| |

| |
| Names | |
|---|---|
|
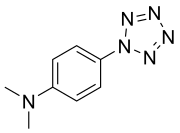
Preferred IUPAC name
N,N-Dimethyl-4-(1H-pentazol-1-yl)aniline | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C8H10N6 | |
| Molar mass | 190.205 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
4-Dimethylaminophenylpentazole is an unstable, explosive compound that contains the rare pentazole ring, which is composed of five nitrogen atoms. The electron donating effect of the 4-dimethylamino substituent on the phenyl ring makes this compound one of the more stable of the phenylpentazoles. At room temperature, its chemical half-life is only a few hours, although storage is possible at cryogenic temperatures. The compound was first prepared in 1956 [1] [2] [3] along with other substituted phenylpentazoles. Studies have been conducted on various other derivatives, though necessarily limited by the instability of these compounds. [4] [5] [6] [7] [8] Some more highly substituted derivatives, such as 2,6-dihydroxy-4-dimethylaminophenylpentazole, are slightly more stable but conversely, more difficult to make. [9] [10] Current research has focused on forming transition metal complexes of these pentazole derivatives, as the pentazole ring should be stabilised by bonding to the metal centre. [11] [12] [13]
- ^ Huisgen R, I. Ugi. Zur Losung eines klassichen Problems der organischen Stickstoff-Chemie. Angewandte Chemie. 1956; 68:705-706.
- ^ Ugi I, R. Huisgen. Pentazole II. Die Zerfallsgeschwindigkeit der Arylpentazole. Chemische Berichte. 1958; 91:531-537.
- ^ Ugi I, Perlinger H, Perlinger L. Pentazole III. Kristallisierte Aryl-pentazole. Chemische Berichte 1958; 98:2324-2329,
- ^ John D. Wallis and Jack D. Dunitz. An all-nitrogen aromatic ring system: structural study of 4-dimethyl-aminophenylpentazole. Journal of the Chemical Society. Chemical Communications. 1983: 910-911.
- ^ Butler RN, Collier S, Fleming AF (1996). "Pentazoles: proton and carbon-13 NMR spectra of some 1-arylpentazoles: kinetics and mechanism of degradation of the arylpentazole system". Journal of the Chemical Society, Perkin Transactions 2 (5): 801. doi: 10.1039/P29960000801.
- ^ Butler RN, Fox A, Collier S, Burke LA (1998). "Pentazole chemistry: the mechanism of the reaction of aryldiazonium chlorides with azide ion at −80 °C: concerted versus stepwise formation of arylpentazoles, detection of a pentazene intermediate, a combined 1H and 15N NMR experimental and ab initio theoretical study". Journal of the Chemical Society, Perkin Transactions 2 (10): 2243–2248. doi: 10.1039/A804040K.
- ^ Benin V, Kaszynski P, Radziszewski JG (February 2002). "Arylpentazoles revisited: experimental and theoretical studies of 4-hydroxyphenylpentazole and 4-oxophenylpentazole anion". The Journal of Organic Chemistry. 67 (4): 1354–8. doi: 10.1021/jo0110754. PMID 11846686.
- ^ Carlqvist P, Östmark H, Brinck T (2004). "The Stability of Arylpentazoles". The Journal of Physical Chemistry A. 108 (36): 7463. Bibcode: 2004JPCA..108.7463C. doi: 10.1021/jp0484480.
- ^ Efforts to synthesize the pentazolate anion
- ^ David Adam. The synthesis and characterisation of halogen and nitro phenyl azide derivatives as highly energetic materials. PhD dissertation, Ludwig-Maximilans-Universität München, 2001 [1]
- ^ Tsipis AC, Chaviara AT (February 2004). "Structure, energetics, and bonding of first row transition metal pentazolato complexes: a DFT study". Inorganic Chemistry. 43 (4): 1273–86. doi: 10.1021/ic035112g. PMID 14966962.
- ^ Burke LA, Fazen PJ (2004). "Electronic Supplementary Information for Chemical Communications". Chemical Communications (9): 1082–3. doi: 10.1039/B315812H. PMID 15116195.
- ^ Burke LA, Fazen PJ (2009). "Correlation analysis of the interconversion and nitrogen loss reactions of aryl pentazenes and pentazoles derived from aryl diazonium and azide ions". International Journal of Quantum Chemistry. 109 (15): 3613. Bibcode: 2009IJQC..109.3613B. doi: 10.1002/qua.22408.

