| |
| Names | |
|---|---|
|
Preferred IUPAC name
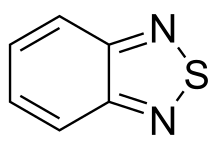
2,1,3-Benzothiadiazole | |
Other names
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.005.442 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H4N2S | |
| Molar mass | 136.17 g·mol−1 |
| Melting point | 54.0 °C (129.2 °F; 327.1 K) |
| Boiling point | 203.0 °C (397.4 °F; 476.1 K) |
| Related compounds | |
Related compounds
|
1,2,3-Benzothiadiazole |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
2,1,3-Benzothiadiazole is a bicyclic molecule composed of a benzene ring that is fused to a 1,2,5-thiadiazole.
Preparation and structure
2,1,3-Benzothiadiazole has been known since the 19th century. It is readily prepared in at least 85% yield from o-phenylenediamine by reaction with two equivalents of thionyl chloride in pyridine. The by-products are sulfur dioxide and HCl. [1]
There are a number of alternative methods used to make this heterocycle and these have been reviewed. [2] [3] The crystal structure of the compound was determined in 1951, when it had the common name piazthiol(e). [4]
Reactions
The extent of the aromaticity of the compound was examined by a study of its proton NMR spectrum and comparison with naphthalene, which allowed the conclusion that it and related oxygen and selenium heterocycles did behave as 10-electron systems in which the 2-heteroatom contributed its lone pair to the ring current, in accordance with Hückel's rule. [5]
As a result, 2,1,3-benzothiadiazole undergoes the standard chemistry of aromatic compounds, for example readily forming nitro [1] and chloro derivatives. [6] The chemistry of this heterocycle and its simple derivatives has been reviewed. [7]
Under reducing conditions, 2,1,3-benzothiadiazoles can be converted back to the 1,2-diaminobenzene compounds from which they were prepared. This can be a useful way to protect a pair of reactive amino groups while other transformations are performed in the benzene ring to which they are attached. [8]
Bromination of 2,1,3-Benzothiadiazole is commonly performed to synthesize 4,7-dibromo-2,1,3-benzothiadiazole. This derivative is extensively used as building block in the design and synthesis of larger molecules and conductive polymers via Suzuki-Miyaura cross-coupling reactions. [9]

Derivatives
2,1,3-Benzothiadiazole derivatives containing carbazole units have been found to be luminiscent, with high emission intensity and quantum efficiency. [10]
Different π-extended molecular systems based on 2,1,3-benzothiadiazole have been built to study fundamental structure–property relationships. [8] One example of this type of oligomer consist of extended thiophene building blocks as electron donors and 2,1,3-benzothiadiazole as electron aceptor. This oligomer was synthesized using a Sonogashira cross-coupling reaction and it showed low HOMO–LUMO gaps which could be interesting for organic semiconductor applications. [11]
Asymmetric derivatives with diphenylamine donors, cyanoacrylic acid acceptors and thiophene linkers bridged by a 2,1,3-benzothiadiazole have been designed as organic dyes with improved charge separation properties [12] when compared to classic cyanine [13] and hemicyanine [14] dyes.
Applications
2,1,3-Benzothiadiazole has been of interest as a redox-active organic component in flow batteries owing to its favourable solubility, low reduction potential and fast electrochemical kinetics. [15]
Such properties in derivatives containing this heterocycle have made it of growing interest in dyestuffs, [16] white light-emitting polymers, [8] [17] solar cells, [18] and in luminescence studies. [19]
References
- ^ a b Pesin, V. G.; Sergeev, V. A. (1969). "Research on 2,1,3-thia- and selenadiazole". Chemistry of Heterocyclic Compounds. 3 (5): 662–666. doi: 10.1007/BF00468340. S2CID 98830770.
- ^ Storr; Gilchrist, eds. (2004). "Product Class 11: 1,2,5-Thiadiazoles and Related Compounds". Category 2, Hetarenes and Related Ring Systems. doi: 10.1055/sos-SD-013-00458. ISBN 978-3-13-112281-0.
- ^ Rakitin, Oleg A. (2019). "Recent Developments in the Synthesis of 1,2,5-Thiadiazoles and 2,1,3-Benzothiadiazoles". Synthesis. 51 (23): 4338–4347. doi: 10.1055/s-0039-1690679. S2CID 204128866.
