| |
| Names | |
|---|---|
|
Preferred IUPAC name
Cyclopentane-1,2,3,4,5-pentol | |
| Other names
1,2,3,4,5-Cyclopentanepentol
1,2,3,4,5-Pentahydroxycyclopentane | |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| Properties | |
| C5H10O5 | |
| Molar mass | 150.130 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
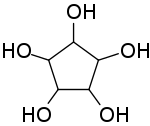
1,2,3,4,5-Cyclopentanepentol, also named cyclopentane-1,2,3,4,5-pentol or 1,2,3,4,5-pentahydroxycyclopentane is a
chemical compound with formula C
5H
10O
5 or (–CHOH–)
5, whose
molecule consists of a ring of five
carbon atoms (a
cyclopentane skeleton), each connected to one
hydrogen and one
hydroxyl
group.
[1] The unqualified term "cyclopentanepentol" usually refers to this compound. There are four distinct
stereoisomers with this same structure.
[2]
The compound is a five-fold alcohol of cyclopentane, and technically a cyclic sugar alcohol (a cyclitol). However it is very rarely found in nature, and therefore it has received much less attention than the ubiquitous six-carbon version, inositol. [3]
There are four distinct stereoisomers of cyclopentane-1,2,3,4,5-pentol, distinguished by the position of the hydroxyls relative to the mean plane of the ring. All have a plane of symmetry, and therefore are not chiral. One naming convention for the isomers labels each carbon number 1-5 with "α" for the side of the ring plane with most hydroxyls (three or more), and "β" for the other side. Another convention lists the hydroxyls on the majority side and then those on the minority side, with the two groups separated by a slash (and a "0" is written when the second list would be empty). [4]
The four possible isomers are: [2]
- 1α,2α,3α,4α,5α, 1,2,3,4,5/0, or all-cis (all hydroxyls on the same side). CAS 34322-89-9. Long needles, melting point 283 °C (darkens ar 220 °C). [2] [5] Obtained from cyclopentane-1α,2α-epoxi-3α,4α,5α-triol by treatment with hydrogen bromide to obtain cyclopentane-1β-bromo-2α,3α,4α,5α-tetrol, followed by benzoylation and treatment with DMSO and sodium bicarbonate. [5]
- 1α,2α,3α,4α,5β or 1,2,3,4/5. CAS 18939-02-1. [2] Soluble in hot ethanol. [3] Melting point 220 °C [3] or 209-210 °C. [6] Obtained by solvolysis of two tetra-O-acetyl-O- tosyl-cyclopentanepentol, [3] and also by hydroxylation of 3,4,5-trihydroxy-1-cyclopentenes. [6]
- 1α,2α,3α,4β,5β or 1,2,3/4,5. CAS 18939-07-6. [2] Soluble in ethanol. [3] Melting point 168-169 °C, [3] or 176 °C. [6] Obtained by deamination of (1,4/2,3,5)-5-amino-1,3-di-O-acetyl-2,3-O-cyclohexylidene-l,2,3,4-cycopentanetetrol, [3] and also by hydroxylation of 3,4,5-trihydroxy-1-cyclopentenes. [6]
- 1α,2α,3β,4α,5β or 1,2,4/3,5. CAS 57784-52-8. [2] Melting point 149.5-150.5 °C. [3] Obtained from cyclopent-4-ene-1,3/2-trio1 by epoxidation and subsequent hydrolysis; [7] also by acid hydrolysis of an anhydro-cyclopentanepentol, DL-1,2-anhydro-4,5-O-cyclohexylidene-1,2,3/4,5-cyclopentanepentol; [3] and also from a D-xylo-pento dialdose. [8]
The last three isomers interconvert by heating them at about 104 °C with 95% acetic acid in the presence of a strong acid. The conversion between 1,2,3,4/5 and 1,2,3/4,5 is faster and entails the hydrogen and hydroxyl switching places on either of the two carbon atoms located between an "α" and a "β" position. The formation of 1,2,4/3,5 is much slower and equilibrium is reached only after many days. The relative stabilities are 1,2,4/3,5 > 1,2,3/4,5 > 1,2,3,4/5, with equilibrium ratios 72 : 17.5 : 10.5. [3]
The isomers can be identified qualitatively by their mobility in paper ionophoresis in a solution of calcium acetate and acetic acid, and revealed with a manganese sulfate/ potassium permanganate reagent. The mobilities of the isomers, relative to that of cis-inositol, are 0.95 (12345/0), 0.44 (1234/5), 0.18 (123/45), and 0.04 (124/35). The variation is attributed to the formation of chelates with the calcium cations, with varying strengths depending on the number of hydroxyl pairs that can bind to the cation. [9] Similar results can be obtained with lanthanum cations and thin-layer chromatography. [10]
Mass spectrometry of the compound generates mainly the ion HO–CH=CH=CH=O+H (mass-charge ratio 73) and a neutral radical C
2H
5O•.
