| |
| Names | |
|---|---|
|
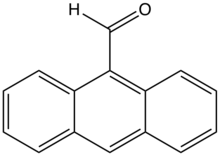
Preferred IUPAC name
Anthracene-9-carbaldehyde | |
| Other names
9-Anthraldehyde, 9-Anthracenaldehyde, Anthracene-9-carbaldehyde
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 639167 | |
| ChemSpider | |
| ECHA InfoCard | 100.010.349 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C15H10O | |
| Molar mass | 206.244 g·mol−1 |
| Appearance | yellow solid |
| Melting point | 104 °C (219 °F; 377 K) |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H302, H312, H315, H319, H332, H335 | |
| P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P312, P304+P340, P305+P351+P338, P312, P321, P322, P330, P332+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Anthracene-9-carbaldehyde is the most common monoaldehyde derivative of anthracene. It is a yellow solid that is soluble in common organic solvents. It is prepared by Vilsmeier formylation of anthracene. [1] [2] The compound is also used as a building block for supramolecular assemblies. [3] Hydrogenation of 9-anthracenecarboxaldehyde gives 9-anthracenemethanol.
- ^ Fieser, L. F.; Hartwell, J. L.; Jones, J. E.; Wood, J. H.; Bost, R. W. (1940). "9-Anthraldehyde; 2-Ethoxy-1-Naphthaldehyde". Organic Syntheses. 20: 11. doi: 10.15227/orgsyn.020.0011.
- ^ Jones, Gurnos; Stanforth, Stephen P. (1996). "The Vilsmeier Reaction of Fully Conjugated Carbocycles and Heterocycles". Organic Reactions. pp. 1–330. doi: 10.1002/0471264180.or049.01. ISBN 0471264180.
- ^ Lindsey, Jonathan S.; Wagner, Richard W. (1989). "Investigation of the Synthesis of ortho-Substituted Tetraphenylporphyrins". The Journal of Organic Chemistry. 54 (4): 828–836. doi: 10.1021/jo00265a021.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
Anthracene-9-carbaldehyde | |
| Other names
9-Anthraldehyde, 9-Anthracenaldehyde, Anthracene-9-carbaldehyde
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 639167 | |
| ChemSpider | |
| ECHA InfoCard | 100.010.349 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C15H10O | |
| Molar mass | 206.244 g·mol−1 |
| Appearance | yellow solid |
| Melting point | 104 °C (219 °F; 377 K) |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H302, H312, H315, H319, H332, H335 | |
| P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P312, P304+P340, P305+P351+P338, P312, P321, P322, P330, P332+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Anthracene-9-carbaldehyde is the most common monoaldehyde derivative of anthracene. It is a yellow solid that is soluble in common organic solvents. It is prepared by Vilsmeier formylation of anthracene. [1] [2] The compound is also used as a building block for supramolecular assemblies. [3] Hydrogenation of 9-anthracenecarboxaldehyde gives 9-anthracenemethanol.
- ^ Fieser, L. F.; Hartwell, J. L.; Jones, J. E.; Wood, J. H.; Bost, R. W. (1940). "9-Anthraldehyde; 2-Ethoxy-1-Naphthaldehyde". Organic Syntheses. 20: 11. doi: 10.15227/orgsyn.020.0011.
- ^ Jones, Gurnos; Stanforth, Stephen P. (1996). "The Vilsmeier Reaction of Fully Conjugated Carbocycles and Heterocycles". Organic Reactions. pp. 1–330. doi: 10.1002/0471264180.or049.01. ISBN 0471264180.
- ^ Lindsey, Jonathan S.; Wagner, Richard W. (1989). "Investigation of the Synthesis of ortho-Substituted Tetraphenylporphyrins". The Journal of Organic Chemistry. 54 (4): 828–836. doi: 10.1021/jo00265a021.