- ^ Luzzati, Z.Z. (1951). "Structure cristalline de piasélénol, piazthiol et benzofurazane". Acta Crystallographica. 4 (3): 193–200. Bibcode: 1951AcCry...4..193L. doi: 10.1107/S0365110X51000702.
- ^ Fedin, E. I.; Todres, Z. V. (1970). "Studies in the field of aromatic heterocycles" (PDF). Chemistry of Heterocyclic Compounds. 4 (3): 308–313. doi: 10.1007/BF00755265. S2CID 91864834.
- ^ Pesin, V. G.; d'Yachenko, E. K. (1969). "Researches on 2,1,3-thia-and selenadiazole". Chemistry of Heterocyclic Compounds. 3: 68–70. doi: 10.1007/BF00944264. S2CID 100997583.
- ^ Houben-Weyl Methods of Organic Chemistry Vol. E 8d, 4th Edition Supplement: Hetarenes III (Five-Membered Rings with Two and More Heteroatoms in the Ring System) - Part 4. Georg Thieme Verlag. 14 May 2014. ISBN 978-3-13-181244-5.
- ^ a b c Neto, Brenno A. D.; Lapis, Alexandre A. M.; da Silva Júnior, Eufrânio N.; Dupont, Jairton (January 2013). "2,1,3-Benzothiadiazole and Derivatives: Synthesis, Properties, Reactions, and Applications in Light Technology of Small Molecules". European Journal of Organic Chemistry. 2013 (2): 228–255. doi: 10.1002/ejoc.201201161.
- ^ Huang, Jian; Niu, Yuhua; Yang, Wei; Mo, Yueqi; Yuan, Ming; Cao, Yong (2002-07-01). "Novel Electroluminescent Polymers Derived from Carbazole and Benzothiadiazole". Macromolecules. 35 (16): 6080–6082. Bibcode: 2002MaMol..35.6080H. doi: 10.1021/ma0255130. ISSN 0024-9297.
- ^ Tao, Yun-Mei; Li, Hong-Yan; Xu, Qiu-Lei; Zhu, Yu-Cheng; Kang, Ling-Chen; Zheng, You-Xuan; Zuo, Jing-Lin; You, Xiao-Zeng (2011). "Synthesis and characterization of efficient luminescent materials based on 2,1,3-benzothiadiazole with carbazole moieties". Synthetic Metals. 161 (9–10): 718–723. doi: 10.1016/j.synthmet.2011.01.020.
- ^ Kitamura, Chitoshi; Saito, Kakuya; Ouchi, Mikio; Yoneda, Akio; Yamashita, Yoshiro (October 2002). "Synthesis and Crystal Structure of 4,7-bis (2-thienylethynyl)-2,1,3-benzothiadiazole". Journal of Chemical Research. 2002 (10): 511–513. doi: 10.3184/030823402103170565. ISSN 1747-5198.
- ^ Velusamy, Marappan; Justin Thomas, K. R.; Lin, Jiann T.; Hsu, Ying-Chan; Ho, Kuo-Chuan (2005-05-01). "Organic Dyes Incorporating Low-Band-Gap Chromophores for Dye-Sensitized Solar Cells". Organic Letters. 7 (10): 1899–1902. doi: 10.1021/ol050417f. ISSN 1523-7060. PMID 15876014.
- ^ Ehret, A.; Stuhl, L.; Spitler, M. T. (2001-10-01). "Spectral Sensitization of TiO 2 Nanocrystalline Electrodes with Aggregated Cyanine Dyes". The Journal of Physical Chemistry B. 105 (41): 9960–9965. doi: 10.1021/jp011952+. ISSN 1520-6106.
- ^ Yao, Qiao-Hong; Meng, Fan-Shun; Li, Fu-You; Tian, He; Huang, Chun-Hui (2003-04-16). "Photoelectric conversion properties of four novel carboxylated hemicyanine dyes on TiO2 electrode". Journal of Materials Chemistry. 13 (5): 1048–1053. doi: 10.1039/b300083b.