[11]
Many esters are known, such as penta benzoyls [2] and penta acetyls. [3]
Trteatment of the derivative 3,4,5-tri-O-acetyl-1,2-O-ethylidene-(1,2,4/3,5)-cyclopentanepentol with
triphenylcarbenium
tetrafluoroborate [(C
6H
5)
3C]+
[BF
4−
to yield the cation 3,4,5-tri-O-acetyl-(1,2,4/3,5)-cyclopentanepentol-1,2-O-acetoxonium, which exhibits a peculiar 10-stage cyclic rearrangement.
[12]
Cyclopentanepentols form dihydrogen phosphate esters analogous to inositol's phytic acid esters. [13]
The related compound 1,2,3,4,5-pentakis(hydroxymethyl)-cyclopentane (a colourless viscous oil, soluble in tert-butanol and DMSO) was synthesized in 1985 by L. M. Tolbert and others. It was hoped that dehydrative dimerization of this product would yield dodecahedrane. [14]
Organic compounds similar to (and possibly mimicking) glycoglycerolipids, [15] with a 1,2,4/3,5-cyclopentanepentol group connected to the glycerol core in place of the sugar group, have been found in several genera of marine sponges with widely separate ranges. [16] [17] [18] [19] [20] [21]
Calditol is a substance found in the archaeobacteria of the order Sulfolobales. It is an ether that can be viewed as the condensation of glycerol and 3α-hydroxymethyl-1α,2α,3β,4α,5β-hydroxy cyclopentane, forming an ether bridge –O– between the 1-carbon of the former and the 4-carbon of the latter. [22] Its seems to protects the cells in acidic environments. [23]
Trace amounts of cyclopentanepentol were identified in some plants, such as the red flower calyces of Hibiscus sabdariffa (roselle); [24] the plant Maclura pomifera (Osage orange); [25] and the cyanobacterium Oscillatoria willei [26] as well as in the products of its hydropyrolysis. [27]
The 1,2,4/3,5 isomer was briefly described in 1963 by H. Z. Sable and others. [7] In 1968, Th. Posternak reported observation of 1,2,3,4/5 and 1,2,3/4,5, without the synthesis method. [28] Better synthesis methods for these three were published by S. J. Angyal and B. M. Luttrell in 1970. [3] Alternative methods for 1,2,3,4/5 and 1,2,3/4,5 were published in the same year by G. Wolczunowicz and others. [6] The remaining all-cis isomer (1,2,3,4,5/0) was synthesized in 1971 by F. G. Cocu and Posternak. [5]
- ^ (2019) Entry 552295: 1,2,3,4-Cyclopentanepentol. Page at the PubChem website, accessed on 2019-07-23.
