| |

| |
| Names | |
|---|---|
|
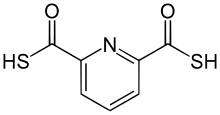
IUPAC name
2,6-Pyridinedicarbothioic acid
| |
|
Preferred IUPAC name
Pyridine-2,6-bis(carbothioic S-acid) | |
| Other names
PDTC, dithiopyridinedicarbothioic acid
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C7H5O2S2 | |
| Molar mass | 185.24 g·mol−1 |
| Appearance | White crystalline solid |
| Density | 1.415 g/cm3 |
| Melting point | 97 to 99 °C (207 to 210 °F; 370 to 372 K) |
| Boiling point | 404.4 °C (759.9 °F; 677.5 K) |
| 1000 g/L (5.02 mol/L) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
acidic |
| Flash point | 198.4 °C (389.1 °F; 471.5 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
2,6-Pyridinedicarbothioic acid (PDTC) is an organosulfur compound that is produced by some bacteria. It functions as a , a low molecular weight compound that scavenges iron. Siderophores solubilize compounds by forming strong complexes. PDTC is secreted by the soil bacteria Pseudomonas stutzeri and Pseudomonas putida. [1]
PDTC can be synthesized in the laboratory by treating the diacid dichloride of pyridine-2,6-dicarboxylic with H2S in pyridine:
- NC5H3(COCl)2 + 2 H2S + 2 C5H5N → [C5H5NH+][HNC5H3(COS)−2] + [C5H5NH]Cl
This route produces the pyridinium salt of pyridinium-2,6-dicarbothioate. Treatment of this orange-colored salt with sulfuric acid gives colorless PDTC, which can then be extracted with dichloromethane. [2]
The biosynthesis of PDTC remains unclear although some insights can be deduced from the genetics. [3] It is suggested that Pseudomonas stutzeri may have acquired at least one of the genes by lateral transfer from mycobacteria. [4] In a proposed biosynthetic sequence pyridine-2,6-dicarboxylic acid, a known bacterial metabolite, [4] is activated as its bis-adenosine monophosphate (AMP) derivative. The sulfur donor and its activation remain uncertain. [5]

PDTC binds to both Fe2+ and Fe3+. The ferric complex is brown, whereas the ferrous complex is blue. In the presence of air, the ferrous complex oxidizes to the ferric compound. [7] It is iron selective [4] as only the Fe complex is soluble in water. PDTC is produced mainly during the exponential phase of bacterial growth. The conditions at which Pseudomonas produces PDTC is 25 °C, pH=8 and sufficient aeration. [5]
- ^ Budzikiewicz, Herbert (2010). "Microbial Siderophores". In Kinghorn, A. Douglas; Falk, Heinz; Kobayashi, Junichi (eds.). Fortschritte der Chemie organischer Naturstoffe / Progress in the Chemistry of Organic Natural Products, Vol. 92 [Progress in the Chemistry of Organic Natural Products]. Vol. 92. pp. 1–75. doi: 10.1007/978-3-211-99661-4_1. ISBN 978-3-211-99660-7. PMID 20198464.
- ^ Hildebrand, U.; Ockels, W.; Lex, J.; Budzikiewicz, H. (1983). "Zur Struktur Eines 1:1-Adduktes von Pyridin-2,6-Dicarbothiosäure und Pyridin". Phosphorus and Sulfur and the Related Elements. 16 (3): 361–364. doi: 10.1080/03086648308080490.
- ^ Cortese, Marc S; Caplan, Allan B; Crawford, Ronald L (2002). "Structural, functional, and evolutionary analysis of moeZ, a gene encoding an enzyme required for the synthesis of the Pseudomonas metabolite, pyridine-2,6-bis(thiocarboxylic acid)". BMC Evolutionary Biology. 2: 8. doi: 10.1186/1471-2148-2-8. PMC 115864. PMID 11972321.
- ^ a b c Cortese, Marc S.; Paszczynski, Andrzej; Lewis, Thomas A.; Sebat, Jonathan L.; Borek, Vladimir; Crawford, Ronald L. (2002). "Metal chelating properties of pyridine-2,6-bis(thiocarboxylic acid) produced by Pseudomonas spp. And the biological activities of the formed complexes". BioMetals. 15 (2): 103–120. doi: 10.1023/A:1015241925322. PMID 12046919. S2CID 5545637.
- ^ a b Budzikiewicz, H. (2003). "Heteroaromatic monothiocarboxylic acids from Pseudomonas spp". Biodegradation. 14 (2): 65–72. doi: 10.1023/A:1024012015127. PMID 12877462. S2CID 29898226.
- ^ Hildebrand, U.; Lex, J.; Taraz, K.; Winkler, S.; Ockels, W.; Budzikiewicz, H. (1984). "Untersuchungen zum Redox-System Bis-(pyridin-2,6-dicarbothioato)-Ferrat(II) /-Ferrat(III) [1]". Zeitschrift für Naturforschung B. 39 (11): 1607–1613. doi: 10.1515/znb-1984-1123. S2CID 94908888.
- ^ Ockels, W., Roemer, A., Budzikiewicz, H., Korth, H., Pulverer, G., "Bacterial constituents. II. An iron(II) complex of pyridine-2,6-di-(monothiocarboxylic acid) - a novel bacterial metabolic product", Tetrahedron Lett. 1978, 3341. doi: 10.1016/S0040-4039(01)85634-3