- ^ Duan, Wentao; Huang, Jinhua; Kowalski, Jeffrey A.; Shkrob, Ilya A.; Vijayakumar, M.; Walter, Eric; Pan, Baofei; Yang, Zheng; Milshtein, Jarrod D.; Li, Bin; Liao, Chen; Zhang, Zhengcheng; Wang, Wei; Liu, Jun; Moore, Jeffery S.; Brushett, Fikile R.; Zhang, Lu; Wei, Xiaoliang (2017). ""Wine-Dark Sea" in an Organic Flow Battery: Storing Negative Charge in 2,1,3-Benzothiadiazole Radicals Leads to Improved Cyclability". ACS Energy Letters. 2 (5): 1156–1161. doi: 10.1021/acsenergylett.7b00261.
- ^ Frizon, Tiago Elias Allievi; Valdivia Martínez, Julio César; Westrup, José Luiz; Duarte, Rodrigo da Costa; Zapp, Eduardo; Domiciano, Kelvin Guessi; Rodembusch, Fabiano Severo; Dal-Bó, Alexandre Gonçalves (December 2016). "2,1,3-Benzothiadiazole-based fluorophores. Synthesis, electrochemical, thermal and photophysical characterization". Dyes and Pigments. 135: 26–35. doi: 10.1016/j.dyepig.2016.07.011.
- ^ Wu, Hongbin; Ying, Lei; Yang, Wei; Cao, Yong (2010). "White-Emitting Polymers and Devices". WOLEDs and Organic Photovoltaics. Green Energy and Technology. pp. 37–78. doi: 10.1007/978-3-642-14935-1_2. ISBN 978-3-642-14934-4. S2CID 54914788.
- ^ Wang, Yang; Michinobu, Tsuyoshi (2016). "Benzothiadiazole and its π-extended, heteroannulated derivatives: Useful acceptor building blocks for high-performance donor–acceptor polymers in organic electronics". Journal of Materials Chemistry C. 4 (26): 6200–6214. doi: 10.1039/C6TC01860B.
- ^ Sukhikh, Taisiya; Ogienko, D.; Bashirov, D.; Konchenkoa, S. (May 21, 2019). "Luminescent complexes of 2,1,3-benzothiadiazole derivatives". Russian Chemical Bulletin. 68 (4): 651–661. doi: 10.1007/s11172-019-2472-9. S2CID 182415426.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
2,1,3-Benzothiadiazole | |
Other names
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.005.442 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H4N2S | |
| Molar mass | 136.17 g·mol−1 |
| Melting point | 54.0 °C (129.2 °F; 327.1 K) |
| Boiling point | 203.0 °C (397.4 °F; 476.1 K) |
| Related compounds | |
Related compounds
|
1,2,3-Benzothiadiazole |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
2,1,3-Benzothiadiazole is a bicyclic molecule composed of a benzene ring that is fused to a 1,2,5-thiadiazole.
Preparation and structure
2,1,3-Benzothiadiazole has been known since the 19th century. It is readily prepared in at least 85% yield from o-phenylenediamine by reaction with two equivalents of thionyl chloride in pyridine. The by-products are sulfur dioxide and HCl. [1]
There are a number of alternative methods used to make this heterocycle and these have been reviewed. [2] [3] The crystal structure of the compound was determined in 1951, when it had the common name piazthiol(e). [4]
Reactions
The extent of the aromaticity of the compound was examined by a study of its proton NMR spectrum and comparison with naphthalene, which allowed the conclusion that it and related oxygen and selenium heterocycles did behave as 10-electron systems in which the 2-heteroatom contributed its lone pair to the ring current, in accordance with Hückel's rule. [5]
As a result, 2,1,3-benzothiadiazole undergoes the standard chemistry of aromatic compounds, for example readily forming nitro [1] and chloro derivatives. [6] The chemistry of this heterocycle and its simple derivatives has been reviewed. [7]
Under reducing conditions, 2,1,3-benzothiadiazoles can be converted back to the 1,2-diaminobenzene compounds from which they were prepared. This can be a useful way to protect a pair of reactive amino groups while other transformations are performed in the benzene ring to which they are attached. [8]
Bromination of 2,1,3-Benzothiadiazole is commonly performed to synthesize 4,7-dibromo-2,1,3-benzothiadiazole. This derivative is extensively used as building block in the design and synthesis of larger molecules and conductive polymers via Suzuki-Miyaura cross-coupling reactions. [9]

Derivatives
2,1,3-Benzothiadiazole derivatives containing carbazole units have been found to be luminiscent, with high emission intensity and quantum efficiency. [10]
Different π-extended molecular systems based on 2,1,3-benzothiadiazole have been built to study fundamental structure–property relationships. [8] One example of this type of oligomer consist of extended thiophene building blocks as electron donors and 2,1,3-benzothiadiazole as electron aceptor. This oligomer was synthesized using a Sonogashira cross-coupling reaction and it showed low HOMO–LUMO gaps which could be interesting for organic semiconductor applications. [11]
Asymmetric derivatives with diphenylamine donors, cyanoacrylic acid acceptors and thiophene linkers bridged by a 2,1,3-benzothiadiazole have been designed as organic dyes with improved charge separation properties [12] when compared to classic cyanine [13] and hemicyanine [14] dyes.