- ^ a b c d e f g Peter M. Collins (2005): Dictionary of Carbohydrates, 2nd edition, page 268. CRC Press, 1304 pages. ISBN 9780849377655
- ^ a b c d e f g h i j k l S. J. Angyal and B. M. Luttrell (1970): "Cyclitols. XXXII. Cyclopentanepentols". Australian Journal of Chemistry, volume 23, issue 9, pages 1831-1838. doi: 10.1071/CH9701831
- ^ CON and CBN IUPAC Commissions on Nomenclature (1968): "The Nomenclature of Cyclitols - Tentative Rules". European Journal of Biochemistry, volume 5, pages 1-12. doi: 10.1111/j.1432-1033.1968.tb00328.x
- ^ a b c F. G. Cocu, Th. Posternak (1971): "Recherches dans la série des cyclitols XLII. Synthèses de cyclitols tout-cis dérivés du cyclopentane; préparation des cyclopentane-tétrol et - pentol tout-cis" Helvetica Chimica Acta, volume 54, issue 6, pages 1676-1687. doi: 10.1002/hlca.19710540619
- ^ a b c d e G. Wolczunowicz, L. Bors, F. Cocu, Th. Posternak (1970): "Recherches dans la série des cyclitols, XLI. Synthèses de deux cyclopentane-pentols". Helvetica Chimica Acta, volume 53, issue 8, pages 2288-2295. doi: 10.1002/hlca.19700530843
- ^ a b H. Z. Sable, Thelma Anderson, Bernadine Tolbert, Th. Posternak (1963): "Recherches dans la série des cyclitols XXXIII. Sur quelques cyclitols dérivés du cyclopentane". Helvetica Chimica Acta, volume 46, issue 4, pages 1157–1165. doi: 10.1002/hlca.19630460410
- ^ Barry V. L. Potter (1998) "Structure-Activity Relationships of Adenophostin A and Related Molecules at the 1-D-myo-Inositol 1,4,5-Trisphosphate Receptor". American Chemical Society Symposium Series, volume 718, chaper 10, pages 158-179 doi: 10.1021/bk-1999-0718.ch010
- ^ S. J. Angyal and J. A. Mills (1979): "Complexes of Carbohydrates with Metal Cations. XI. Paper Electrophoresis of Polyols in Solutions of Calcium Ions". Australian Journal of Chemistry, volume 32, issue 9, pages 1993–2001. doi: 10.1071/CH9791993
- ^ S. J. Angyal and J. A. Mills (1985): "Complexes of Carbohydrates with Metal Cations. XIV. Separation of Sugars and Alditols by Means of Their Lanthanum Complexes". Australian Journal of Chemistry, volume 38, issue 8, pages 1279–1285. doi: 10.1071/CH9851279
- ^ Gérard A. Singy and Armand Buchs (1975): "Etude par spectrométrie de masse de la fragmentation des cyclopentane-triols-1,2,3, des cyclopentane-tétrols et des cyclopentane-pentols". Helvetica Chimica Acta, volume 58, issue 7, pages 2164-2172. doi: 10.1002/hlca.19750580729
- ^ H. Paulsen, H. Behre (1969): "Cyclorearrangement of 3,4,5-Tri-O-acetyl-(1,2,4/3,5)-cyclopentanepentol-1,2-O-acetoxonium Tetrafluoroborate". Angewandte chemie - International Edition, volume 8, issue 11, pages 887–887. doi: 10.1002/anie.196908871
- ^ Marc Felemez, Gilbert Schlewer, David J. Jenkins, Vanessa Correa, Colin W. Taylor, Barry V. L. Potter, and Bernard Spiess (1999): "Inframolecular studies of the protonation of 1D-1,2,4/3,5-cyclopentanepentaol 1,3,4-trisphosphate, a ring-contracted analogue of 1D-myo-inositol 1,4,5-trisphosphate". Carbohydrate Research, volume 322, issues 1–2, pages 95-101. doi: 10.1016/S0008-6215(99)00193-7
- ^ Laren M. Tolbert, J. Carlin Gregory, Carolyn P. Brock (1985): "cis,cis,cis,cis-1,2,3,4,5-Pentakis(hydroxymethyl)-cyclopentane". Journal of Organic Chemistry, volume 50, issue 4, pages 548-548. doi: 10.1021/jo00204a030
- ^ G. Hölzl, P. Dörmann(2007): "Structure and function of glycoglycerolipids in plants and bacteria". Progress in Lipid Research, volume 46, issue 5, pages 225-243. PMID 17599463 doi: 10.1016/j.plipres.2007.05.001
- ^ V. Costantino, E. Fattorusso, and A. Mangoni (1993): "Isolation of five-membered cyclitol glycolipids, crasserides: unique glycerides from the sponge Pseudoceratina crassa" Journal of Organic Chemistry, volume 58, issue 1, pages 186–191. doi: 10.1021/jo00053a034
