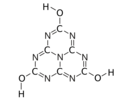
Trihydroxy (
imidic acid)
tautomer
| |
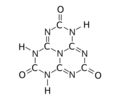
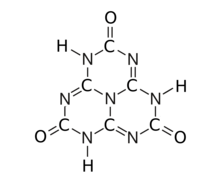 Tricarbonyl (
amide) tautomer
| |
| Names | |
|---|---|
|
IUPAC name
2,4,6,8,10,12,13-heptazatricyclo[7.3.1.05,13]trideca-1,4,8-triene-3,7,11-trione
| |
| Other names
1,3,4,6,7,9,9b-Heptaazaphenalene-2,5,8(1H,3H,6H)-trione; 2,5,8-trihydroxy-s-heptazine; 1,4,7-trihydro-2,5,8-trioxo-s-heptazine
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 34555, 542266 | |
| ChEBI | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H3N7O3 | |
| Molar mass | 221.13 g/mol |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyameluric acid or 2,5,8-trihydroxy-s-heptazine is a chemical compound with formula C
6N
7O
3H
3, usually described as a
heptazine molecule with the
hydrogen atoms replaced by
hydroxyl
groups –OH; or any of its
tautomers.
The substance exists as an equilibrium of 17 tautomers that easily interconvert among each other. Calculations show that the symmetric tri-oxo form (1,4,7-trihydro-2,5,8-trioxo-s-heptazine) is the most stable. [1] Therefore, this compound contains amide groups rather than imidic acids.
In 1834
Justus von Liebig described the compounds that he named
melamine,
melam, and
melon.
[2] In 1835
Leopold Gmelin prepared novel
salts by heating
potassium ferrocyanide with
sulfur); recognizing their connection to the compounds described by Liebig, he named the salts "
hydromelonates" and the corresponding acid "
hydromelonic".
[3] In the following years Liebig prepared the same salts by other methods, such as by fusing
potassium thiocyanate with
antimony trichloride,
[4] and eventually determined the formula C
9N
13H
3 for the acid.
[5]
[6]
Cyameluric acid, H
3O
3C
6N
7 and salts were prepared in 1850 by
W. Henneberg, by treating Gmelin's "hydromelonate" with alkali.
[7]
[6]
The first person to suggest a structure for cyameluric acid was J. Loschmidt, as far back as 1861. His structure was in fact a meta- cyclophane, but it is remarkable since at that time cyclic compounds of any type were not widely recognised. [8] [9]
The correct structure (for the trihydroxy tautomer) was published in 1937 by Linus Pauling and J. H. Sturdivant. [6]

The various tautomeric forms differ in the position of the hydrogen atoms. Each oxygen is connected to one of the corner carbons; it may be bonded to a hydrogen, forming a hydroxy group; or may have a double bond to the carbon, in which case the hydrogen is bonded to one of several adjacent nitrogen atoms. [1]
The trihydroxy tautomer is one of several that have more than one planar conformational isomer. In this case, there is a symmetric one, with all three hydroxyls bent in the same direction around the ring, and an asymmetric one, with one of them bent in the opposite direction compared to the other two. Calculations show that the symmetric form 1,4,7-trihydro-2,5,8-trioxo is the most stable. The energy of the asymmetric 1,3,7-trihydro-2,5,8-trioxo form is estimated to be 5.61 kcal/ mol higher, and that of the two conformations of the trihydroxy form are 19.84 (symmetric) and 20.18 (asymmetric) kcal/mol higher. [1]
- Melem, 2,5,8-triamino-heptazine.
- ^ a b c Alkorta, Ibon; Jagerovic, Nadine; Elguero, José (2004-03-06). Muthyala, Ramaiah (ed.). "Theoretical study of cyameluric acid and related compounds". Arkivoc. 2004 (4): 130–136. doi: 10.3998/ark.5550190.0005.415. hdl: 2027/spo.5550190.0005.415. ISSN 1551-7012.