| |

| |
| Names | |
|---|---|
|
IUPAC name
2,6-Pyridinedicarbothioic acid
| |
|
Preferred IUPAC name
Pyridine-2,6-bis(carbothioic S-acid) | |
| Other names
PDTC, dithiopyridinedicarbothioic acid
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C7H5O2S2 | |
| Molar mass | 185.24 g·mol−1 |
| Appearance | White crystalline solid |
| Density | 1.415 g/cm3 |
| Melting point | 97 to 99 °C (207 to 210 °F; 370 to 372 K) |
| Boiling point | 404.4 °C (759.9 °F; 677.5 K) |
| 1000 g/L (5.02 mol/L) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
acidic |
| Flash point | 198.4 °C (389.1 °F; 471.5 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
2,6-Pyridinedicarbothioic acid (PDTC) is an organosulfur compound that is produced by some bacteria. It functions as a , a low molecular weight compound that scavenges iron. Siderophores solubilize compounds by forming strong complexes. PDTC is secreted by the soil bacteria Pseudomonas stutzeri and Pseudomonas putida. [1]
PDTC can be synthesized in the laboratory by treating the diacid dichloride of pyridine-2,6-dicarboxylic with H2S in pyridine:
- NC5H3(COCl)2 + 2 H2S + 2 C5H5N → [C5H5NH+][HNC5H3(COS)−2] + [C5H5NH]Cl
This route produces the pyridinium salt of pyridinium-2,6-dicarbothioate. Treatment of this orange-colored salt with sulfuric acid gives colorless PDTC, which can then be extracted with dichloromethane. [2]
The biosynthesis of PDTC remains unclear although some insights can be deduced from the genetics. [3] It is suggested that Pseudomonas stutzeri may have acquired at least one of the genes by lateral transfer from mycobacteria. [4] In a proposed biosynthetic sequence pyridine-2,6-dicarboxylic acid, a known bacterial metabolite, [4] is activated as its bis-adenosine monophosphate (AMP) derivative. The sulfur donor and its activation remain uncertain. [5]

PDTC binds to both Fe2+ and Fe3+. The ferric complex is brown, whereas the ferrous complex is blue. In the presence of air, the ferrous complex oxidizes to the ferric compound. [7] It is iron selective [4] as only the Fe complex is soluble in water. PDTC is produced mainly during the exponential phase of bacterial growth. The conditions at which Pseudomonas produces PDTC is 25 °C, pH=8 and sufficient aeration. [5]
- ^ Budzikiewicz, Herbert (2010). "Microbial Siderophores". In Kinghorn, A. Douglas; Falk, Heinz; Kobayashi, Junichi (eds.). Fortschritte der Chemie organischer Naturstoffe / Progress in the Chemistry of Organic Natural Products, Vol. 92 [Progress in the Chemistry of Organic Natural Products]. Vol. 92. pp. 1–75. doi: 10.1007/978-3-211-99661-4_1. ISBN 978-3-211-99660-7. PMID 20198464.
- ^ Hildebrand, U.; Ockels, W.; Lex, J.; Budzikiewicz, H. (1983). "Zur Struktur Eines 1:1-Adduktes von Pyridin-2,6-Dicarbothiosäure und Pyridin". Phosphorus and Sulfur and the Related Elements. 16 (3): 361–364. doi: 10.1080/03086648308080490.
- ^ Cortese, Marc S; Caplan, Allan B; Crawford, Ronald L (2002). "Structural, functional, and evolutionary analysis of moeZ, a gene encoding an enzyme required for the synthesis of the Pseudomonas metabolite, pyridine-2,6-bis(thiocarboxylic acid)". BMC Evolutionary Biology. 2: 8. doi: 10.1186/1471-2148-2-8. PMC 115864. PMID 11972321.
- ^ a b c Cortese, Marc S.; Paszczynski, Andrzej; Lewis, Thomas A.; Sebat, Jonathan L.; Borek, Vladimir; Crawford, Ronald L. (2002). "Metal chelating properties of pyridine-2,6-bis(thiocarboxylic acid) produced by Pseudomonas spp. And the biological activities of the formed complexes". BioMetals. 15 (2): 103–120. doi: 10.1023/A:1015241925322. PMID 12046919. S2CID 5545637.
- ^ a b Budzikiewicz, H. (2003). "Heteroaromatic monothiocarboxylic acids from Pseudomonas spp". Biodegradation. 14 (2): 65–72. doi: 10.1023/A:1024012015127. PMID 12877462. S2CID 29898226.
- ^ Hildebrand, U.; Lex, J.; Taraz, K.; Winkler, S.; Ockels, W.; Budzikiewicz, H. (1984). "Untersuchungen zum Redox-System Bis-(pyridin-2,6-dicarbothioato)-Ferrat(II) /-Ferrat(III) [1]". Zeitschrift für Naturforschung B. 39 (11): 1607–1613. doi: 10.1515/znb-1984-1123. S2CID 94908888.
- ^ Ockels, W., Roemer, A., Budzikiewicz, H., Korth, H., Pulverer, G., "Bacterial constituents. II. An iron(II) complex of pyridine-2,6-di-(monothiocarboxylic acid) - a novel bacterial metabolic product", Tetrahedron Lett. 1978, 3341. doi: 10.1016/S0040-4039(01)85634-3