- ^ Jun'Ichi Kobayashi, Chun-Min Zeng and Masami Ishibashi (1993): "Keruffaride, a new all-ciscyclopentanepentol-containing metabolite from the okinawan marine sponge luffariella sp.". Journal of the Chemical Society, Chemical Communications, volume 1993, issue 1, pages 79–81. doi: 10.1039/C39930000079
- ^ Masami Ishibashi, Chun-min Zeng, Jun'ichi Kobayashi (1993): "Keruffaride: Structure Revision and Isolation from Plural Genera of Okinawan Marine Sponges". Journal of Natural Products, volume 56, issue 10, pages 1856-1860. doi: 10.1021/np50100a033
- ^ E. Fattorusso, A. Mangoni (1997) "Marine Glycolipids". In Fortschritte der Chemie organischer Naturstoffe / Progress in the Chemistry of Organic Natural Products, volume 72, pp 215-301. doi: 10.1007/978-3-7091-6527-0_3 ISBN 978-3-7091-7342-8
- ^ Dong-Kyoo Kim, Young Ja Lim. Jung Sun Kim, Jong Hee Park, Nam Deuk Kim, Kwang Sik Im, Jongki Hong, Jee H. Jung (1999) "A Cyclitol Derivative as a Replication Inhibitor from the Marine Sponge Petrosia sp. Journal of Natural Products, volume 62, issue 5, pages 773-776. doi: 10.1021/np9804785
- ^ Yonghong Liu, Jee H. Jung, Tunhai Xu, Lijuan Long, Xiuping Lin, Hao Yin, Bin Yang, Xue-Feng Zhou, and Xianwen Yang (2011): "New cyclitol derivative from a sponge Sarcotragus species". Natural Product Research, volume 25, issue 6, pages 648–652. doi: 10.1080/14786419.2010.490213
- ^ Blériot, Yves; Untersteller, Edouard; Fritz, Benoît; Sinaÿ, Pierre (2001). "Total Synthesis of Calditol: Structural Clarification of this Typical Component of Archaea Order Sulfolobales". Chemistry: A European Journal. 8 (1): 240–246. doi: 10.1002/1521-3765(20020104)8:1<240::AID-CHEM240>3.0.CO;2-G. PMID 11822455.
- ^ William W. Christie (2019): " Di- and Tetra-Alkyl Ether Lipids of the Archaea". Article on the LipidWeb site, accessed on 2019-07-25.
- ^ F. T. D. Bothon, J. Adovelande, F. Cazier, F. Avlessi, and D. C. K. Sohounhloue (2016): " Phytochemical Analysis and Medecinal Evaluation of Hydro Ethanolic Extract of Two Varieties Hibiscus sabdariffa Calyxes". International Journal of Green and Herbal Chemistry, Green Chemistry series, volume 5, issue 3, pages 349-354. ISSN 2278-3229 CODEN IJGHAY
- ^ V. V. Platonov, A. A. Khadartsev L. N. Belozerova (2017): " Хромато-масс-спектрометрия спиртового экстракта маклюры" ("Chromato-Mass-Spectrometry of Alcohol Extraction of Maclura"). Вестник новых медицинских технологий (Journal of New Medical Technologies), electronic edition, volume 2017, issue 2. UDC: 581.1 doi: 10.12737/article_5909a300d1fcc8.29871271
- ^ Antony V.Samrot and T.Thirunalasundari (2012): "[wprim.whocc.org.cn/admin/article/downloadAppendix?article=/upload/articleFile/P020160926333045048197.pdf&articleId=625657 Effect of Oscillatoria willei – a marine cyanobacterium on hydrazine induced toxicity]". Malaysian Journal of Microbiology, volume 8, issue 4, pages 229-234. ISSN 1823-8262
- ^ Harshal D. Kawale and Nanda Kishore (2019): "Production of hydrocarbons from a green algae (Oscillatoria) with exploration of its fuel characteristics over different reaction atmospheres". Energy, volume 178, pages 344-355. doi: 10.1016/j.energy.2019.04.103
- ^ T. Posternak, and G. Wolczunovicz (1968): "Two new cyclopentanepentols". Naturwissenschaften, volume 55, issue 2, pages 82–83. doi: 10.1007/BF00599491

| |
| Names | |
|---|---|
|
Preferred IUPAC name
Cyclopentane-1,2,3,4,5-pentol | |
| Other names
1,2,3,4,5-Cyclopentanepentol
1,2,3,4,5-Pentahydroxycyclopentane | |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| Properties | |
| C5H10O5 | |
| Molar mass | 150.130 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
1,2,3,4,5-Cyclopentanepentol, also named cyclopentane-1,2,3,4,5-pentol or 1,2,3,4,5-pentahydroxycyclopentane is a
chemical compound with formula C
5H
10O
5 or (–CHOH–)
5, whose
molecule consists of a ring of five
carbon atoms (a
cyclopentane skeleton), each connected to one
hydrogen and one
hydroxyl
group.
