| CSTB | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | CSTB, CST6, EPM1, EPM1A, PME, STFB, ULD, Cystatin B, CPI-B | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 601145; MGI: 109514; HomoloGene: 79; GeneCards: CSTB; OMA: CSTB - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Cystatin-B is a protein that in humans is encoded by the CSTB gene. [5] [6]
The cystatin superfamily encompasses proteins that contain multiple cystatin-like sequences. Some of the members are active cysteine protease inhibitors, while others have lost or perhaps never acquired this inhibitory activity. There are three inhibitory families in the superfamily, including the type 1 cystatins (stefins), type 2 cystatins and kininogens. This gene encodes a stefin that functions as an intracellular cysteine protease inhibitor. The protein is able to form a dimer stabilized by noncovalent forces, inhibiting papain and cathepsins L, H and B. The protein is thought to play a role in protecting against the proteases leaking from lysosomes. Evidence indicates that mutations in this gene are responsible for the primary defects in patients with Unverricht–Lundborg disease, a form of progressive myoclonic epilepsy (EPM1). [6]
Interactions
Cystatin B has been shown to interact with Cathepsin B. [7] [8]
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000160213 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000005054 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Pennacchio LA, Lehesjoki AE, Stone NE, Willour VL, Virtaneva K, Miao J, D'Amato E, Ramirez L, Faham M, Koskiniemi M, Warrington JA, Norio R, de la Chapelle A, Cox DR, Myers RM (Apr 1996). "Mutations in the gene encoding cystatin B in progressive myoclonus epilepsy (EPM1)". Science. 271 (5256): 1731–4. Bibcode: 1996Sci...271.1731P. doi: 10.1126/science.271.5256.1731. PMID 8596935. S2CID 84361089.
- ^ a b "Entrez Gene: CSTB cystatin B (stefin B)".
- ^ Pavlova A, Björk Ingemar (Sep 2003). "Grafting of features of cystatins C or B into the N-terminal region or second binding loop of cystatin A (stefin A) substantially enhances inhibition of cysteine proteinases". Biochemistry. 42 (38). United States: 11326–33. doi: 10.1021/bi030119v. ISSN 0006-2960. PMID 14503883.
- ^ Pol E, Björk I (Sep 2001). "Role of the single cysteine residue, Cys 3, of human and bovine cystatin B (stefin B) in the inhibition of cysteine proteinases". Protein Sci. 10 (9). United States: 1729–38. doi: 10.1110/ps.11901. ISSN 0961-8368. PMC 2253190. PMID 11514663.
Further reading
- Turk V, Bode W (1991). "The cystatins: protein inhibitors of cysteine proteinases". FEBS Lett. 285 (2): 213–9. doi: 10.1016/0014-5793(91)80804-C. PMID 1855589. S2CID 40444629.
- Järvinen M, Rinne A, Hopsu-Havu VK (1988). "Human cystatins in normal and diseased tissues--a review". Acta Histochem. 82 (1): 5–18. doi: 10.1016/s0065-1281(87)80043-0. PMID 3122506.
- Brown WM, Dziegielewska KM (1997). "Friends and relations of the cystatin superfamily--new members and their evolution". Protein Sci. 6 (1): 5–12. doi: 10.1002/pro.5560060102. PMC 2143511. PMID 9007972.
- Kos J, Lah TT (1998). "Cysteine proteinases and their endogenous inhibitors: target proteins for prognosis, diagnosis and therapy in cancer (review)". Oncol. Rep. 5 (6): 1349–61. doi: 10.3892/or.5.6.1349. PMID 9769367.
- Stubbs MT, Laber B, Bode W, et al. (1990). "The refined 2.4 A X-ray crystal structure of recombinant human stefin B in complex with the cysteine proteinase papain: a novel type of proteinase inhibitor interaction". EMBO J. 9 (6): 1939–47. doi: 10.1002/j.1460-2075.1990.tb08321.x. PMC 551902. PMID 2347312.
- Jerala R, Trstenjak M, Lenarcic B, Turk V (1988). "Cloning a synthetic gene for human stefin B and its expression in E. coli". FEBS Lett. 239 (1): 41–4. doi: 10.1016/0014-5793(88)80541-6. PMID 3053245. S2CID 33859701.
- Lenarcic B, Kos J, Dolenc I, et al. (1988). "Cathepsin D inactivates cysteine proteinase inhibitors, cystatins". Biochem. Biophys. Res. Commun. 154 (2): 765–72. doi: 10.1016/0006-291X(88)90206-9. PMID 3261170.
- Ritonja A, Machleidt W, Barrett AJ (1985). "Amino acid sequence of the intracellular cysteine proteinase inhibitor cystatin B from human liver". Biochem. Biophys. Res. Commun. 131 (3): 1187–92. doi: 10.1016/0006-291X(85)90216-5. PMID 3902020.
- Spiess E, Brüning A, Gack S, et al. (1994). "Cathepsin B activity in human lung tumor cell lines: ultrastructural localization, pH sensitivity, and inhibitor status at the cellular level". J. Histochem. Cytochem. 42 (7): 917–29. doi: 10.1177/42.7.8014475. PMID 8014475. S2CID 24509312.
