
| |
| |
| Names | |
|---|---|
|
IUPAC name
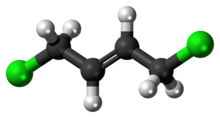
1,4-Dichlorobut-2-ene
| |
| Other names
trans-1,4-Dichlorobutene
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.437 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
| UN number | 2920 2927 2922 |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C4H6Cl2 | |
| Molar mass | 124.99 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.13 g/mL |
| Melting point | 1 °C (34 °F; 274 K) |
| Boiling point | 125.5 °C (257.9 °F; 398.6 K) |
| Hazards [1] | |
| GHS labelling: | |

 

| |
| Danger | |
| H301, H311, H314, H330, H350, H410 | |
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P264, P270, P271, P273, P280, P281, P284, P301+P310, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P307+P311, P308+P313, P310, P312, P314, P320, P321, P322, P330, P361, P363, P370+P378, P391, P403+P233, P403+P235, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
1,4-Dichlorobut-2-ene are organochlorine compounds with the formula ClCH2CH=CHCH2L. Cis and trans isomers exist. These compounds are intermediates in the industrial production of chloroprene. They are main impurity in technical grade chloroprene. [2]
Chloroprene is a monomer for the production of synthetic rubbers such as Neoprene. It is produced from butadiene in a three-step process. The first step is the liquid- or vapour-phase chlorination of butadiene to a mixture of 3,4-dichlorobut-1-ene and 1,4-dichlorobut-2-ene (both isomers). In the second step, the mixture of 1,4-dichlorobut-2-ene and 3,4-dichlorobut-1-ene is isomerized to 3,4-dichlorobut-1-ene by heating to temperatures of 60–120 °C in the presence of a catalyst. Finally, dehydrochlorination ( elimination of hydrogen chloride) of 3,4-dichlorobut-1-ene with dilute sodium hydroxide solution in the presence of polymerization inhibitors gives crude chloroprene. [3]
1,4-Dichlorobut-2-ene is a precursor to various heterocycles. [4]
The (E)-isomer is also one of the starting materials for the poriferic natural product sceptrin. [5]
- ^ Index no. 602-073-00-X of Annex VI, Part 3, to Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006. OJEU L353, 31.12.2008, pp 1–1355 at p 473.
- ^ Re-evaluation of Some Organic Chemicals, Hydrazine and Hydrogen Peroxide (PDF), IARC Monographs on the Evaluation of Carcinogenic Risks to Humans 71, Lyon, France: International Agency for Research on Cancer, 1999, pp. 227–50, ISBN 92-832-1271-1.
-
^ Kleinschmidt, P. (1986), "Chlorinated hydrocarbons. 6.4. 2-Chloro-1,3-butadiene", in Gerhartz, W.; Yamamoto, Y. S. (eds.), Ullmann's Encyclopedia of Industrial Chemistry, vol. A6 (5th ed.), Weinheim: VCH, pp. 315–18
Stewart, C. A. Jr. (1993), "Chlorocarbons, -hydrocarbons (chloroprene)", in Kroschwitz, J. I.; Howe-Grant, M. (eds.), Kirk-Othmer Encyclopedia of Chemical Technology, vol. 6 (4th ed.), New York: John Wiley, pp. 70–78. -
^ Albert I. Meyers, Joseph S. Warmus, Garrett J. Dilley (1996). "3-Pyrroline". Organic Syntheses. 73: 246.
doi:
10.15227/orgsyn.073.0246.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Birman, Vladimir B.; Jiang, Xun-Tian (2004), "Synthesis of Sceptrin Alkaloids", Org. Lett., 6 (14): 2369–71, doi: 10.1021/ol049283g, PMID 15228281. Baran, Phil S.; Zografos, Alexandros L.; O'Malley, Daniel P. (2004), "Short Total Synthesis of (±)-Sceptrin", J. Am. Chem. Soc., 126 (12): 3726–27, doi: 10.1021/ja049648s, PMID 15038721.