[1] The unqualified term "cyclopentanepentol" usually refers to this compound. There are four distinct
stereoisomers with this same structure.
[2]
The compound is a five-fold alcohol of cyclopentane, and technically a cyclic sugar alcohol (a cyclitol). However it is very rarely found in nature, and therefore it has received much less attention than the ubiquitous six-carbon version, inositol. [3]
There are four distinct stereoisomers of cyclopentane-1,2,3,4,5-pentol, distinguished by the position of the hydroxyls relative to the mean plane of the ring. All have a plane of symmetry, and therefore are not chiral. One naming convention for the isomers labels each carbon number 1-5 with "α" for the side of the ring plane with most hydroxyls (three or more), and "β" for the other side. Another convention lists the hydroxyls on the majority side and then those on the minority side, with the two groups separated by a slash (and a "0" is written when the second list would be empty). [4]
The four possible isomers are: [2]
- 1α,2α,3α,4α,5α, 1,2,3,4,5/0, or all-cis (all hydroxyls on the same side). CAS 34322-89-9. Long needles, melting point 283 °C (darkens ar 220 °C). [2] [5] Obtained from cyclopentane-1α,2α-epoxi-3α,4α,5α-triol by treatment with hydrogen bromide to obtain cyclopentane-1β-bromo-2α,3α,4α,5α-tetrol, followed by benzoylation and treatment with DMSO and sodium bicarbonate. [5]
- 1α,2α,3α,4α,5β or 1,2,3,4/5. CAS 18939-02-1. [2] Soluble in hot ethanol. [3] Melting point 220 °C [3] or 209-210 °C. [6] Obtained by solvolysis of two tetra-O-acetyl-O- tosyl-cyclopentanepentol, [3] and also by hydroxylation of 3,4,5-trihydroxy-1-cyclopentenes. [6]
- 1α,2α,3α,4β,5β or 1,2,3/4,5. CAS 18939-07-6. [2] Soluble in ethanol. [3] Melting point 168-169 °C, [3] or 176 °C. [6] Obtained by deamination of (1,4/2,3,5)-5-amino-1,3-di-O-acetyl-2,3-O-cyclohexylidene-l,2,3,4-cycopentanetetrol, [3] and also by hydroxylation of 3,4,5-trihydroxy-1-cyclopentenes. [6]
- 1α,2α,3β,4α,5β or 1,2,4/3,5. CAS 57784-52-8. [2] Melting point 149.5-150.5 °C. [3] Obtained from cyclopent-4-ene-1,3/2-trio1 by epoxidation and subsequent hydrolysis; [7] also by acid hydrolysis of an anhydro-cyclopentanepentol, DL-1,2-anhydro-4,5-O-cyclohexylidene-1,2,3/4,5-cyclopentanepentol; [3] and also from a D-xylo-pento dialdose. [8]
The last three isomers interconvert by heating them at about 104 °C with 95% acetic acid in the presence of a strong acid. The conversion between 1,2,3,4/5 and 1,2,3/4,5 is faster and entails the hydrogen and hydroxyl switching places on either of the two carbon atoms located between an "α" and a "β" position. The formation of 1,2,4/3,5 is much slower and equilibrium is reached only after many days. The relative stabilities are 1,2,4/3,5 > 1,2,3/4,5 > 1,2,3,4/5, with equilibrium ratios 72 : 17.5 : 10.5. [3]
The isomers can be identified qualitatively by their mobility in paper ionophoresis in a solution of calcium acetate and acetic acid, and revealed with a manganese sulfate/ potassium permanganate reagent. The mobilities of the isomers, relative to that of cis-inositol, are 0.95 (12345/0), 0.44 (1234/5), 0.18 (123/45), and 0.04 (124/35). The variation is attributed to the formation of chelates with the calcium cations, with varying strengths depending on the number of hydroxyl pairs that can bind to the cation. [9] Similar results can be obtained with lanthanum cations and thin-layer chromatography. [10]
Mass spectrometry of the compound generates mainly the ion HO–CH=CH=CH=O+H (mass-charge ratio 73) and a neutral radical C
2H
5O•.
