| |
| Names | |
|---|---|
|
Preferred IUPAC name
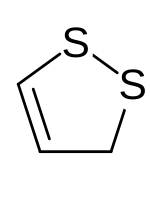
3H-1,2-Dithiole | |
Other names
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C3H4S2 | |
| Molar mass | 104.19 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
In organosulfur chemistry, a 1,2-dithiole is a type of heterocycle. The parent of this class of compounds is 1,2-dithiacyclopentene. The anticancer drug oltipraz is a dithiole. [1]
See also
References
- ^ Ogurtsov, Vladimir A; Rakitin, Oleg A (2012). "Reactivity of 1,2-dithioles". Russian Chemical Reviews. 81 (7): 638. Bibcode: 2012RuCRv..81..638O. doi: 10.1070/RC2012v081n07ABEH004231. S2CID 97817545.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
3H-1,2-Dithiole | |
Other names
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C3H4S2 | |
| Molar mass | 104.19 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
In organosulfur chemistry, a 1,2-dithiole is a type of heterocycle. The parent of this class of compounds is 1,2-dithiacyclopentene. The anticancer drug oltipraz is a dithiole. [1]
See also
References
- ^ Ogurtsov, Vladimir A; Rakitin, Oleg A (2012). "Reactivity of 1,2-dithioles". Russian Chemical Reviews. 81 (7): 638. Bibcode: 2012RuCRv..81..638O. doi: 10.1070/RC2012v081n07ABEH004231. S2CID 97817545.
