| |

| |
| Names | |
|---|---|
|
IUPAC name
hexanedial
| |
| Other names
1,4-Butane dicarboxaldehyde, 1,6-hexanedial
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.012.731 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H10O2 | |
| Molar mass | 114.144 g·mol−1 |
| Appearance | colorless liquid |
| Density | 1.003 g/cm3 |
| Melting point | −8 °C (18 °F; 265 K) |
| Boiling point | 68–70 °C (154–158 °F; 341–343 K) 3 mm Hg |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
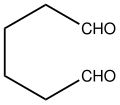
Adipaldehyde is the organic compound with the formula (CH2)4(CHO)2. It is a colorless oil that is usually encountered as an aqueous solution because it is highly reactive like many dialdehydes. [1] The compound has attracted interest as a precursor to nylon-related polymers. It can be produced by double hydroformylation of 1,3- butadiene, but this methodology has not achieved commercialization. [2] It has been prepared by oxidation of 1,6-hexanediol with pyridinium chlorochromate. [3]
References
- ^ Hardy, P. M.; Nicholls, A. C.; Rydon, H. N. (1972). "The Hydration and Polymerisation of Succinaldehyde, Glutaraldehyde, and Adipaldehyde". Journal of the Chemical Society, Perkin Transactions 2 (15): 2270. doi: 10.1039/P29720002270.
- ^ Yu, Si-min; Snavely, William K.; Chaudhari, Raghunath V.; Subramaniam, Bala (2020). "Butadiene hydroformylation to adipaldehyde with Rh-based catalysts: Insights into ligand effects". Molecular Catalysis. 484. doi: 10.1016/j.mcat.2019.110721. S2CID 213312335.
- ^ Corey, E.J.; Suggs, J.William (1975). "Pyridinium Chlorochromate. An efficient reagent for oxidation of primary and secondary alcohols to carbonyl compounds". Tetrahedron Letters. 16 (31): 2647–2650. doi: 10.1016/s0040-4039(00)75204-x.

| |

| |
| Names | |
|---|---|
|
IUPAC name
hexanedial
| |
| Other names
1,4-Butane dicarboxaldehyde, 1,6-hexanedial
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.012.731 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H10O2 | |
| Molar mass | 114.144 g·mol−1 |
| Appearance | colorless liquid |
| Density | 1.003 g/cm3 |
| Melting point | −8 °C (18 °F; 265 K) |
| Boiling point | 68–70 °C (154–158 °F; 341–343 K) 3 mm Hg |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Adipaldehyde is the organic compound with the formula (CH2)4(CHO)2. It is a colorless oil that is usually encountered as an aqueous solution because it is highly reactive like many dialdehydes. [1] The compound has attracted interest as a precursor to nylon-related polymers. It can be produced by double hydroformylation of 1,3- butadiene, but this methodology has not achieved commercialization. [2] It has been prepared by oxidation of 1,6-hexanediol with pyridinium chlorochromate. [3]
References
- ^ Hardy, P. M.; Nicholls, A. C.; Rydon, H. N. (1972). "The Hydration and Polymerisation of Succinaldehyde, Glutaraldehyde, and Adipaldehyde". Journal of the Chemical Society, Perkin Transactions 2 (15): 2270. doi: 10.1039/P29720002270.
- ^ Yu, Si-min; Snavely, William K.; Chaudhari, Raghunath V.; Subramaniam, Bala (2020). "Butadiene hydroformylation to adipaldehyde with Rh-based catalysts: Insights into ligand effects". Molecular Catalysis. 484. doi: 10.1016/j.mcat.2019.110721. S2CID 213312335.
- ^ Corey, E.J.; Suggs, J.William (1975). "Pyridinium Chlorochromate. An efficient reagent for oxidation of primary and secondary alcohols to carbonyl compounds". Tetrahedron Letters. 16 (31): 2647–2650. doi: 10.1016/s0040-4039(00)75204-x.