| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
2-Phenylpyridine | |||
| Other names
2-Azabiphenyl
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.012.512 | ||
| EC Number |
| ||
| MeSH | C058324 | ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C11H9N | |||
| Molar mass | 155.200 g·mol−1 | ||
| Appearance | Colorless oil | ||
| Density | 1.086 g/mL | ||
| Boiling point | 268–270 °C (514–518 °F; 541–543 K) | ||
| Low | |||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
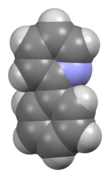
2-Phenylpyridine is an organic compound with the formula C6H5C5H4N (or C11H9N). It is a colourless viscous liquid. The compound and related derivatives have attracted interest as precursors to highly fluorescent metal complexes of possible value as organic light emitting diodes (OLEDs). [1]
The compound is prepared by the reaction of phenyl lithium with pyridine: [2]
- C6H5Li + C5H5N → C6H5-C5H4N + LiH
The reaction of iridium trichloride with 2-phenylpyridine proceeds via cyclometallation to give the chloride-bridged complex: [3] [4]
- 4 C6H5-C5H4N + 2 IrCl3(H2O)3 → Ir2Cl2(C6H4-C5H4N)4 + 4 HCl
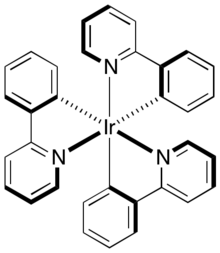
This complex can be converted to the pictured tris(cyclometallated) derivative tris(2-phenylpyridine)iridium.
References
- ^ Eli Zysman-Colman, ed. (2017). Iridium(III) in Optoelectronic and Photonics Applications. John Wiley & Sons. ISBN 978-1-119-00713-5.
- ^ Evans, J. C. W.; Allen, C. F. H. (1938). "2-Phenylpyridine". Organic Syntheses. 18: 70. doi: 10.15227/orgsyn.018.0070.
- ^ Lamansky, S.; Djurovich, P.; Murphy, D.; et al. (2001). "Synthesis and Characterization of Phosphorescent Cyclometalated Iridium Complexes". Inorganic Chemistry. 40 (7): 1704–1711. doi: 10.1021/ic0008969. PMID 11261983.
- ^ Kip A. Teegardin, Jimmie D. Weaver (2018). "Preparation of Fac-Tris(2-Phenylpyridinato) Iridium(III)". Org. Synth. 95: 29–45. doi: 10.15227/orgsyn.095.0029. PMC 6022758. PMID 29962554.
Further reading
- Zhou, Guijiang; Wong, Wai-Yeung; Yang, Xiaolong (2011). "New Design Tactics in OLEDs Using Functionalized 2-Phenylpyridine-Type Cyclometalates of Iridium(III) and Platinum(II)". Chemistry: An Asian Journal. 6 (7): 1706–1727. doi: 10.1002/asia.201000928. PMID 21557486.
| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
2-Phenylpyridine | |||
| Other names
2-Azabiphenyl
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.012.512 | ||
| EC Number |
| ||
| MeSH | C058324 | ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C11H9N | |||
| Molar mass | 155.200 g·mol−1 | ||
| Appearance | Colorless oil | ||
| Density | 1.086 g/mL | ||
| Boiling point | 268–270 °C (514–518 °F; 541–543 K) | ||
| Low | |||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
2-Phenylpyridine is an organic compound with the formula C6H5C5H4N (or C11H9N). It is a colourless viscous liquid. The compound and related derivatives have attracted interest as precursors to highly fluorescent metal complexes of possible value as organic light emitting diodes (OLEDs). [1]
The compound is prepared by the reaction of phenyl lithium with pyridine: [2]
- C6H5Li + C5H5N → C6H5-C5H4N + LiH
The reaction of iridium trichloride with 2-phenylpyridine proceeds via cyclometallation to give the chloride-bridged complex: [3] [4]
- 4 C6H5-C5H4N + 2 IrCl3(H2O)3 → Ir2Cl2(C6H4-C5H4N)4 + 4 HCl
This complex can be converted to the pictured tris(cyclometallated) derivative tris(2-phenylpyridine)iridium.

References
- ^ Eli Zysman-Colman, ed. (2017). Iridium(III) in Optoelectronic and Photonics Applications. John Wiley & Sons. ISBN 978-1-119-00713-5.
- ^ Evans, J. C. W.; Allen, C. F. H. (1938). "2-Phenylpyridine". Organic Syntheses. 18: 70. doi: 10.15227/orgsyn.018.0070.
- ^ Lamansky, S.; Djurovich, P.; Murphy, D.; et al. (2001). "Synthesis and Characterization of Phosphorescent Cyclometalated Iridium Complexes". Inorganic Chemistry. 40 (7): 1704–1711. doi: 10.1021/ic0008969. PMID 11261983.
- ^ Kip A. Teegardin, Jimmie D. Weaver (2018). "Preparation of Fac-Tris(2-Phenylpyridinato) Iridium(III)". Org. Synth. 95: 29–45. doi: 10.15227/orgsyn.095.0029. PMC 6022758. PMID 29962554.
Further reading
- Zhou, Guijiang; Wong, Wai-Yeung; Yang, Xiaolong (2011). "New Design Tactics in OLEDs Using Functionalized 2-Phenylpyridine-Type Cyclometalates of Iridium(III) and Platinum(II)". Chemistry: An Asian Journal. 6 (7): 1706–1727. doi: 10.1002/asia.201000928. PMID 21557486.