| |

| |
| Names | |
|---|---|
|
IUPAC name
1,4-Dichlorobut-2-ene
| |
| Other names
trans-1,4-Dichlorobutene
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.437 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
| UN number | 2920 2927 2922 |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C4H6Cl2 | |
| Molar mass | 124.99 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.13 g/mL |
| Melting point | 1 °C (34 °F; 274 K) |
| Boiling point | 125.5 °C (257.9 °F; 398.6 K) |
| Hazards [1] | |
| GHS labelling: | |

 

| |
| Danger | |
| H301, H311, H314, H330, H350, H410 | |
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P264, P270, P271, P273, P280, P281, P284, P301+P310, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P307+P311, P308+P313, P310, P312, P314, P320, P321, P322, P330, P361, P363, P370+P378, P391, P403+P233, P403+P235, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
1,4-Dichlorobut-2-ene are organochlorine compounds with the formula ClCH2CH=CHCH2L. Cis and trans isomers exist. These compounds are intermediates in the industrial production of chloroprene. They are main impurity in technical grade chloroprene. [2]
Chloroprene is a monomer for the production of synthetic rubbers such as Neoprene. It is produced from butadiene in a three-step process. The first step is the liquid- or vapour-phase chlorination of butadiene to a mixture of 3,4-dichlorobut-1-ene and 1,4-dichlorobut-2-ene (both isomers). In the second step, the mixture of 1,4-dichlorobut-2-ene and 3,4-dichlorobut-1-ene is isomerized to 3,4-dichlorobut-1-ene by heating to temperatures of 60–120 °C in the presence of a catalyst. Finally, dehydrochlorination ( elimination of hydrogen chloride) of 3,4-dichlorobut-1-ene with dilute sodium hydroxide solution in the presence of polymerization inhibitors gives crude chloroprene. [3]
1,4-Dichlorobut-2-ene is a precursor to various heterocycles. [4]
The (E)-isomer is also one of the starting materials for the poriferic natural product sceptrin. [5]
- ^ Index no. 602-073-00-X of Annex VI, Part 3, to Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006. OJEU L353, 31.12.2008, pp 1–1355 at p 473.
- ^ Re-evaluation of Some Organic Chemicals, Hydrazine and Hydrogen Peroxide (PDF), IARC Monographs on the Evaluation of Carcinogenic Risks to Humans 71, Lyon, France: International Agency for Research on Cancer, 1999, pp. 227–50, ISBN 92-832-1271-1.
-
^ Kleinschmidt, P. (1986), "Chlorinated hydrocarbons. 6.4. 2-Chloro-1,3-butadiene", in Gerhartz, W.; Yamamoto, Y. S. (eds.), Ullmann's Encyclopedia of Industrial Chemistry, vol. A6 (5th ed.), Weinheim: VCH, pp. 315–18
Stewart, C. A. Jr. (1993), "Chlorocarbons, -hydrocarbons (chloroprene)", in Kroschwitz, J. I.; Howe-Grant, M. (eds.), Kirk-Othmer Encyclopedia of Chemical Technology, vol. 6 (4th ed.), New York: John Wiley, pp. 70–78. -
^ Albert I. Meyers, Joseph S. Warmus, Garrett J. Dilley (1996). "3-Pyrroline". Organic Syntheses. 73: 246.
doi:
10.15227/orgsyn.073.0246.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Birman, Vladimir B.; Jiang, Xun-Tian (2004), "Synthesis of Sceptrin Alkaloids", Org. Lett., 6 (14): 2369–71, doi: 10.1021/ol049283g, PMID 15228281. Baran, Phil S.; Zografos, Alexandros L.; O'Malley, Daniel P. (2004), "Short Total Synthesis of (±)-Sceptrin", J. Am. Chem. Soc., 126 (12): 3726–27, doi: 10.1021/ja049648s, PMID 15038721.