 | |
| Clinical data | |
|---|---|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.164.003 |
| Chemical and physical data | |
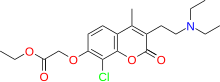
| Formula | C20H26ClNO5 |
| Molar mass | 395.88 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Cloricromen is a platelet aggregation inhibitor. [1] Coronary vasodilator.
Synthesis

Base catalyzed alkylation of Ethyl Acetoacetate [141-97-9] (1) with 2-Chlorotriethylamine [100-35-6] (2) gives ethyl 2-(2-diethylaminoethyl)acetoacetate [23999-02-2] (3). Disulfonation of resorcinol [108-46-3] (4) with 96% sulfuric acid gives 4,6-dihydroxybenzene-1,3-disulfonic Acid [17724-11-7] (5). This is reacted with potassium chlorate [3811-04-9] in acid and alkalized to give 5-Chloro-4,6-dihydroxybenzene-1,3-disulfonic acid, PC20077643 (6). Hydrolysis of the sulfonate groups in dilute acid occurs to give 2-chlororesorcinol [6201-65-6] (7). The acid catalyzed condensation between (3) and (7) led to 8-chloro-3-[2-(diethylamino)ethyl]-7-hydroxy-4-methylchromen-2-one, PC20144545 [70665-54-2] (8). Ether formation with ethyl bromoacetate [105-36-2] (9) completed the synthesis of Carbocromen (10).
See also
- Carbocromen is ditto sans chlorine.
References
- ^ Orefice G, Grasso A, Fazio N, Del Vecchio G, Volpe G, Coppola M, D'Alessio A, Carrieri PB (1994). "No effect of cloricromen on some coagulation parameters in patients with ischaemic cerebrovascular disease". The Journal of International Medical Research. 22 (5): 287–91. doi: 10.1177/030006059402200506. PMID 7867874. S2CID 36081514.
- ^ Castaer, J.; Hillier, K.; Serradell, MN; Blancafort, P.; AD-6. Drugs Fut 1979, 4, 10, 701.
- ^ BE871315 idem Francesco Della Valle, U.S. patent 4,452,811 (1984 to Fidia S.P.A.).
- ^ Weizmann, Ch.; Bergmann, E.; Sulzbacher, M. (1950). "THE USE, FOR CONDENSATION REACTIONS, OF POTASSIUM HYDROXIDE IN SOLVENTS OF THE ACETAL TYPE". The Journal of Organic Chemistry. 15 (5): 918–929. doi:10.1021/jo01151a002.
- ^ Francesco D. Valle & Aurelio Romeo, U.S. patent 4,704,469 (1987 to Fidia Farmaceutici SpA).
- ^ Wanzlick, H., Mohrmann, S. (August 1963). "Notiz über einen neuen Weg zum 2‐Chlor‐resorcin". Chemische Berichte. 96 (8): 2257–2258. doi:10.1002/cber.19630960843.
 | |
| Clinical data | |
|---|---|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.164.003 |
| Chemical and physical data | |
| Formula | C20H26ClNO5 |
| Molar mass | 395.88 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Cloricromen is a platelet aggregation inhibitor. [1] Coronary vasodilator.
Synthesis

Base catalyzed alkylation of Ethyl Acetoacetate [141-97-9] (1) with 2-Chlorotriethylamine [100-35-6] (2) gives ethyl 2-(2-diethylaminoethyl)acetoacetate [23999-02-2] (3). Disulfonation of resorcinol [108-46-3] (4) with 96% sulfuric acid gives 4,6-dihydroxybenzene-1,3-disulfonic Acid [17724-11-7] (5). This is reacted with potassium chlorate [3811-04-9] in acid and alkalized to give 5-Chloro-4,6-dihydroxybenzene-1,3-disulfonic acid, PC20077643 (6). Hydrolysis of the sulfonate groups in dilute acid occurs to give 2-chlororesorcinol [6201-65-6] (7). The acid catalyzed condensation between (3) and (7) led to 8-chloro-3-[2-(diethylamino)ethyl]-7-hydroxy-4-methylchromen-2-one, PC20144545 [70665-54-2] (8). Ether formation with ethyl bromoacetate [105-36-2] (9) completed the synthesis of Carbocromen (10).
See also
- Carbocromen is ditto sans chlorine.
References
- ^ Orefice G, Grasso A, Fazio N, Del Vecchio G, Volpe G, Coppola M, D'Alessio A, Carrieri PB (1994). "No effect of cloricromen on some coagulation parameters in patients with ischaemic cerebrovascular disease". The Journal of International Medical Research. 22 (5): 287–91. doi: 10.1177/030006059402200506. PMID 7867874. S2CID 36081514.
- ^ Castaer, J.; Hillier, K.; Serradell, MN; Blancafort, P.; AD-6. Drugs Fut 1979, 4, 10, 701.
- ^ BE871315 idem Francesco Della Valle, U.S. patent 4,452,811 (1984 to Fidia S.P.A.).
- ^ Weizmann, Ch.; Bergmann, E.; Sulzbacher, M. (1950). "THE USE, FOR CONDENSATION REACTIONS, OF POTASSIUM HYDROXIDE IN SOLVENTS OF THE ACETAL TYPE". The Journal of Organic Chemistry. 15 (5): 918–929. doi:10.1021/jo01151a002.
- ^ Francesco D. Valle & Aurelio Romeo, U.S. patent 4,704,469 (1987 to Fidia Farmaceutici SpA).
- ^ Wanzlick, H., Mohrmann, S. (August 1963). "Notiz über einen neuen Weg zum 2‐Chlor‐resorcin". Chemische Berichte. 96 (8): 2257–2258. doi:10.1002/cber.19630960843.