| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
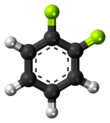
1,2-Difluorobenzene | |||
| Other names
o-Difluorobenzene
ortho-Difluorobenzene | |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.006.074 | ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C6H4F2 | |||
| Molar mass | 114.093 g/mol | ||
| Appearance | colorless liquid | ||
| Density | 1.1599 g/cm3 | ||
| Melting point | −34 °C (−29 °F; 239 K) | ||
| Boiling point | 92 °C (198 °F; 365 K) | ||
| (insoluble) 1.14 g/L | |||
| Related compounds | |||
Related compounds
|
1,2-Dichlorobenzene | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
1,2-Difluorobenzene, also known as DFB, is an aromatic compound with formula C6H4F2. This colorless flammable liquid is a solvent used in the electrochemical studies of transition metal complexes. Compared to most conventional halogenated aliphatic and aromatic solvents, it possesses an exceptionally high dielectric constant (ε0 = 13.8 at 300 K). Thus, it can be a suitable solvent for cationic, and/or highly electrophilic organometallic complexes. [2]
Difluorobenzenes can be prepared by the Balz-Schiemann reaction, which entails conversion of diazonium tetrafluoroborate salts to their fluorides. The synthesis of 1,2-difluorobenzene starts with 2- fluoroaniline: [3]
- C6H4F(NH2) + HNO2 + HBF4 → [C6H4F(N2)]BF4 + 2 H2O
- [C6H4F(N2)]BF4 → C6H4F2 + N2 + BF3
The syntheses of 1,3- and 1,4-difluorobenzene proceed respectively from 1,3- and 1,4-diaminobenzene, which are doubly diazotized. [4]
Organometallic derivatives of 1,2-difluorobenzene have been well developed. It is found to be a weaker base than benzene. [5]
1,2-Difluorobenzene has been used as solvent for the electrochemical analysis of transition metal complexes. It is relatively chemically inert, weakly coordinating and has a relatively high dielectric constant. It is a weakly coordinating for metal complexes, in contrast to acetonitrile, DMSO, and DMF. [6]
It has anaesthetic properties. [7]
1,2-Difluorobenzene can be acylated to 3',4'-difluoropropiophenone. [8]
- ^ David R. Lide, ed., CRC Handbook of Chemistry and Physics, 89th Edition (Internet Version 2009), CRC Press/Taylor and Francis, Boca Raton, FL.
- ^ Pike, Sebastian D.; Crimmin, Mark R.; Chaplin, Adrian B. (2017). "Organometallic chemistry using partially fluorinated benzenes" (PDF). Chemical Communications. 53 (26): 3615–3633. doi: 10.1039/C6CC09575E. PMID 28304406.
- ^ Yu, Zhiqun; Lv, Yanwen; Yu, Chuanming (2012). "A Continuous Kilogram-Scale Process for the Manufacture of o-Difluorobenzene". Organic Process Research & Development. 16 (10): 1669–1672. doi: 10.1021/op300127x.
- ^ Siegemund, Günter; Schwertfeger, Werner; Feiring, Andrew; Smart, Bruce; Behr, Fred; Vogel, Herward; McKusick, Blaine (2000). "Fluorine Compounds, Organic". Ullmann's Encyclopedia of Industrial Chemistry. doi: 10.1002/14356007.a11_349. ISBN 3527306730.
- ^ Pike, Sebastian D.; Crimmin, Mark R.; Chaplin, Adrian B. (2017). "Organometallic chemistry using partially fluorinated benzenes" (PDF). Chemical Communications. 53 (26): 3615–3633. doi: 10.1039/c6cc09575e. PMID 28304406.
- ^ O'toole, Terrence R.; Younathan, Janet N.; Sullivan, B. Patrick; Meyer, Thomas J. (1989). "1,2-Difluorobenzene: a relatively inert and noncoordinating solvent for electrochemical studies on transition-metal complexes". Inorganic Chemistry. 28 (20): 3923. doi: 10.1021/ic00319a032.
- ^ "1,2-Difluorobenzene". PubChem. Retrieved 11 February 2021.
