| |
| Names | |
|---|---|
|
IUPAC name
[4-(hydroxymethyl)cyclohexyl]methanol
| |
|
Preferred IUPAC name
(cyclohexane-1,4-diyl)dimethanol | |
| Other names
1,4–Cyclohexanedimethanol; CHDM; 1,4-Bis(hydroxymethyl)cyclohexane
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.002.972 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C8H16O2 | |
| Molar mass | 144.21 g/mol |
| Appearance | White waxy solid |
| Density | 1.02 g/ml |
| Melting point | 41 to 61 °C (106 to 142 °F; 314 to 334 K) |
| Boiling point | 284 to 288 °C (543 to 550 °F; 557 to 561 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
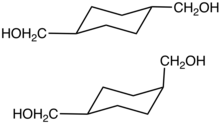
Cyclohexanedimethanol (CHDM) is a mixture of isomeric organic compounds with formula C6H10(CH2OH)2. It is a colorless low-melting solid used in the production of polyester resins. Commercial samples consist of a mixture of cis and trans isomers. It is a di-substituted derivative of cyclohexane and is classified as a diol, meaning that it has two OH functional groups. Commercial CHDM typically has a cis/trans ratio of 30:70.
CHDM is produced by catalytic hydrogenation of dimethyl terephthalate (DMT). The reaction conducted in two steps beginning with the conversion of DMT to the diester dimethyl 1,4-cyclohexanedicarboxylate (DMCD):
- C6H4(CO2CH3)2 + 3 H2 → C6H10(CO2CH3)2
In the second step DMCD is further hydrogenated to CHDM:
- C6H10(CO2CH3)2 + 4 H2 → C6H10(CH2OH)2 + 2 CH3OH
A copper chromite catalyst is usually used industrially. [1] The cis/trans ratio of the CHDM is affected by the catalyst. [2]
Byproduct of this process are 4-methylcyclohexanemethanol (CH3C6H10CH2OH) and the monoester methyl 4-methyl-4-cyclohexanecarboxylate (CH3C6H10CO2CH3, CAS registry number 51181-40-9). [3] The leading producers in CHDM are Eastman Chemical in US and SK Chemicals in South Korea.
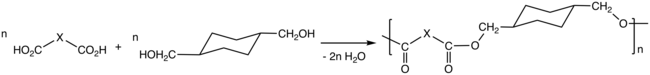
Via the process called polycondensation, CHDM is a precursor to polyesters. It is one of the most important comonomers for production of polyethylene terephthalate (PET), or polyethylene terephthalic ester (PETE), from which plastic bottles are made. [4] [5] In addition it maybe spun to form carpet fibers. [6]
Thermoplastic polyesters containing CHDM exhibit enhanced strength, clarity, and solvent resistance. The properties of the polyesters vary from the high melting crystalline poly(1,4-cyclohexylenedimethylene terephthalate), PCT, to the non-crystalline copolyesters derived from both ethylene glycol and CHDM. The properties of these polyesters also is affected by the cis/trans ratio of the CHDM monomer. [7] CHDM reduces the degree of crystallinity of PET homopolymer, improving its processability. The copolymer tends to resist degradation, e.g. to acetaldehyde. The copolymer with PET is known as glycol-modified polyethylene terephthalate, PETG. PETG is used in many fields, including electronics, automobiles, barrier, and medical, etc.
CHDM is a raw material for the production of 1,4-cyclohexanedimethanol diglycidyl ether, an epoxy diluent. [8] The key use for this diglycidyl ether is to reduce the viscosity of epoxy resins. [9]
- ^ S.R. Turner; Y. Li (2010). "Synthesis and Properties of Cyclic Diester Based Aliphatic Copolyesters". Journal of Polymer Science Part A: Polymer Chemistry. 48 (10): 2162–2169. doi: 10.1002/pola.23985.