Applications
2,1,3-Benzothiadiazole has been of interest as a redox-active organic component in flow batteries owing to its favourable solubility, low reduction potential and fast electrochemical kinetics. [15]
Such properties in derivatives containing this heterocycle have made it of growing interest in dyestuffs, [16] white light-emitting polymers, [8] [17] solar cells, [18] and in luminescence studies. [19]
References
- ^ a b Pesin, V. G.; Sergeev, V. A. (1969). "Research on 2,1,3-thia- and selenadiazole". Chemistry of Heterocyclic Compounds. 3 (5): 662–666. doi: 10.1007/BF00468340. S2CID 98830770.
- ^ Storr; Gilchrist, eds. (2004). "Product Class 11: 1,2,5-Thiadiazoles and Related Compounds". Category 2, Hetarenes and Related Ring Systems. doi: 10.1055/sos-SD-013-00458. ISBN 978-3-13-112281-0.
- ^ Rakitin, Oleg A. (2019). "Recent Developments in the Synthesis of 1,2,5-Thiadiazoles and 2,1,3-Benzothiadiazoles". Synthesis. 51 (23): 4338–4347. doi: 10.1055/s-0039-1690679. S2CID 204128866.
- ^ Luzzati, Z.Z. (1951). "Structure cristalline de piasélénol, piazthiol et benzofurazane". Acta Crystallographica. 4 (3): 193–200. Bibcode: 1951AcCry...4..193L. doi: 10.1107/S0365110X51000702.
- ^ Fedin, E. I.; Todres, Z. V. (1970). "Studies in the field of aromatic heterocycles" (PDF). Chemistry of Heterocyclic Compounds. 4 (3): 308–313. doi: 10.1007/BF00755265. S2CID 91864834.
- ^ Pesin, V. G.; d'Yachenko, E. K. (1969). "Researches on 2,1,3-thia-and selenadiazole". Chemistry of Heterocyclic Compounds. 3: 68–70. doi: 10.1007/BF00944264. S2CID 100997583.
- ^ Houben-Weyl Methods of Organic Chemistry Vol. E 8d, 4th Edition Supplement: Hetarenes III (Five-Membered Rings with Two and More Heteroatoms in the Ring System) - Part 4. Georg Thieme Verlag. 14 May 2014. ISBN 978-3-13-181244-5.
- ^ a b c Neto, Brenno A. D.; Lapis, Alexandre A. M.; da Silva Júnior, Eufrânio N.; Dupont, Jairton (January 2013). "2,1,3-Benzothiadiazole and Derivatives: Synthesis, Properties, Reactions, and Applications in Light Technology of Small Molecules". European Journal of Organic Chemistry. 2013 (2): 228–255. doi: 10.1002/ejoc.201201161.
- ^ Huang, Jian; Niu, Yuhua; Yang, Wei; Mo, Yueqi; Yuan, Ming; Cao, Yong (2002-07-01). "Novel Electroluminescent Polymers Derived from Carbazole and Benzothiadiazole". Macromolecules. 35 (16): 6080–6082. Bibcode: 2002MaMol..35.6080H. doi: 10.1021/ma0255130. ISSN 0024-9297.