- ^ GB 1140754, Danilewicz, John Christopher & Szelke, Michael, "3,4-difluorophenyl compounds", published 1969-01-22, assigned to Pfizer Ltd.
| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
1,2-Difluorobenzene | |||
| Other names
o-Difluorobenzene
ortho-Difluorobenzene | |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.006.074 | ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C6H4F2 | |||
| Molar mass | 114.093 g/mol | ||
| Appearance | colorless liquid | ||
| Density | 1.1599 g/cm3 | ||
| Melting point | −34 °C (−29 °F; 239 K) | ||
| Boiling point | 92 °C (198 °F; 365 K) | ||
| (insoluble) 1.14 g/L | |||
| Related compounds | |||
Related compounds
|
1,2-Dichlorobenzene | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
1,2-Difluorobenzene, also known as DFB, is an aromatic compound with formula C6H4F2. This colorless flammable liquid is a solvent used in the electrochemical studies of transition metal complexes. Compared to most conventional halogenated aliphatic and aromatic solvents, it possesses an exceptionally high dielectric constant (ε0 = 13.8 at 300 K). Thus, it can be a suitable solvent for cationic, and/or highly electrophilic organometallic complexes. [2]
Difluorobenzenes can be prepared by the Balz-Schiemann reaction, which entails conversion of diazonium tetrafluoroborate salts to their fluorides. The synthesis of 1,2-difluorobenzene starts with 2- fluoroaniline: [3]
- C6H4F(NH2) + HNO2 + HBF4 → [C6H4F(N2)]BF4 + 2 H2O
- [C6H4F(N2)]BF4 → C6H4F2 + N2 + BF3
The syntheses of 1,3- and 1,4-difluorobenzene proceed respectively from 1,3- and 1,4-diaminobenzene, which are doubly diazotized. [4]
Organometallic derivatives of 1,2-difluorobenzene have been well developed. It is found to be a weaker base than benzene. [5]
1,2-Difluorobenzene has been used as solvent for the electrochemical analysis of transition metal complexes. It is relatively chemically inert, weakly coordinating and has a relatively high dielectric constant. It is a weakly coordinating for metal complexes, in contrast to acetonitrile, DMSO, and DMF. [6]
It has anaesthetic properties. [7]
1,2-Difluorobenzene can be acylated to 3',4'-difluoropropiophenone. [8]
- ^ David R. Lide, ed., CRC Handbook of Chemistry and Physics, 89th Edition (Internet Version 2009), CRC Press/Taylor and Francis, Boca Raton, FL.
- ^ Pike, Sebastian D.; Crimmin, Mark R.; Chaplin, Adrian B. (2017). "Organometallic chemistry using partially fluorinated benzenes" (PDF). Chemical Communications. 53 (26): 3615–3633. doi: 10.1039/C6CC09575E. PMID 28304406.
- ^ Yu, Zhiqun; Lv, Yanwen; Yu, Chuanming (2012). "A Continuous Kilogram-Scale Process for the Manufacture of o-Difluorobenzene". Organic Process Research & Development. 16 (10): 1669–1672. doi: 10.1021/op300127x.
- ^ Siegemund, Günter; Schwertfeger, Werner; Feiring, Andrew; Smart, Bruce; Behr, Fred; Vogel, Herward; McKusick, Blaine (2000). "Fluorine Compounds, Organic". Ullmann's Encyclopedia of Industrial Chemistry. doi: 10.1002/14356007.a11_349. ISBN 3527306730.
- ^ Pike, Sebastian D.; Crimmin, Mark R.; Chaplin, Adrian B. (2017). "Organometallic chemistry using partially fluorinated benzenes" (PDF). Chemical Communications. 53 (26): 3615–3633. doi: 10.1039/c6cc09575e. PMID 28304406.
- ^ O'toole, Terrence R.; Younathan, Janet N.; Sullivan, B. Patrick; Meyer, Thomas J. (1989). "1,2-Difluorobenzene: a relatively inert and noncoordinating solvent for electrochemical studies on transition-metal complexes". Inorganic Chemistry. 28 (20): 3923. doi: 10.1021/ic00319a032.
- ^ "1,2-Difluorobenzene". PubChem. Retrieved 11 February 2021.
- ^ GB 1140754, Danilewicz, John Christopher & Szelke, Michael, "3,4-difluorophenyl compounds", published 1969-01-22, assigned to Pfizer Ltd.