[11]
Many esters are known, such as penta benzoyls [2] and penta acetyls. [3]
Trteatment of the derivative 3,4,5-tri-O-acetyl-1,2-O-ethylidene-(1,2,4/3,5)-cyclopentanepentol with
triphenylcarbenium
tetrafluoroborate [(C
6H
5)
3C]+
[BF
4−
to yield the cation 3,4,5-tri-O-acetyl-(1,2,4/3,5)-cyclopentanepentol-1,2-O-acetoxonium, which exhibits a peculiar 10-stage cyclic rearrangement.
[12]
Cyclopentanepentols form dihydrogen phosphate esters analogous to inositol's phytic acid esters. [13]
The related compound 1,2,3,4,5-pentakis(hydroxymethyl)-cyclopentane (a colourless viscous oil, soluble in tert-butanol and DMSO) was synthesized in 1985 by L. M. Tolbert and others. It was hoped that dehydrative dimerization of this product would yield dodecahedrane. [14]
Organic compounds similar to (and possibly mimicking) glycoglycerolipids, [15] with a 1,2,4/3,5-cyclopentanepentol group connected to the glycerol core in place of the sugar group, have been found in several genera of marine sponges with widely separate ranges. [16] [17] [18] [19] [20] [21]
Calditol is a substance found in the archaeobacteria of the order Sulfolobales. It is an ether that can be viewed as the condensation of glycerol and 3α-hydroxymethyl-1α,2α,3β,4α,5β-hydroxy cyclopentane, forming an ether bridge –O– between the 1-carbon of the former and the 4-carbon of the latter. [22] Its seems to protects the cells in acidic environments. [23]
Trace amounts of cyclopentanepentol were identified in some plants, such as the red flower calyces of Hibiscus sabdariffa (roselle); [24] the plant Maclura pomifera (Osage orange); [25] and the cyanobacterium Oscillatoria willei [26] as well as in the products of its hydropyrolysis. [27]
The 1,2,4/3,5 isomer was briefly described in 1963 by H. Z. Sable and others. [7] In 1968, Th. Posternak reported observation of 1,2,3,4/5 and 1,2,3/4,5, without the synthesis method. [28] Better synthesis methods for these three were published by S. J. Angyal and B. M. Luttrell in 1970. [3] Alternative methods for 1,2,3,4/5 and 1,2,3/4,5 were published in the same year by G. Wolczunowicz and others. [6] The remaining all-cis isomer (1,2,3,4,5/0) was synthesized in 1971 by F. G. Cocu and Posternak. [5]
- ^ (2019) Entry 552295: 1,2,3,4-Cyclopentanepentol. Page at the PubChem website, accessed on 2019-07-23.