- ^ Liebig, Justus (1834). "Analyse der Harnsäure". Annalen der Pharmacie. 10 (1): 47–48. doi: 10.1002/jlac.18340100103. ISSN 0365-5490.
- ^ Gmelin, Leopold (1835). "Ueber einige Verbindungen des Melon's". Annalen der Pharmacie. 15 (3): 252–258. doi: 10.1002/jlac.18350150306. ISSN 0365-5490.
- ^ Liebig, Justus (1844). "Ueber Mellon und Mellonverbindungen". Justus Liebigs Annalen der Chemie. 50 (3): 337–363. doi: 10.1002/jlac.18440500302. ISSN 0075-4617.
- ^ Liebig, Justus (1855). "Ueber die Constitution der Mellonverbindungen". Justus Liebigs Annalen der Chemie. 95 (3): 257–282. doi: 10.1002/jlac.18550950302. ISSN 0075-4617.
- ^ a b c Pauling, Linus; Sturdivant, J. H. (December 1937). "The Structure of Cyameluric Acid, Hydromelonic Acid and Related Substances". Proceedings of the National Academy of Sciences. 23 (12): 615–620. doi: 10.1073/pnas.23.12.615. ISSN 0027-8424. PMC 1077007. PMID 16577829.
- ^ Henneberg, W. (1850). "Ueber einige Zersetzungsproducte des Mellonkaliums". Justus Liebigs Annalen der Chemie. 73 (2): 228–255. doi: 10.1002/jlac.18500730217. ISSN 0075-4617.
- ^ Henrich, F. (1913-11-15). "Konstitutionsformeln der organischen Chemie in graphischer Darstellung". Zeitschrift für Elektrochemie und angewandte physikalische Chemie. 19 (22). doi: 10.1002/bbpc.191300036. ISSN 0372-8323.
- ^ Henry S. Rzepa, Joseph Loschmidt: Structural formulae, 1861. Accessed on 2009-06-30.
 Trihydroxy (
imidic acid)
tautomer
| |
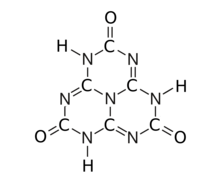 Tricarbonyl (
amide) tautomer
| |
| Names | |
|---|---|
|
IUPAC name
2,4,6,8,10,12,13-heptazatricyclo[7.3.1.05,13]trideca-1,4,8-triene-3,7,11-trione
| |
| Other names
1,3,4,6,7,9,9b-Heptaazaphenalene-2,5,8(1H,3H,6H)-trione; 2,5,8-trihydroxy-s-heptazine; 1,4,7-trihydro-2,5,8-trioxo-s-heptazine
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 34555, 542266 | |
| ChEBI | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H3N7O3 | |
| Molar mass | 221.13 g/mol |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyameluric acid or 2,5,8-trihydroxy-s-heptazine is a chemical compound with formula C
6N
7O
3H
3, usually described as a
heptazine molecule with the
hydrogen atoms replaced by
hydroxyl
groups –OH; or any of its
tautomers.
The substance exists as an equilibrium of 17 tautomers that easily interconvert among each other. Calculations show that the symmetric tri-oxo form (1,4,7-trihydro-2,5,8-trioxo-s-heptazine) is the most stable. [1] Therefore, this compound contains amide groups rather than imidic acids.
In 1834
Justus von Liebig described the compounds that he named
melamine,
melam, and
melon.
[2] In 1835
Leopold Gmelin prepared novel
salts by heating
potassium ferrocyanide with
sulfur); recognizing their connection to the compounds described by Liebig, he named the salts "
hydromelonates" and the corresponding acid "
hydromelonic".
[3] In the following years Liebig prepared the same salts by other methods, such as by fusing
potassium thiocyanate with
antimony trichloride,
[4] and eventually determined the formula C
9N
13H
3 for the acid.
