| |

| |
| Names | |
|---|---|
|
Preferred IUPAC name
(Propane-1,3-diyl)bis(diphenylphosphane) | |
| Identifiers | |
3D model (
JSmol)
|
|
| Abbreviations | DPPP |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.027.084 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C27H26P2 | |
| Molar mass | 412.453 g·mol−1 |
| Appearance | white solid |
| chlorocarbons | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
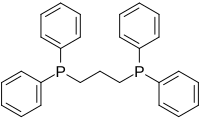
1,3-Bis(diphenylphosphino)propane (dppp) is an organophosphorus compound with the formula Ph2P(CH2)3PPh2. The compound is a white solid that is soluble in organic solvents. It is slightly air-sensitive, degrading in air to the phosphine oxide. It is classified as a diphosphine ligand in coordination chemistry and homogeneous catalysis.
The diphosphine can be prepared by the reaction of lithium diphenylphosphide and 1,3-dichloropropane (Ph = C6H5):
- 2 Ph2PLi + Cl(CH2)3Cl → Ph2P(CH2)3PPh2 + 2 LiCl
However, it can be synthesised via a much more controllable (and cheaper) route, via metal-halogen exchange and then metathesis:
- Br(CH2)3Br + 2 tBuLi → Li(CH2)3Li + 2 tBuBr
- Li(CH2)3Li + 2 PCl3 → Cl2P(CH2)3PCl2 + 2 LiCl
- Cl2P(CH2)3PCl2 + 4 PhLi → Ph2P(CH2)3PPh2 + 4 LiCl
The diphosphine serves as a bidentate ligand forming six-membered C3P2M chelate ring with a natural bite angle of 91°. [1] For example, the complex dichloro(1,3-bis(diphenylphosphino)propane)nickel is prepared by combining equimolar portions of the ligand and nickel(II) chloride hexahydrate. This nickel complex serves as a catalyst for the Kumada coupling reaction. [2] Dppp is also used as a ligand for palladium(II) catalysts to co-polymerize carbon monoxide and ethylene to give polyketones. [3] Dppp can sometimes be used in palladium-catalyzed arylation under Heck reaction conditions to control regioselectivity. [4]
- ^ Birkholz (née Gensow), Mandy-Nicole; Freixa, Zoraida; van Leeuwen, Piet W. N. M. (2009). "Bite angle effects of diphosphines in C–C and C–X bond forming cross coupling reactions". Chemical Society Reviews. 38 (4): 1099–1118. doi: 10.1039/B806211K. PMID 19421583.
- ^ Kumada, Makota; Tamao, Kohei; Sumitani, Koji (1988). "Phosphine-Nickel Complex Catalyzed Cross-Coupling of Grignard Reagents with Aryl and Alkenyl Halides: 1,2-dibutylbenzene". Organic Syntheses; Collected Volumes, vol. 6, p. 407.
- ^ Drent, E.; Mul, W. P.; Smaardijk, A. A. (2001). "Polyketones". Encyclopedia Of Polymer Science and Technology. doi: 10.1002/0471440264.pst273. ISBN 9781118633892.
- ^ Cabri, Walter; Candiani, Ilaria; Bedeschi Angelo; Penco, Sergio"α-Regioselectivity in Palladium-Catalyzed Arylation of Acyclic Enol Ethers" journal= Journal of Organic Chemistry, 1991, volume 57, p. 1481. doi: 10.1021/jo00031a029

| |

| |
| Names | |
|---|---|
|
Preferred IUPAC name
(Propane-1,3-diyl)bis(diphenylphosphane) | |
| Identifiers | |
3D model (
JSmol)
|
|
| Abbreviations | DPPP |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.027.084 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C27H26P2 | |
| Molar mass | 412.453 g·mol−1 |
| Appearance | white solid |
| chlorocarbons | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
1,3-Bis(diphenylphosphino)propane (dppp) is an organophosphorus compound with the formula Ph2P(CH2)3PPh2. The compound is a white solid that is soluble in organic solvents. It is slightly air-sensitive, degrading in air to the phosphine oxide. It is classified as a diphosphine ligand in coordination chemistry and homogeneous catalysis.
The diphosphine can be prepared by the reaction of lithium diphenylphosphide and 1,3-dichloropropane (Ph = C6H5):
- 2 Ph2PLi + Cl(CH2)3Cl → Ph2P(CH2)3PPh2 + 2 LiCl
However, it can be synthesised via a much more controllable (and cheaper) route, via metal-halogen exchange and then metathesis:
- Br(CH2)3Br + 2 tBuLi → Li(CH2)3Li + 2 tBuBr
- Li(CH2)3Li + 2 PCl3 → Cl2P(CH2)3PCl2 + 2 LiCl
- Cl2P(CH2)3PCl2 + 4 PhLi → Ph2P(CH2)3PPh2 + 4 LiCl
The diphosphine serves as a bidentate ligand forming six-membered C3P2M chelate ring with a natural bite angle of 91°. [1] For example, the complex dichloro(1,3-bis(diphenylphosphino)propane)nickel is prepared by combining equimolar portions of the ligand and nickel(II) chloride hexahydrate. This nickel complex serves as a catalyst for the Kumada coupling reaction. [2] Dppp is also used as a ligand for palladium(II) catalysts to co-polymerize carbon monoxide and ethylene to give polyketones. [3] Dppp can sometimes be used in palladium-catalyzed arylation under Heck reaction conditions to control regioselectivity. [4]
- ^ Birkholz (née Gensow), Mandy-Nicole; Freixa, Zoraida; van Leeuwen, Piet W. N. M. (2009). "Bite angle effects of diphosphines in C–C and C–X bond forming cross coupling reactions". Chemical Society Reviews. 38 (4): 1099–1118. doi: 10.1039/B806211K. PMID 19421583.
- ^ Kumada, Makota; Tamao, Kohei; Sumitani, Koji (1988). "Phosphine-Nickel Complex Catalyzed Cross-Coupling of Grignard Reagents with Aryl and Alkenyl Halides: 1,2-dibutylbenzene". Organic Syntheses; Collected Volumes, vol. 6, p. 407.
- ^ Drent, E.; Mul, W. P.; Smaardijk, A. A. (2001). "Polyketones". Encyclopedia Of Polymer Science and Technology. doi: 10.1002/0471440264.pst273. ISBN 9781118633892.
- ^ Cabri, Walter; Candiani, Ilaria; Bedeschi Angelo; Penco, Sergio"α-Regioselectivity in Palladium-Catalyzed Arylation of Acyclic Enol Ethers" journal= Journal of Organic Chemistry, 1991, volume 57, p. 1481. doi: 10.1021/jo00031a029