- ^ a b c d e f g Peter M. Collins (2005): Dictionary of Carbohydrates, 2nd edition, page 268. CRC Press, 1304 pages. ISBN 9780849377655
- ^ a b c d e f g h i j k l S. J. Angyal and B. M. Luttrell (1970): "Cyclitols. XXXII. Cyclopentanepentols". Australian Journal of Chemistry, volume 23, issue 9, pages 1831-1838. doi: 10.1071/CH9701831
- ^ CON and CBN IUPAC Commissions on Nomenclature (1968): "The Nomenclature of Cyclitols - Tentative Rules". European Journal of Biochemistry, volume 5, pages 1-12. doi: 10.1111/j.1432-1033.1968.tb00328.x
- ^ a b c F. G. Cocu, Th. Posternak (1971): "Recherches dans la série des cyclitols XLII. Synthèses de cyclitols tout-cis dérivés du cyclopentane; préparation des cyclopentane-tétrol et - pentol tout-cis" Helvetica Chimica Acta, volume 54, issue 6, pages 1676-1687. doi: 10.1002/hlca.19710540619
- ^ a b c d e G. Wolczunowicz, L. Bors, F. Cocu, Th. Posternak (1970): "Recherches dans la série des cyclitols, XLI. Synthèses de deux cyclopentane-pentols". Helvetica Chimica Acta, volume 53, issue 8, pages 2288-2295. doi: 10.1002/hlca.19700530843
- ^ a b H. Z. Sable, Thelma Anderson, Bernadine Tolbert, Th. Posternak (1963): "Recherches dans la série des cyclitols XXXIII. Sur quelques cyclitols dérivés du cyclopentane". Helvetica Chimica Acta, volume 46, issue 4, pages 1157–1165. doi: 10.1002/hlca.19630460410
- ^ Barry V. L. Potter (1998) "Structure-Activity Relationships of Adenophostin A and Related Molecules at the 1-D-myo-Inositol 1,4,5-Trisphosphate Receptor". American Chemical Society Symposium Series, volume 718, chaper 10, pages 158-179 doi: 10.1021/bk-1999-0718.ch010
- ^ S. J. Angyal and J. A. Mills (1979): "Complexes of Carbohydrates with Metal Cations. XI. Paper Electrophoresis of Polyols in Solutions of Calcium Ions". Australian Journal of Chemistry, volume 32, issue 9, pages 1993–2001. doi: 10.1071/CH9791993
- ^ S. J. Angyal and J. A. Mills (1985): "Complexes of Carbohydrates with Metal Cations. XIV. Separation of Sugars and Alditols by Means of Their Lanthanum Complexes". Australian Journal of Chemistry, volume 38, issue 8, pages 1279–1285. doi: 10.1071/CH9851279
- ^ Gérard A. Singy and Armand Buchs (1975): "Etude par spectrométrie de masse de la fragmentation des cyclopentane-triols-1,2,3, des cyclopentane-tétrols et des cyclopentane-pentols". Helvetica Chimica Acta, volume 58, issue 7, pages 2164-2172. doi: 10.1002/hlca.19750580729
- ^ H. Paulsen, H. Behre (1969): "Cyclorearrangement of 3,4,5-Tri-O-acetyl-(1,2,4/3,5)-cyclopentanepentol-1,2-O-acetoxonium Tetrafluoroborate". Angewandte chemie - International Edition, volume 8, issue 11, pages 887–887. doi: 10.1002/anie.196908871
- ^ Marc Felemez, Gilbert Schlewer, David J. Jenkins, Vanessa Correa, Colin W. Taylor, Barry V. L. Potter, and Bernard Spiess (1999): "Inframolecular studies of the protonation of 1D-1,2,4/3,5-cyclopentanepentaol 1,3,4-trisphosphate, a ring-contracted analogue of 1D-myo-inositol 1,4,5-trisphosphate". Carbohydrate Research, volume 322, issues 1–2, pages 95-101. doi: 10.1016/S0008-6215(99)00193-7
- ^ Laren M. Tolbert, J. Carlin Gregory, Carolyn P. Brock (1985): "cis,cis,cis,cis-1,2,3,4,5-Pentakis(hydroxymethyl)-cyclopentane". Journal of Organic Chemistry, volume 50, issue 4, pages 548-548. doi: 10.1021/jo00204a030
- ^ G. Hölzl, P. Dörmann(2007): "Structure and function of glycoglycerolipids in plants and bacteria". Progress in Lipid Research, volume 46, issue 5, pages 225-243. PMID 17599463 doi: 10.1016/j.plipres.2007.05.001