- ^ J. M. Thomas; R. Raja (2002). "The materials Chemistry of Inorganic Catalyst". Australian Journal of Chemistry. 54: 551–560. doi: 10.1071/CH01150.
- ^ Peter Werle, Marcus Morawietz, Stefan Lundmark, Kent Sörensen, Esko Karvinen and Juha Lehtonen "Alcohols, Polyhydric" Ullmann's Encyclopedia of Industrial Chemistry, 2008, Wiley-VCH, Weinheim. doi: 10.1002/14356007.a01_305.pub2
- ^ S.R. Turner (2004). "Development of amorphous copolyesters based on 1,4- cyclohexane-dimethanol". Journal of Polymer Science Part A: Polymer Chemistry. 42 (23): 5847–5852. doi: 10.1002/pola.20460.
- ^ S. Andjelic; D.D. Jamiolkowski; R. Bezwada (2007). "Mini-review The Polyoxaesters". Polymer International. 56: 1063–1077. doi: 10.1002/pi.2257.
- ^ Hatton. "Nylon vs. Polyester Carpet Fibers: Comparison Guide". Homedit. Retrieved 2023-08-17.
- ^ S. R. Turner; R.W. Seymour; T.W. Smith (2001). "Cyclohexanedimethanol Polyesters". Encyclopedia of Polymer Science and Technology. doi: 10.1002/0471440264.pst257. ISBN 0471440264.
- ^ Crivello, James V. (2006). "Design and synthesis of multifunctional glycidyl ethers that undergo frontal polymerization". Journal of Polymer Science Part A: Polymer Chemistry. 44 (21): 6435–6448. Bibcode: 2006JPoSA..44.6435C. doi: 10.1002/pola.21761. ISSN 0887-624X.
- ^ Monte, Salvatore J. (1998), Pritchard, Geoffrey (ed.), "Diluents and viscosity modifiers for epoxy resins", Plastics Additives: An A-Z reference, Polymer Science and Technology Series, vol. 1, Dordrecht: Springer Netherlands, pp. 211–216, doi: 10.1007/978-94-011-5862-6_24, ISBN 978-94-011-5862-6, archived from the original on 2022-04-11, retrieved 2022-03-29

| |
| Names | |
|---|---|
|
IUPAC name
[4-(hydroxymethyl)cyclohexyl]methanol
| |
|
Preferred IUPAC name
(cyclohexane-1,4-diyl)dimethanol | |
| Other names
1,4–Cyclohexanedimethanol; CHDM; 1,4-Bis(hydroxymethyl)cyclohexane
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.002.972 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C8H16O2 | |
| Molar mass | 144.21 g/mol |
| Appearance | White waxy solid |
| Density | 1.02 g/ml |
| Melting point | 41 to 61 °C (106 to 142 °F; 314 to 334 K) |
| Boiling point | 284 to 288 °C (543 to 550 °F; 557 to 561 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyclohexanedimethanol (CHDM) is a mixture of isomeric organic compounds with formula C6H10(CH2OH)2. It is a colorless low-melting solid used in the production of polyester resins. Commercial samples consist of a mixture of cis and trans isomers. It is a di-substituted derivative of cyclohexane and is classified as a diol, meaning that it has two OH functional groups. Commercial CHDM typically has a cis/trans ratio of 30:70.
CHDM is produced by catalytic hydrogenation of dimethyl terephthalate (DMT). The reaction conducted in two steps beginning with the conversion of DMT to the diester dimethyl 1,4-cyclohexanedicarboxylate (DMCD):
- C6H4(CO2CH3)2 + 3 H2 → C6H10(CO2CH3)2
In the second step DMCD is further hydrogenated to CHDM:
- C6H10(CO2CH3)2 + 4 H2 → C6H10(CH2OH)2 + 2 CH3OH
A copper chromite catalyst is usually used industrially. [1] The cis/trans ratio of the CHDM is affected by the catalyst. [2]
Byproduct of this process are 4-methylcyclohexanemethanol (CH3C6H10CH2OH) and the monoester methyl 4-methyl-4-cyclohexanecarboxylate (CH3C6H10CO2CH3, CAS registry number 51181-40-9). [3] The leading producers in CHDM are Eastman Chemical in US and SK Chemicals in South Korea.
