| |
| Names | |
|---|---|
|
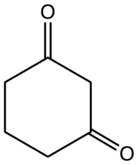
Preferred IUPAC name
Cyclohexane-1,3-dione | |
| Other names
CHD, dihydroresorcinol
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 385888 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.255 |
| EC Number |
|
| 200899 | |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H8O2 | |
| Molar mass | 112.128 g·mol−1 |
| Appearance | Colorless or white solid |
| Density | 1.0861 g/cm3 |
| Melting point | 105.5 °C (221.9 °F; 378.6 K) |
| Acidity (pKa) | 5.20 (H2O) [1] |
| Hazards | |
| GHS labelling: | |


| |
| Danger | |
| H302, H318, H412 | |
| P264, P270, P273, P280, P301+P312, P305+P351+P338, P310, P330, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
1,3-Cyclohexanedione is an organic compound with the formula (CH2)4(CO)2. It is one of three isomeric cyclohexanediones. It is a colorless compound that occurs naturally. It is the substrate for cyclohexanedione hydrolase. The compound exists mainly as the enol tautomer. [2]
Synthesis, structure, and reactivity
1,3-Cyclohexanedione is produced by semi- hydrogenation of resorcinol: [3] [4]
- C6H4(OH)2 + H2 → C6H8O2
1,3-Cyclohexanedione exists in solution predominantly as the enol tautomer.
It reacts under acid catalysis with alcohols to 3-alkoxyenones. [2] Its pKa is 5.26. Treatment of the sodium salt of the enolate with methyl iodide gives 2-methyl-1,3-cyclohexanedione, which also exists predominantly as the enol. [4]
Derivatives
Dimedone (5,5-dimethyl-1,3-cyclohexanedione) is a well established reagent. [5]
Several herbicides against grasses are formal derivatives of 1,3-cyclohexanedione. Examples of commercial products include cycloxydim, clethodim, tralkoxydim, butroxydim, sethoxydim, profoxydim, and mesotrione. [6]
References
- ^ Terasawa, Tadao; Okada, Toshihiko (1977). "Novel heterocyclic synthons. Synthesis and properties of thia- and oxacyclohexane-3,5-diones". J. Org. Chem. 42 (7): 1163–1169. doi: 10.1021/jo00427a014.
- ^ a b Guppi, Sanjeeva Rao; O'Doherty, George A. (2008). "1,3-Cyclohexadiene". E-EROS Encyclopedia of Reagents for Organic Synthesis. doi: 10.1002/047084289X.rn00921. ISBN 978-0471936237.
- ^ Thompson, R. B. (1947). "Dihydroresorcinol". Org. Synth. 27: 21. doi: 10.15227/orgsyn.027.0021.
- ^ a b Mekler, A. B.; Ramachandran, S.; Swaminathan, S.; Newman, Melvin S. (1961). "Methyl-1,3-Cyclohexanedione". Org. Synth. 41: 56. doi: 10.15227/orgsyn.041.0056.
- ^ Strittmatter, Harald; Hildbrand, Stefan; Pollak, Peter (2007). "Malonic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. doi: 10.1002/14356007.a16_063.pub2. ISBN 978-3527306732.
- ^ Keith G. Watson (2011). "Cyclohexane-1,3-dione Oxime Ether Grass-Specific Herbicides and the Discovery of Butroxydim". Aust. J. Chem. 64 (4): 367–372. doi: 10.1071/CH10366.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
Cyclohexane-1,3-dione | |
| Other names
CHD, dihydroresorcinol
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 385888 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.255 |
| EC Number |
|
| 200899 | |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H8O2 | |
| Molar mass | 112.128 g·mol−1 |
| Appearance | Colorless or white solid |
| Density | 1.0861 g/cm3 |
| Melting point | 105.5 °C (221.9 °F; 378.6 K) |
| Acidity (pKa) | 5.20 (H2O) [1] |
| Hazards | |
| GHS labelling: | |


| |
| Danger | |
| H302, H318, H412 | |
| P264, P270, P273, P280, P301+P312, P305+P351+P338, P310, P330, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
1,3-Cyclohexanedione is an organic compound with the formula (CH2)4(CO)2. It is one of three isomeric cyclohexanediones. It is a colorless compound that occurs naturally. It is the substrate for cyclohexanedione hydrolase. The compound exists mainly as the enol tautomer. [2]
Synthesis, structure, and reactivity
1,3-Cyclohexanedione is produced by semi- hydrogenation of resorcinol: [3] [4]
- C6H4(OH)2 + H2 → C6H8O2
1,3-Cyclohexanedione exists in solution predominantly as the enol tautomer.
It reacts under acid catalysis with alcohols to 3-alkoxyenones. [2] Its pKa is 5.26. Treatment of the sodium salt of the enolate with methyl iodide gives 2-methyl-1,3-cyclohexanedione, which also exists predominantly as the enol. [4]
Derivatives
Dimedone (5,5-dimethyl-1,3-cyclohexanedione) is a well established reagent. [5]
Several herbicides against grasses are formal derivatives of 1,3-cyclohexanedione. Examples of commercial products include cycloxydim, clethodim, tralkoxydim, butroxydim, sethoxydim, profoxydim, and mesotrione. [6]
References
- ^ Terasawa, Tadao; Okada, Toshihiko (1977). "Novel heterocyclic synthons. Synthesis and properties of thia- and oxacyclohexane-3,5-diones". J. Org. Chem. 42 (7): 1163–1169. doi: 10.1021/jo00427a014.
- ^ a b Guppi, Sanjeeva Rao; O'Doherty, George A. (2008). "1,3-Cyclohexadiene". E-EROS Encyclopedia of Reagents for Organic Synthesis. doi: 10.1002/047084289X.rn00921. ISBN 978-0471936237.
- ^ Thompson, R. B. (1947). "Dihydroresorcinol". Org. Synth. 27: 21. doi: 10.15227/orgsyn.027.0021.
- ^ a b Mekler, A. B.; Ramachandran, S.; Swaminathan, S.; Newman, Melvin S. (1961). "Methyl-1,3-Cyclohexanedione". Org. Synth. 41: 56. doi: 10.15227/orgsyn.041.0056.
- ^ Strittmatter, Harald; Hildbrand, Stefan; Pollak, Peter (2007). "Malonic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. doi: 10.1002/14356007.a16_063.pub2. ISBN 978-3527306732.
- ^ Keith G. Watson (2011). "Cyclohexane-1,3-dione Oxime Ether Grass-Specific Herbicides and the Discovery of Butroxydim". Aust. J. Chem. 64 (4): 367–372. doi: 10.1071/CH10366.