- ^ V. Costantino, E. Fattorusso, and A. Mangoni (1993): "Isolation of five-membered cyclitol glycolipids, crasserides: unique glycerides from the sponge Pseudoceratina crassa" Journal of Organic Chemistry, volume 58, issue 1, pages 186–191. doi: 10.1021/jo00053a034
- ^ Jun'Ichi Kobayashi, Chun-Min Zeng and Masami Ishibashi (1993): "Keruffaride, a new all-ciscyclopentanepentol-containing metabolite from the okinawan marine sponge luffariella sp.". Journal of the Chemical Society, Chemical Communications, volume 1993, issue 1, pages 79–81. doi: 10.1039/C39930000079
- ^ Masami Ishibashi, Chun-min Zeng, Jun'ichi Kobayashi (1993): "Keruffaride: Structure Revision and Isolation from Plural Genera of Okinawan Marine Sponges". Journal of Natural Products, volume 56, issue 10, pages 1856-1860. doi: 10.1021/np50100a033
- ^ E. Fattorusso, A. Mangoni (1997) "Marine Glycolipids". In Fortschritte der Chemie organischer Naturstoffe / Progress in the Chemistry of Organic Natural Products, volume 72, pp 215-301. doi: 10.1007/978-3-7091-6527-0_3 ISBN 978-3-7091-7342-8
- ^ Dong-Kyoo Kim, Young Ja Lim. Jung Sun Kim, Jong Hee Park, Nam Deuk Kim, Kwang Sik Im, Jongki Hong, Jee H. Jung (1999) "A Cyclitol Derivative as a Replication Inhibitor from the Marine Sponge Petrosia sp. Journal of Natural Products, volume 62, issue 5, pages 773-776. doi: 10.1021/np9804785
- ^ Yonghong Liu, Jee H. Jung, Tunhai Xu, Lijuan Long, Xiuping Lin, Hao Yin, Bin Yang, Xue-Feng Zhou, and Xianwen Yang (2011): "New cyclitol derivative from a sponge Sarcotragus species". Natural Product Research, volume 25, issue 6, pages 648–652. doi: 10.1080/14786419.2010.490213
- ^ Blériot, Yves; Untersteller, Edouard; Fritz, Benoît; Sinaÿ, Pierre (2001). "Total Synthesis of Calditol: Structural Clarification of this Typical Component of Archaea Order Sulfolobales". Chemistry: A European Journal. 8 (1): 240–246. doi: 10.1002/1521-3765(20020104)8:1<240::AID-CHEM240>3.0.CO;2-G. PMID 11822455.
- ^ William W. Christie (2019): " Di- and Tetra-Alkyl Ether Lipids of the Archaea". Article on the LipidWeb site, accessed on 2019-07-25.
- ^ F. T. D. Bothon, J. Adovelande, F. Cazier, F. Avlessi, and D. C. K. Sohounhloue (2016): " Phytochemical Analysis and Medecinal Evaluation of Hydro Ethanolic Extract of Two Varieties Hibiscus sabdariffa Calyxes". International Journal of Green and Herbal Chemistry, Green Chemistry series, volume 5, issue 3, pages 349-354. ISSN 2278-3229 CODEN IJGHAY
- ^ V. V. Platonov, A. A. Khadartsev L. N. Belozerova (2017): " Хромато-масс-спектрометрия спиртового экстракта маклюры" ("Chromato-Mass-Spectrometry of Alcohol Extraction of Maclura"). Вестник новых медицинских технологий (Journal of New Medical Technologies), electronic edition, volume 2017, issue 2. UDC: 581.1 doi: 10.12737/article_5909a300d1fcc8.29871271
- ^ Antony V.Samrot and T.Thirunalasundari (2012): "[wprim.whocc.org.cn/admin/article/downloadAppendix?article=/upload/articleFile/P020160926333045048197.pdf&articleId=625657 Effect of Oscillatoria willei – a marine cyanobacterium on hydrazine induced toxicity]". Malaysian Journal of Microbiology, volume 8, issue 4, pages 229-234. ISSN 1823-8262
- ^ Harshal D. Kawale and Nanda Kishore (2019): "Production of hydrocarbons from a green algae (Oscillatoria) with exploration of its fuel characteristics over different reaction atmospheres". Energy, volume 178, pages 344-355. doi: 10.1016/j.energy.2019.04.103
- ^ T. Posternak, and G. Wolczunovicz (1968): "Two new cyclopentanepentols". Naturwissenschaften, volume 55, issue 2, pages 82–83. doi: 10.1007/BF00599491