
| |
| Names | |
|---|---|
|
Preferred IUPAC name
Carbamoylcarbamic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| 3DMet | |
| 1756623 | |
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C2H4N2O3 | |
| Molar mass | 104.065 g·mol−1 |
| Appearance | white solid |
| Melting point | 114 °C (237 °F; 387 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Allophanic acid is the organic compound with the formula H2NC(O)NHCO2H. It is a carbamic acid, the carboxylated derivative of urea. Biuret can be viewed as the amide of allophanic acid. The compound can be prepared by treating urea with sodium bicarbonate: [1]
- H2NC(O)NH2 + NaHCO3 → H2NC(O)NHCO2H + NaOH
The anionicconjugate base, H2NC(O)NHCO2−, is called allophanate. Salts of this anion have been characterized by X-ray crystallography. [2] [3] The allophanate anion is the substrate for the enzyme allophanate hydrolase.
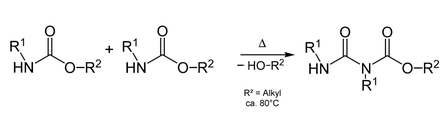
Allophanate esters arise from the condensation of carbamates.
- ^ Karachinskii, S. V.; Dragalov, V. V.; Chimishkyan, A. L.; Tsvetkov, V. Yu. (1987). "Reaction of urea with alkali metal carbonates". Zhurnal Organicheskoi Khimii. 23: 93–6.
- ^ Mak, Thomas C. W.; Yip, Wai Hing; Li, Qi (1995). "Novel Hydrogen-Bonded Host Lattices Built of Urea and the Elusive Allophanate Ion". Journal of the American Chemical Society. 117 (48): 11995–11996. doi: 10.1021/ja00153a022.
- ^ Li, Qi; Mak, Thomas C. W. (1996). "A Novel Inclusion Compound Consolidated by Host-host and Host-guest Hydrogen Bonding: (2-hydro-xyethyl)trimethylammonium Ions Included in a Channel Host Lattice Built of Urea Molecules and Allophanate Ions". Supramolecular Chemistry. 8: 73–80. doi: 10.1080/10610279608233970.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
Carbamoylcarbamic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| 3DMet | |
| 1756623 | |
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C2H4N2O3 | |
| Molar mass | 104.065 g·mol−1 |
| Appearance | white solid |
| Melting point | 114 °C (237 °F; 387 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Allophanic acid is the organic compound with the formula H2NC(O)NHCO2H. It is a carbamic acid, the carboxylated derivative of urea. Biuret can be viewed as the amide of allophanic acid. The compound can be prepared by treating urea with sodium bicarbonate: [1]
- H2NC(O)NH2 + NaHCO3 → H2NC(O)NHCO2H + NaOH
The anionicconjugate base, H2NC(O)NHCO2−, is called allophanate. Salts of this anion have been characterized by X-ray crystallography. [2] [3] The allophanate anion is the substrate for the enzyme allophanate hydrolase.
Allophanate esters arise from the condensation of carbamates.

- ^ Karachinskii, S. V.; Dragalov, V. V.; Chimishkyan, A. L.; Tsvetkov, V. Yu. (1987). "Reaction of urea with alkali metal carbonates". Zhurnal Organicheskoi Khimii. 23: 93–6.
- ^ Mak, Thomas C. W.; Yip, Wai Hing; Li, Qi (1995). "Novel Hydrogen-Bonded Host Lattices Built of Urea and the Elusive Allophanate Ion". Journal of the American Chemical Society. 117 (48): 11995–11996. doi: 10.1021/ja00153a022.
- ^ Li, Qi; Mak, Thomas C. W. (1996). "A Novel Inclusion Compound Consolidated by Host-host and Host-guest Hydrogen Bonding: (2-hydro-xyethyl)trimethylammonium Ions Included in a Channel Host Lattice Built of Urea Molecules and Allophanate Ions". Supramolecular Chemistry. 8: 73–80. doi: 10.1080/10610279608233970.