[5]
[6]
Cyameluric acid, H
3O
3C
6N
7 and salts were prepared in 1850 by
W. Henneberg, by treating Gmelin's "hydromelonate" with alkali.
[7]
[6]
The first person to suggest a structure for cyameluric acid was J. Loschmidt, as far back as 1861. His structure was in fact a meta- cyclophane, but it is remarkable since at that time cyclic compounds of any type were not widely recognised. [8] [9]
The correct structure (for the trihydroxy tautomer) was published in 1937 by Linus Pauling and J. H. Sturdivant. [6]

The various tautomeric forms differ in the position of the hydrogen atoms. Each oxygen is connected to one of the corner carbons; it may be bonded to a hydrogen, forming a hydroxy group; or may have a double bond to the carbon, in which case the hydrogen is bonded to one of several adjacent nitrogen atoms. [1]
The trihydroxy tautomer is one of several that have more than one planar conformational isomer. In this case, there is a symmetric one, with all three hydroxyls bent in the same direction around the ring, and an asymmetric one, with one of them bent in the opposite direction compared to the other two. Calculations show that the symmetric form 1,4,7-trihydro-2,5,8-trioxo is the most stable. The energy of the asymmetric 1,3,7-trihydro-2,5,8-trioxo form is estimated to be 5.61 kcal/ mol higher, and that of the two conformations of the trihydroxy form are 19.84 (symmetric) and 20.18 (asymmetric) kcal/mol higher. [1]
- Melem, 2,5,8-triamino-heptazine.
- ^ a b c Alkorta, Ibon; Jagerovic, Nadine; Elguero, José (2004-03-06). Muthyala, Ramaiah (ed.). "Theoretical study of cyameluric acid and related compounds". Arkivoc. 2004 (4): 130–136. doi: 10.3998/ark.5550190.0005.415. hdl: 2027/spo.5550190.0005.415. ISSN 1551-7012.
- ^ Liebig, Justus (1834). "Analyse der Harnsäure". Annalen der Pharmacie. 10 (1): 47–48. doi: 10.1002/jlac.18340100103. ISSN 0365-5490.
- ^ Gmelin, Leopold (1835). "Ueber einige Verbindungen des Melon's". Annalen der Pharmacie. 15 (3): 252–258. doi: 10.1002/jlac.18350150306. ISSN 0365-5490.
- ^ Liebig, Justus (1844). "Ueber Mellon und Mellonverbindungen". Justus Liebigs Annalen der Chemie. 50 (3): 337–363. doi: 10.1002/jlac.18440500302. ISSN 0075-4617.
- ^ Liebig, Justus (1855). "Ueber die Constitution der Mellonverbindungen". Justus Liebigs Annalen der Chemie. 95 (3): 257–282. doi: 10.1002/jlac.18550950302. ISSN 0075-4617.
- ^ a b c Pauling, Linus; Sturdivant, J. H. (December 1937). "The Structure of Cyameluric Acid, Hydromelonic Acid and Related Substances". Proceedings of the National Academy of Sciences. 23 (12): 615–620. doi: 10.1073/pnas.23.12.615. ISSN 0027-8424. PMC 1077007. PMID 16577829.
- ^ Henneberg, W. (1850). "Ueber einige Zersetzungsproducte des Mellonkaliums". Justus Liebigs Annalen der Chemie. 73 (2): 228–255. doi: 10.1002/jlac.18500730217. ISSN 0075-4617.
- ^ Henrich, F. (1913-11-15). "Konstitutionsformeln der organischen Chemie in graphischer Darstellung". Zeitschrift für Elektrochemie und angewandte physikalische Chemie. 19 (22). doi: 10.1002/bbpc.191300036. ISSN 0372-8323.
- ^ Henry S. Rzepa, Joseph Loschmidt: Structural formulae, 1861. Accessed on 2009-06-30.