| |||
| Names | |||
|---|---|---|---|
IUPAC name
| |||
| Identifiers | |||
| |||
3D model (
JSmol)
|
| ||
| EC Number |
| ||
PubChem
CID
|
|||
| UNII |
| ||
| |||
| |||
| Properties | |||
| C15H24 | |||
| Molar mass | 204.357 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
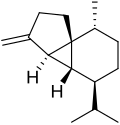
Cubebenes are a pair of chemical compounds, classified as sesquiterpenes, first isolated from Piper cubeba berries, known as cubebs.
The volatile oil from the distillation of cubebs is a pale green or blue-yellow viscous liquid with a warm woody, slightly camphoraceous odor [1] consisting of cubebene which comes in two forms, α- and β-cubebene, both with the molecular formula C15H24. [2] They differ only in the position of a double bond which is endocyclic (part of the five-membered ring) in α-cubebene, but exocyclic in β-cubebene. [3]
References
- ^ Lawless, Julia (1995), The Illustrated Encyclopedia of Essential Oils: The Complete Guide to the Use of Oils in Aromatherapy and Herbalism, Element Books, ISBN 978-1-85230-721-9
- ^ This article incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). " Cubebs". Encyclopædia Britannica. Vol. 7 (11th ed.). Cambridge University Press. p. 607.
- ^ Ohta, Yoshimoto; Sakai, Tsutomu; Hirose, Toshio (1966). "Sesquiterpene hydrocarbons from the oil of cubeb α-cubebene and β-cubebene". Tetrahedron Letters. 7 (51): 6365–6370. doi: 10.1016/S0040-4039(00)70179-1.
| |||
| Names | |||
|---|---|---|---|
IUPAC name
| |||
| Identifiers | |||
| |||
3D model (
JSmol)
|
| ||
| EC Number |
| ||
PubChem
CID
|
|||
| UNII |
| ||
| |||
| |||
| Properties | |||
| C15H24 | |||
| Molar mass | 204.357 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Cubebenes are a pair of chemical compounds, classified as sesquiterpenes, first isolated from Piper cubeba berries, known as cubebs.
The volatile oil from the distillation of cubebs is a pale green or blue-yellow viscous liquid with a warm woody, slightly camphoraceous odor [1] consisting of cubebene which comes in two forms, α- and β-cubebene, both with the molecular formula C15H24. [2] They differ only in the position of a double bond which is endocyclic (part of the five-membered ring) in α-cubebene, but exocyclic in β-cubebene. [3]
References
- ^ Lawless, Julia (1995), The Illustrated Encyclopedia of Essential Oils: The Complete Guide to the Use of Oils in Aromatherapy and Herbalism, Element Books, ISBN 978-1-85230-721-9
- ^ This article incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). " Cubebs". Encyclopædia Britannica. Vol. 7 (11th ed.). Cambridge University Press. p. 607.
- ^ Ohta, Yoshimoto; Sakai, Tsutomu; Hirose, Toshio (1966). "Sesquiterpene hydrocarbons from the oil of cubeb α-cubebene and β-cubebene". Tetrahedron Letters. 7 (51): 6365–6370. doi: 10.1016/S0040-4039(00)70179-1.