treatment. [3] [4] The trial was conducted at 431 sites in 35 countries. [4]
The FDA granted the approval of Jesduvroq to GlaxoSmithKline LLC. [3]
Society and culture
Due to its potential applications in athletic doping, it has also been incorporated into screens for performance-enhancing drugs. [11]
Research
Daprodustat is in phase III clinical trials for the treatment of anemia caused by chronic kidney disease. [12] [13] [14]
References
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ a b c d e f "Jesduvroq- daprodustat tablet, film coated". DailyMed. 1 February 2023. Archived from the original on 11 February 2023. Retrieved 11 February 2023.
- ^
a
b
c
d
e
f
g
h
i
"FDA Approves First Oral Treatment for Anemia Caused by Chronic Kidney Disease for Adults on Dialysis". U.S.
Food and Drug Administration (FDA) (Press release). 1 February 2023.
Archived from the original on 4 February 2023. Retrieved 3 February 2023.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^
a
b
c
d
e
f
g
h
i
j
"Drug Trials Snapshots: Jesduvroq". U.S.
Food and Drug Administration (FDA). 1 February 2023.
Archived from the original on 24 February 2024. Retrieved 24 February 2024.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ Dhillon S (September 2020). "Daprodustat: First Approval". Drugs. 80 (14): 1491–1497. doi: 10.1007/s40265-020-01384-y. PMC 7471535. PMID 32880805.
- ^ "GSK receives first regulatory approval for Duvroq (daprodustat) in Japan for patients with anaemia due to chronic kidney disease" (Press release). GSK. 29 June 2020. Archived from the original on 4 February 2023. Retrieved 29 March 2021.
- ^ Harris E (15 February 2023). "FDA Approves First Oral Treatment for Kidney Disease–Induced Anemia". JAMA. 329 (9): 704. doi: 10.1001/jama.2023.1556. PMID 36790833.
- ^ "Jesduvroq (daprodustat) approved by US FDA for anemia of chronic kidney disease in adults on dialysis" (Press release). GSK US. 1 February 2023. Archived from the original on 4 February 2023. Retrieved 24 February 2024.
- ^ New Drug Therapy Approvals 2023 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2024. Archived from the original on 10 January 2024. Retrieved 9 January 2024.
- ^ Clinical trial number NCT02879305 for "Anemia Studies in Chronic Kidney Disease: Erythropoiesis Via a Novel Prolyl Hydroxylase Inhibitor Daprodustat-Dialysis (ASCEND-D)" at ClinicalTrials.gov
- ^ Thevis M, Milosovich S, Licea-Perez H, Knecht D, Cavalier T, Schänzer W (August 2016). "Mass spectrometric characterization of a prolyl hydroxylase inhibitor GSK1278863, its bishydroxylated metabolite, and its implementation into routine doping controls". Drug Testing and Analysis. 8 (8): 858–63. doi: 10.1002/dta.1870. PMID 26361079.
- ^ Schmid H, Jelkmann W (August 2016). "Investigational therapies for renal disease-induced anemia". Expert Opinion on Investigational Drugs. 25 (8): 901–16. doi: 10.1080/13543784.2016.1182981. PMID 27122198. S2CID 32493057.
- ^ Ariazi JL, Duffy KJ, Adams DF, Fitch DM, Luo L, Pappalardi M, et al. (December 2017). "Discovery and Preclinical Characterization of GSK1278863 (Daprodustat), a Small Molecule Hypoxia Inducible Factor-Prolyl Hydroxylase Inhibitor for Anemia". The Journal of Pharmacology and Experimental Therapeutics. 363 (3): 336–347. doi: 10.1124/jpet.117.242503. PMID 28928122.
- ^ Singh AK, Carroll K, McMurray JJ, Solomon S, Jha V, Johansen KL, et al. (December 2021). "Daprodustat for the Treatment of Anemia in Patients Not Undergoing Dialysis". The New England Journal of Medicine. 385 (25): 2313–2324. doi: 10.1056/NEJMoa2113380. PMID 34739196. S2CID 243761990.