In organic chemistry, an aminal or aminoacetal is a functional group or type of organic compound that has two amine groups attached to the same carbon atom: −C(NR2)(NR2)−. (As is customary in organic chemistry, R can represent hydrogen or an alkyl group). [1] A common aminal is bis(dimethylamino)methane, a colorless liquid that is prepared by the reaction of dimethylamine and formaldehyde: [2]
- 2 (CH3)2NH + CH2O → [(CH3)2N]2CH2 + H2O
Aminals are encountered in, for instance, the Fischer indole synthesis. Several examples exist in nature. [3]
- Naturally occurring aminals
-
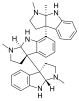
Hodgkinsine, an alkaloid with antiviral, antibacterial and antifungal effects
-
5,10-Methylenetetrahydrofolate, an intermediate in one-carbon metabolism
Hexahydro-1,3,5-triazine ((CH2NH)3), an intermediate in the condensation of formaldehyde and ammonia, tends to degrade to hexamethylene tetraamine.
Cyclic aminals can be obtained by the condensation of a diamine and an aldehyde. [4] Imidazolidines are one class of these cyclic aminals.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) " aminals". doi: 10.1351/goldbook.A00270
- ^ Gaudry, Michel; Jasor, Yves; Khac, Trung Bui (1979). "Regioselective Mannich Condensation with Dimethyl(Methylene)ammonium Trifluoroacetate: 1-(Dimethylamino)-4-methyl-3-pentanone". Org. Synth. 59: 153. doi: 10.15227/orgsyn.059.0153.
- ^ Duhamel, Lucette (1982). "Aminals". In Saul Patai (ed.). Amino, Nitroso and Nitro Compounds and Their Derivatives: Vol. 2. PATAI'S Chemistry of Functional Groups. pp. 849–907. doi: 10.1002/9780470771679.ch5. ISBN 9780470771679.
- ^ Hiersemann, M. "Functions bearing two nitrogens" in Comprehensive Organic Functional Group Transformations II 2005, volume 4, 411-441. Edited by Katritzky, Alan R.; Taylor, Richard J. K. doi: 10.1016/B0-08-044655-8/00075-1

In organic chemistry, an aminal or aminoacetal is a functional group or type of organic compound that has two amine groups attached to the same carbon atom: −C(NR2)(NR2)−. (As is customary in organic chemistry, R can represent hydrogen or an alkyl group). [1] A common aminal is bis(dimethylamino)methane, a colorless liquid that is prepared by the reaction of dimethylamine and formaldehyde: [2]
- 2 (CH3)2NH + CH2O → [(CH3)2N]2CH2 + H2O
Aminals are encountered in, for instance, the Fischer indole synthesis. Several examples exist in nature. [3]
- Naturally occurring aminals
-
Hodgkinsine, an alkaloid with antiviral, antibacterial and antifungal effects
-
5,10-Methylenetetrahydrofolate, an intermediate in one-carbon metabolism
Hexahydro-1,3,5-triazine ((CH2NH)3), an intermediate in the condensation of formaldehyde and ammonia, tends to degrade to hexamethylene tetraamine.
Cyclic aminals can be obtained by the condensation of a diamine and an aldehyde. [4] Imidazolidines are one class of these cyclic aminals.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) " aminals". doi: 10.1351/goldbook.A00270
- ^ Gaudry, Michel; Jasor, Yves; Khac, Trung Bui (1979). "Regioselective Mannich Condensation with Dimethyl(Methylene)ammonium Trifluoroacetate: 1-(Dimethylamino)-4-methyl-3-pentanone". Org. Synth. 59: 153. doi: 10.15227/orgsyn.059.0153.
- ^ Duhamel, Lucette (1982). "Aminals". In Saul Patai (ed.). Amino, Nitroso and Nitro Compounds and Their Derivatives: Vol. 2. PATAI'S Chemistry of Functional Groups. pp. 849–907. doi: 10.1002/9780470771679.ch5. ISBN 9780470771679.
- ^ Hiersemann, M. "Functions bearing two nitrogens" in Comprehensive Organic Functional Group Transformations II 2005, volume 4, 411-441. Edited by Katritzky, Alan R.; Taylor, Richard J. K. doi: 10.1016/B0-08-044655-8/00075-1