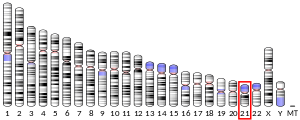
- Lehesjoki AE, Koskiniemi M, Norio R, et al. (1993). "Localization of the EPM1 gene for progressive myoclonus epilepsy on chromosome 21: linkage disequilibrium allows high resolution mapping". Hum. Mol. Genet. 2 (8): 1229–34. doi: 10.1093/hmg/2.8.1229. PMID 8104628.
- Pennacchio LA, Myers RM (1997). "Isolation and characterization of the mouse cystatin B gene". Genome Res. 6 (11): 1103–9. doi: 10.1101/gr.6.11.1103. PMID 8938434.
- Lalioti MD, Mirotsou M, Buresi C, et al. (1997). "Identification of mutations in cystatin B, the gene responsible for the Unverricht-Lundborg type of progressive myoclonus epilepsy (EPM1)". Am. J. Hum. Genet. 60 (2): 342–51. PMC 1712389. PMID 9012407.
- Lafrenière RG, Rochefort DL, Chrétien N, et al. (1997). "Unstable insertion in the 5' flanking region of the cystatin B gene is the most common mutation in progressive myoclonus epilepsy type 1, EPM1". Nat. Genet. 15 (3): 298–302. doi: 10.1038/ng0397-298. PMID 9054946. S2CID 21180258.
- Virtaneva K, D'Amato E, Miao J, et al. (1997). "Unstable minisatellite expansion causing recessively inherited myoclonus epilepsy, EPM1". Nat. Genet. 15 (4): 393–6. doi: 10.1038/ng0497-393. PMID 9090386. S2CID 24971949.
- Bespalova IN, Adkins S, Pranzatelli M, Burmeister M (1997). "Novel cystatin B mutation and diagnostic PCR assay in an Unverricht-Lundborg progressive myoclonus epilepsy patient" (PDF). Am. J. Med. Genet. 74 (5): 467–71. doi: 10.1002/(SICI)1096-8628(19970919)74:5<467::AID-AJMG1>3.0.CO;2-L. hdl: 2027.42/38271. PMID 9342192.
External links
- GeneReviews/NCBI/NIH/UW entry on Unverricht-Lundborg Disease or EPM1
- The MEROPS online database for peptidases and their inhibitors: I25.003
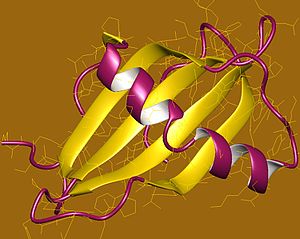
- PDBe-KB provides an overview of all the structure information available in the PDB for Human Cystatin-B

| CSTB | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | CSTB, CST6, EPM1, EPM1A, PME, STFB, ULD, Cystatin B, CPI-B | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 601145; MGI: 109514; HomoloGene: 79; GeneCards: CSTB; OMA: CSTB - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Cystatin-B is a protein that in humans is encoded by the CSTB gene. [5] [6]
The cystatin superfamily encompasses proteins that contain multiple cystatin-like sequences. Some of the members are active cysteine protease inhibitors, while others have lost or perhaps never acquired this inhibitory activity. There are three inhibitory families in the superfamily, including the type 1 cystatins (stefins), type 2 cystatins and kininogens. This gene encodes a stefin that functions as an intracellular cysteine protease inhibitor. The protein is able to form a dimer stabilized by noncovalent forces, inhibiting papain and cathepsins L, H and B. The protein is thought to play a role in protecting against the proteases leaking from lysosomes. Evidence indicates that mutations in this gene are responsible for the primary defects in patients with Unverricht–Lundborg disease, a form of progressive myoclonic epilepsy (EPM1). [6]
Interactions
Cystatin B has been shown to interact with Cathepsin B. [7] [8]
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000160213 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000005054 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Pennacchio LA, Lehesjoki AE, Stone NE, Willour VL, Virtaneva K, Miao J, D'Amato E, Ramirez L, Faham M, Koskiniemi M, Warrington JA, Norio R, de la Chapelle A, Cox DR, Myers RM (Apr 1996). "Mutations in the gene encoding cystatin B in progressive myoclonus epilepsy (EPM1)". Science. 271 (5256): 1731–4. Bibcode: 1996Sci...271.1731P. doi: 10.1126/science.271.5256.1731. PMID 8596935. S2CID 84361089.
- ^ a b "Entrez Gene: CSTB cystatin B (stefin B)".
- ^ Pavlova A, Björk Ingemar (Sep 2003). "Grafting of features of cystatins C or B into the N-terminal region or second binding loop of cystatin A (stefin A) substantially enhances inhibition of cysteine proteinases". Biochemistry. 42 (38). United States: 11326–33. doi: 10.1021/bi030119v. ISSN 0006-2960. PMID 14503883.
- ^ Pol E, Björk I (Sep 2001). "Role of the single cysteine residue, Cys 3, of human and bovine cystatin B (stefin B) in the inhibition of cysteine proteinases". Protein Sci. 10 (9). United States: 1729–38. doi: 10.1110/ps.11901. ISSN 0961-8368. PMC 2253190. PMID 11514663.