| |

| |
| Names | |
|---|---|
|
Preferred IUPAC name
N,N-Dimethyl-4-(1H-pentazol-1-yl)aniline | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C8H10N6 | |
| Molar mass | 190.205 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
4-Dimethylaminophenylpentazole is an unstable, explosive compound that contains the rare pentazole ring, which is composed of five nitrogen atoms. The electron donating effect of the 4-dimethylamino substituent on the phenyl ring makes this compound one of the more stable of the phenylpentazoles. At room temperature, its chemical half-life is only a few hours, although storage is possible at cryogenic temperatures. The compound was first prepared in 1956 [1] [2] [3] along with other substituted phenylpentazoles. Studies have been conducted on various other derivatives, though necessarily limited by the instability of these compounds. [4] [5] [6] [7] [8] Some more highly substituted derivatives, such as 2,6-dihydroxy-4-dimethylaminophenylpentazole, are slightly more stable but conversely, more difficult to make. [9] [10] Current research has focused on forming transition metal complexes of these pentazole derivatives, as the pentazole ring should be stabilised by bonding to the metal centre. [11] [12] [13]
- ^ Huisgen R, I. Ugi. Zur Losung eines klassichen Problems der organischen Stickstoff-Chemie. Angewandte Chemie. 1956; 68:705-706.
- ^ Ugi I, R. Huisgen. Pentazole II. Die Zerfallsgeschwindigkeit der Arylpentazole. Chemische Berichte. 1958; 91:531-537.
- ^ Ugi I, Perlinger H, Perlinger L. Pentazole III. Kristallisierte Aryl-pentazole. Chemische Berichte 1958; 98:2324-2329,
- ^ John D. Wallis and Jack D. Dunitz. An all-nitrogen aromatic ring system: structural study of 4-dimethyl-aminophenylpentazole. Journal of the Chemical Society. Chemical Communications. 1983: 910-911.
- ^ Butler RN, Collier S, Fleming AF (1996). "Pentazoles: proton and carbon-13 NMR spectra of some 1-arylpentazoles: kinetics and mechanism of degradation of the arylpentazole system". Journal of the Chemical Society, Perkin Transactions 2 (5): 801. doi: 10.1039/P29960000801.
- ^ Butler RN, Fox A, Collier S, Burke LA (1998). "Pentazole chemistry: the mechanism of the reaction of aryldiazonium chlorides with azide ion at −80 °C: concerted versus stepwise formation of arylpentazoles, detection of a pentazene intermediate, a combined 1H and 15N NMR experimental and ab initio theoretical study". Journal of the Chemical Society, Perkin Transactions 2 (10): 2243–2248. doi: 10.1039/A804040K.
- ^ Benin V, Kaszynski P, Radziszewski JG (February 2002). "Arylpentazoles revisited: experimental and theoretical studies of 4-hydroxyphenylpentazole and 4-oxophenylpentazole anion". The Journal of Organic Chemistry. 67 (4): 1354–8. doi: 10.1021/jo0110754. PMID 11846686.
- ^ Carlqvist P, Östmark H, Brinck T (2004). "The Stability of Arylpentazoles". The Journal of Physical Chemistry A. 108 (36): 7463. Bibcode: 2004JPCA..108.7463C. doi: 10.1021/jp0484480.
- ^ Efforts to synthesize the pentazolate anion
- ^ David Adam. The synthesis and characterisation of halogen and nitro phenyl azide derivatives as highly energetic materials. PhD dissertation, Ludwig-Maximilans-Universität München, 2001 [1]
- ^ Tsipis AC, Chaviara AT (February 2004). "Structure, energetics, and bonding of first row transition metal pentazolato complexes: a DFT study". Inorganic Chemistry. 43 (4): 1273–86. doi: 10.1021/ic035112g. PMID 14966962.
- ^ Burke LA, Fazen PJ (2004). "Electronic Supplementary Information for Chemical Communications". Chemical Communications (9): 1082–3. doi: 10.1039/B315812H. PMID 15116195.
- ^ Burke LA, Fazen PJ (2009). "Correlation analysis of the interconversion and nitrogen loss reactions of aryl pentazenes and pentazoles derived from aryl diazonium and azide ions". International Journal of Quantum Chemistry. 109 (15): 3613. Bibcode: 2009IJQC..109.3613B. doi: 10.1002/qua.22408.