Via the process called polycondensation, CHDM is a precursor to polyesters. It is one of the most important comonomers for production of polyethylene terephthalate (PET), or polyethylene terephthalic ester (PETE), from which plastic bottles are made. [4] [5] In addition it maybe spun to form carpet fibers. [6]
Thermoplastic polyesters containing CHDM exhibit enhanced strength, clarity, and solvent resistance. The properties of the polyesters vary from the high melting crystalline poly(1,4-cyclohexylenedimethylene terephthalate), PCT, to the non-crystalline copolyesters derived from both ethylene glycol and CHDM. The properties of these polyesters also is affected by the cis/trans ratio of the CHDM monomer. [7] CHDM reduces the degree of crystallinity of PET homopolymer, improving its processability. The copolymer tends to resist degradation, e.g. to acetaldehyde. The copolymer with PET is known as glycol-modified polyethylene terephthalate, PETG. PETG is used in many fields, including electronics, automobiles, barrier, and medical, etc.
CHDM is a raw material for the production of 1,4-cyclohexanedimethanol diglycidyl ether, an epoxy diluent. [8] The key use for this diglycidyl ether is to reduce the viscosity of epoxy resins. [9]
- ^ S.R. Turner; Y. Li (2010). "Synthesis and Properties of Cyclic Diester Based Aliphatic Copolyesters". Journal of Polymer Science Part A: Polymer Chemistry. 48 (10): 2162–2169. doi: 10.1002/pola.23985.
- ^ J. M. Thomas; R. Raja (2002). "The materials Chemistry of Inorganic Catalyst". Australian Journal of Chemistry. 54: 551–560. doi: 10.1071/CH01150.
- ^ Peter Werle, Marcus Morawietz, Stefan Lundmark, Kent Sörensen, Esko Karvinen and Juha Lehtonen "Alcohols, Polyhydric" Ullmann's Encyclopedia of Industrial Chemistry, 2008, Wiley-VCH, Weinheim. doi: 10.1002/14356007.a01_305.pub2
- ^ S.R. Turner (2004). "Development of amorphous copolyesters based on 1,4- cyclohexane-dimethanol". Journal of Polymer Science Part A: Polymer Chemistry. 42 (23): 5847–5852. doi: 10.1002/pola.20460.
- ^ S. Andjelic; D.D. Jamiolkowski; R. Bezwada (2007). "Mini-review The Polyoxaesters". Polymer International. 56: 1063–1077. doi: 10.1002/pi.2257.
- ^ Hatton. "Nylon vs. Polyester Carpet Fibers: Comparison Guide". Homedit. Retrieved 2023-08-17.
- ^ S. R. Turner; R.W. Seymour; T.W. Smith (2001). "Cyclohexanedimethanol Polyesters". Encyclopedia of Polymer Science and Technology. doi: 10.1002/0471440264.pst257. ISBN 0471440264.
- ^ Crivello, James V. (2006). "Design and synthesis of multifunctional glycidyl ethers that undergo frontal polymerization". Journal of Polymer Science Part A: Polymer Chemistry. 44 (21): 6435–6448. Bibcode: 2006JPoSA..44.6435C. doi: 10.1002/pola.21761. ISSN 0887-624X.
- ^ Monte, Salvatore J. (1998), Pritchard, Geoffrey (ed.), "Diluents and viscosity modifiers for epoxy resins", Plastics Additives: An A-Z reference, Polymer Science and Technology Series, vol. 1, Dordrecht: Springer Netherlands, pp. 211–216, doi: 10.1007/978-94-011-5862-6_24, ISBN 978-94-011-5862-6, archived from the original on 2022-04-11, retrieved 2022-03-29