Further reading
- Turk V, Bode W (1991). "The cystatins: protein inhibitors of cysteine proteinases". FEBS Lett. 285 (2): 213–9. doi: 10.1016/0014-5793(91)80804-C. PMID 1855589. S2CID 40444629.
- Järvinen M, Rinne A, Hopsu-Havu VK (1988). "Human cystatins in normal and diseased tissues--a review". Acta Histochem. 82 (1): 5–18. doi: 10.1016/s0065-1281(87)80043-0. PMID 3122506.
- Brown WM, Dziegielewska KM (1997). "Friends and relations of the cystatin superfamily--new members and their evolution". Protein Sci. 6 (1): 5–12. doi: 10.1002/pro.5560060102. PMC 2143511. PMID 9007972.
- Kos J, Lah TT (1998). "Cysteine proteinases and their endogenous inhibitors: target proteins for prognosis, diagnosis and therapy in cancer (review)". Oncol. Rep. 5 (6): 1349–61. doi: 10.3892/or.5.6.1349. PMID 9769367.
- Stubbs MT, Laber B, Bode W, et al. (1990). "The refined 2.4 A X-ray crystal structure of recombinant human stefin B in complex with the cysteine proteinase papain: a novel type of proteinase inhibitor interaction". EMBO J. 9 (6): 1939–47. doi: 10.1002/j.1460-2075.1990.tb08321.x. PMC 551902. PMID 2347312.
- Jerala R, Trstenjak M, Lenarcic B, Turk V (1988). "Cloning a synthetic gene for human stefin B and its expression in E. coli". FEBS Lett. 239 (1): 41–4. doi: 10.1016/0014-5793(88)80541-6. PMID 3053245. S2CID 33859701.
- Lenarcic B, Kos J, Dolenc I, et al. (1988). "Cathepsin D inactivates cysteine proteinase inhibitors, cystatins". Biochem. Biophys. Res. Commun. 154 (2): 765–72. doi: 10.1016/0006-291X(88)90206-9. PMID 3261170.
- Ritonja A, Machleidt W, Barrett AJ (1985). "Amino acid sequence of the intracellular cysteine proteinase inhibitor cystatin B from human liver". Biochem. Biophys. Res. Commun. 131 (3): 1187–92. doi: 10.1016/0006-291X(85)90216-5. PMID 3902020.
- Spiess E, Brüning A, Gack S, et al. (1994). "Cathepsin B activity in human lung tumor cell lines: ultrastructural localization, pH sensitivity, and inhibitor status at the cellular level". J. Histochem. Cytochem. 42 (7): 917–29. doi: 10.1177/42.7.8014475. PMID 8014475. S2CID 24509312.
- Lehesjoki AE, Koskiniemi M, Norio R, et al. (1993). "Localization of the EPM1 gene for progressive myoclonus epilepsy on chromosome 21: linkage disequilibrium allows high resolution mapping". Hum. Mol. Genet. 2 (8): 1229–34. doi: 10.1093/hmg/2.8.1229. PMID 8104628.
- Pennacchio LA, Myers RM (1997). "Isolation and characterization of the mouse cystatin B gene". Genome Res. 6 (11): 1103–9. doi: 10.1101/gr.6.11.1103. PMID 8938434.
- Lalioti MD, Mirotsou M, Buresi C, et al. (1997). "Identification of mutations in cystatin B, the gene responsible for the Unverricht-Lundborg type of progressive myoclonus epilepsy (EPM1)". Am. J. Hum. Genet. 60 (2): 342–51. PMC 1712389. PMID 9012407.
- Lafrenière RG, Rochefort DL, Chrétien N, et al. (1997). "Unstable insertion in the 5' flanking region of the cystatin B gene is the most common mutation in progressive myoclonus epilepsy type 1, EPM1". Nat. Genet. 15 (3): 298–302. doi: 10.1038/ng0397-298. PMID 9054946. S2CID 21180258.
- Virtaneva K, D'Amato E, Miao J, et al. (1997). "Unstable minisatellite expansion causing recessively inherited myoclonus epilepsy, EPM1". Nat. Genet. 15 (4): 393–6. doi: 10.1038/ng0497-393. PMID 9090386. S2CID 24971949.
- Bespalova IN, Adkins S, Pranzatelli M, Burmeister M (1997). "Novel cystatin B mutation and diagnostic PCR assay in an Unverricht-Lundborg progressive myoclonus epilepsy patient" (PDF). Am. J. Med. Genet. 74 (5): 467–71. doi: 10.1002/(SICI)1096-8628(19970919)74:5<467::AID-AJMG1>3.0.CO;2-L. hdl: 2027.42/38271. PMID 9342192.
External links
- GeneReviews/NCBI/NIH/UW entry on Unverricht-Lundborg Disease or EPM1
- The MEROPS online database for peptidases and their inhibitors: I25.003
- PDBe-KB provides an overview of all the structure information available in the PDB for Human Cystatin-B