- ^ Tao, Yun-Mei; Li, Hong-Yan; Xu, Qiu-Lei; Zhu, Yu-Cheng; Kang, Ling-Chen; Zheng, You-Xuan; Zuo, Jing-Lin; You, Xiao-Zeng (2011). "Synthesis and characterization of efficient luminescent materials based on 2,1,3-benzothiadiazole with carbazole moieties". Synthetic Metals. 161 (9–10): 718–723. doi: 10.1016/j.synthmet.2011.01.020.
- ^ Kitamura, Chitoshi; Saito, Kakuya; Ouchi, Mikio; Yoneda, Akio; Yamashita, Yoshiro (October 2002). "Synthesis and Crystal Structure of 4,7-bis (2-thienylethynyl)-2,1,3-benzothiadiazole". Journal of Chemical Research. 2002 (10): 511–513. doi: 10.3184/030823402103170565. ISSN 1747-5198.
- ^ Velusamy, Marappan; Justin Thomas, K. R.; Lin, Jiann T.; Hsu, Ying-Chan; Ho, Kuo-Chuan (2005-05-01). "Organic Dyes Incorporating Low-Band-Gap Chromophores for Dye-Sensitized Solar Cells". Organic Letters. 7 (10): 1899–1902. doi: 10.1021/ol050417f. ISSN 1523-7060. PMID 15876014.
- ^ Ehret, A.; Stuhl, L.; Spitler, M. T. (2001-10-01). "Spectral Sensitization of TiO 2 Nanocrystalline Electrodes with Aggregated Cyanine Dyes". The Journal of Physical Chemistry B. 105 (41): 9960–9965. doi: 10.1021/jp011952+. ISSN 1520-6106.
- ^ Yao, Qiao-Hong; Meng, Fan-Shun; Li, Fu-You; Tian, He; Huang, Chun-Hui (2003-04-16). "Photoelectric conversion properties of four novel carboxylated hemicyanine dyes on TiO2 electrode". Journal of Materials Chemistry. 13 (5): 1048–1053. doi: 10.1039/b300083b.
- ^ Duan, Wentao; Huang, Jinhua; Kowalski, Jeffrey A.; Shkrob, Ilya A.; Vijayakumar, M.; Walter, Eric; Pan, Baofei; Yang, Zheng; Milshtein, Jarrod D.; Li, Bin; Liao, Chen; Zhang, Zhengcheng; Wang, Wei; Liu, Jun; Moore, Jeffery S.; Brushett, Fikile R.; Zhang, Lu; Wei, Xiaoliang (2017). ""Wine-Dark Sea" in an Organic Flow Battery: Storing Negative Charge in 2,1,3-Benzothiadiazole Radicals Leads to Improved Cyclability". ACS Energy Letters. 2 (5): 1156–1161. doi: 10.1021/acsenergylett.7b00261.
- ^ Frizon, Tiago Elias Allievi; Valdivia Martínez, Julio César; Westrup, José Luiz; Duarte, Rodrigo da Costa; Zapp, Eduardo; Domiciano, Kelvin Guessi; Rodembusch, Fabiano Severo; Dal-Bó, Alexandre Gonçalves (December 2016). "2,1,3-Benzothiadiazole-based fluorophores. Synthesis, electrochemical, thermal and photophysical characterization". Dyes and Pigments. 135: 26–35. doi: 10.1016/j.dyepig.2016.07.011.
- ^ Wu, Hongbin; Ying, Lei; Yang, Wei; Cao, Yong (2010). "White-Emitting Polymers and Devices". WOLEDs and Organic Photovoltaics. Green Energy and Technology. pp. 37–78. doi: 10.1007/978-3-642-14935-1_2. ISBN 978-3-642-14934-4. S2CID 54914788.
- ^ Wang, Yang; Michinobu, Tsuyoshi (2016). "Benzothiadiazole and its π-extended, heteroannulated derivatives: Useful acceptor building blocks for high-performance donor–acceptor polymers in organic electronics". Journal of Materials Chemistry C. 4 (26): 6200–6214. doi: 10.1039/C6TC01860B.
- ^ Sukhikh, Taisiya; Ogienko, D.; Bashirov, D.; Konchenkoa, S. (May 21, 2019). "Luminescent complexes of 2,1,3-benzothiadiazole derivatives". Russian Chemical Bulletin. 68 (4): 651–661. doi: 10.1007/s11172-019-2472-9. S2CID 182415426.
