
| |
 Crystal structure
| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
PubChem
CID
|
|
| |
| |
| Properties | |
| Cl3Cm | |
| Molar mass | 353 g·mol−1 |
| Appearance | White solid (anhydrous) Light green solid (hydrate) |
| Melting point | 695 °C (1,283 °F; 968 K)[ citation needed] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Curium(III) chloride is the chemical compound with the formula CmCl3.
Structure
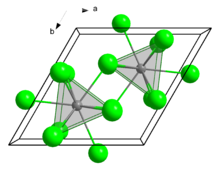
Curium(III) chloride has a 9 coordinate tricapped trigonal prismatic geometry. [1]
Synthesis
Curium(III) chloride can be obtained from the reaction of hydrogen chloride gas with curium dioxide, curium(III) oxide, or curium(III) oxychloride at a temperature of 400-600 °C:
- CmOCl + 2HCl → CmCl3 + H2O
It can also be obtained from the dissolution of metallic curium in dilute hydrochloric acid: [2]
- 2Cm + 6HCl → 2CmCl3 + 3H2
This method has a number of disadvantages associated with the ongoing processes of hydrolysis and hydration of the resulting compound in an aqueous solution, making it problematic to obtain a pure product using this reaction.
It can be obtained from the reaction of curium nitride with cadmium chloride: [3]
- 2 CmN + 3 CdCl2 → 2 CmCl3 + Cd3N2
References
-
^ Greenwood, N. N.; Earnshaw, A. (1997). Chemistry of the Elements (2nd ed.). Butterworth, UK. p. 1270.
{{ cite book}}: CS1 maint: location missing publisher ( link) - ^ Wallmann, J. C.; Fuger, J.; Peterson, J. R.; Green, J. L. (1 November 1967). "Crystal structure and lattice parameters of curium trichloride". Journal of Inorganic and Nuclear Chemistry. 29 (11): 2745–2751. doi: 10.1016/0022-1902(67)80013-7. ISSN 0022-1902. S2CID 97334114. Retrieved 3 July 2023.
- ^ Hayashi, Hirokazu; Takano, Masahide; Otobe, Haruyoshi; Koyama, Tadafumi (July 2013). "Syntheses and thermal analyses of curium trichloride". Journal of Radioanalytical and Nuclear Chemistry. 297 (1): 139–144. doi: 10.1007/s10967-012-2413-7. S2CID 95792512.

| |
 Crystal structure
| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
PubChem
CID
|
|
| |
| |
| Properties | |
| Cl3Cm | |
| Molar mass | 353 g·mol−1 |
| Appearance | White solid (anhydrous) Light green solid (hydrate) |
| Melting point | 695 °C (1,283 °F; 968 K)[ citation needed] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Curium(III) chloride is the chemical compound with the formula CmCl3.
Structure
Curium(III) chloride has a 9 coordinate tricapped trigonal prismatic geometry. [1]
Synthesis
Curium(III) chloride can be obtained from the reaction of hydrogen chloride gas with curium dioxide, curium(III) oxide, or curium(III) oxychloride at a temperature of 400-600 °C:
- CmOCl + 2HCl → CmCl3 + H2O
It can also be obtained from the dissolution of metallic curium in dilute hydrochloric acid: [2]
- 2Cm + 6HCl → 2CmCl3 + 3H2
This method has a number of disadvantages associated with the ongoing processes of hydrolysis and hydration of the resulting compound in an aqueous solution, making it problematic to obtain a pure product using this reaction.
It can be obtained from the reaction of curium nitride with cadmium chloride: [3]
- 2 CmN + 3 CdCl2 → 2 CmCl3 + Cd3N2
References
-
^ Greenwood, N. N.; Earnshaw, A. (1997). Chemistry of the Elements (2nd ed.). Butterworth, UK. p. 1270.
{{ cite book}}: CS1 maint: location missing publisher ( link) - ^ Wallmann, J. C.; Fuger, J.; Peterson, J. R.; Green, J. L. (1 November 1967). "Crystal structure and lattice parameters of curium trichloride". Journal of Inorganic and Nuclear Chemistry. 29 (11): 2745–2751. doi: 10.1016/0022-1902(67)80013-7. ISSN 0022-1902. S2CID 97334114. Retrieved 3 July 2023.
- ^ Hayashi, Hirokazu; Takano, Masahide; Otobe, Haruyoshi; Koyama, Tadafumi (July 2013). "Syntheses and thermal analyses of curium trichloride". Journal of Radioanalytical and Nuclear Chemistry. 297 (1): 139–144. doi: 10.1007/s10967-012-2413-7. S2CID 95792512.